Advanced Synthesis of Alpha Beta Deuterated Amines for Pharmaceutical Applications
The pharmaceutical industry is increasingly recognizing the strategic value of deuterated drugs, which offer improved metabolic stability and pharmacokinetic profiles compared to their non-deuterated counterparts. Patent CN112430187A introduces a groundbreaking methodology for the synthesis of alpha,beta-deuterated amine compounds, addressing critical limitations in existing technologies. This innovation provides a robust pathway for producing high-purity pharmaceutical intermediates essential for modern drug discovery and development. The core of this technology lies in a sequential two-step process: first, the conversion of nitrile compounds into alpha-deuterated nitrile intermediates using a deuterium donor and base, followed by a reductive deuteration step utilizing a divalent lanthanide transition metal compound. This approach effectively solves the historical challenges of low deuteration rates and poor regioselectivity associated with prior art methods.
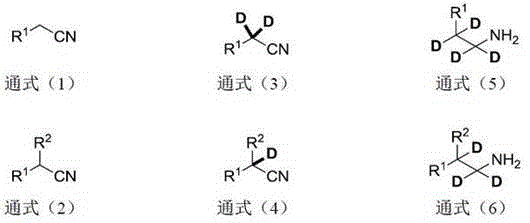
For R&D directors and process chemists, understanding the mechanistic superiority of this route is paramount. The method described in the patent circumvents the need for harsh reaction conditions often required by traditional H/D exchange processes. By leveraging the unique reactivity of lanthanide reagents, specifically samarium diiodide (SmI2) or similar divalent species, the process achieves selective reduction of the carbon-nitrogen triple bond while preserving the newly installed deuterium atoms at the alpha position. This precision is crucial for maintaining the integrity of the isotopic label throughout the synthesis, ensuring that the final active pharmaceutical ingredient (API) retains the desired metabolic advantages. The ability to operate under argon protection at room temperature further underscores the operational simplicity and safety profile of this novel synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,beta-deuterated amines has been plagued by significant technical and economic hurdles. Prior art methods, such as those catalyzed by rubidium complexes, often necessitate extreme reaction conditions including high temperatures and elevated pressures, which pose safety risks and increase energy consumption. Furthermore, these traditional transition metal catalysts are frequently expensive, toxic, and highly sensitive to air and moisture, complicating handling and storage requirements in a manufacturing setting. A major drawback of these conventional routes is their poor regioselectivity, leading to random H/D exchange at unwanted positions and resulting in a complex mixture of isotopologues that are difficult and costly to separate. Additionally, the low product yields and suboptimal deuteration rates observed in older methodologies directly impact the overall cost of goods and the feasibility of scaling these processes for commercial production.
The Novel Approach
In stark contrast, the methodology disclosed in CN112430187A offers a streamlined and efficient alternative that directly addresses these pain points. By employing a mild base-catalyzed exchange followed by lanthanide-mediated reduction, the new process achieves high regioselectivity, targeting specifically the alpha and beta positions relative to the amine group without affecting other sensitive functional groups on the molecule. The reaction conditions are remarkably gentle, typically proceeding at room temperature within a timeframe of 15 minutes to 72 hours depending on the substrate, which significantly reduces thermal stress on the equipment and the product. The use of divalent lanthanide compounds allows for a clean transformation with minimal side reactions, thereby simplifying the downstream purification process. This novel approach not only enhances the chemical purity of the final product but also aligns with green chemistry principles by avoiding toxic heavy metal contaminants and reducing waste generation.
Mechanistic Insights into Lanthanide-Catalyzed Reductive Deuteration
The success of this synthetic route hinges on the precise control of deuterium incorporation through a well-defined mechanistic pathway. In the first stage, the nitrile substrate undergoes deprotonation at the alpha-position by a strong base in the presence of a deuterium source, such as heavy water or deuterated alcohols. This equilibrium process facilitates the exchange of hydrogen for deuterium, generating an alpha-deuterated nitrile intermediate with high isotopic enrichment. The subsequent addition of the divalent lanthanide reagent initiates a single-electron transfer mechanism that reduces the nitrile group to an imine or amine species. Crucially, the reaction environment is controlled to prevent the loss of the labile deuterium atoms during this reduction phase. The lanthanide center coordinates with the nitrogen atom, activating the triple bond for nucleophilic attack by deuterium species present in the medium, ultimately yielding the fully saturated alpha,beta-deuterated amine.
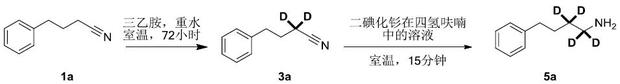
Impurity control is another critical aspect where this mechanism excels. Because the reaction proceeds under mild conditions without the need for high-energy activation, the formation of thermal degradation products is minimized. The high chemoselectivity of the lanthanide reagent ensures that other reducible functional groups, such as esters or halides present on the aromatic ring, remain intact, as demonstrated in the synthesis of various substituted benzylamines. For instance, the patent details the successful conversion of substrates containing methoxy groups and heterocycles without compromising the integrity of these moieties. This level of control is essential for pharmaceutical applications where impurity profiles are strictly regulated. The ability to quench the reaction simply by introducing air further simplifies the workup, allowing for the rapid isolation of the product with high purity, thus reducing the burden on analytical quality control teams.
How to Synthesize Alpha Beta Deuterated Amines Efficiently
The practical implementation of this technology involves a straightforward protocol that can be easily adapted for scale-up. The process begins with the preparation of the reaction vessel under an inert atmosphere to exclude moisture and oxygen, which could deactivate the sensitive lanthanide catalyst. Following the addition of the base and deuterium donor, the nitrile substrate is introduced, and the mixture is allowed to stir for the requisite period to ensure complete alpha-deuteration. Once the intermediate is formed, the lanthanide solution is added dropwise to control the exotherm and ensure uniform mixing. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and quenching procedures, are outlined below to guide process engineers in replicating these high-yielding transformations.
- React nitrile compounds with a deuterium donor and base under argon protection at room temperature for 24-72 hours to form alpha-deuterated nitrile intermediates.
- Add a divalent lanthanide transition metal compound solution (e.g., SmI2) in an organic solvent to the reaction mixture.
- Stir the mixture, quench with air, extract with organic solvents, and purify to obtain the final alpha,beta-deuterated amine compound or its salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic methodology offers substantial strategic benefits that extend beyond mere technical performance. The shift towards milder reaction conditions and the elimination of expensive noble metal catalysts translate directly into reduced operational expenditures and a more resilient supply chain. By utilizing readily available lanthanide reagents and common organic solvents, manufacturers can mitigate the risks associated with the volatility of precious metal markets. Furthermore, the simplified workup and purification requirements mean that production cycles can be shortened, enhancing the overall throughput of the facility. This efficiency is critical for meeting the tight deadlines often imposed by drug development timelines, ensuring that key intermediates are available when needed for preclinical and clinical studies.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of costly transition metal catalysts with more affordable lanthanide alternatives. Traditional methods often require stoichiometric amounts of expensive metals or specialized ligands, whereas this method utilizes catalytic or near-stoichiometric amounts of cheaper reagents. Additionally, the high yields reported, often exceeding 90%, minimize the loss of valuable starting materials and reduce the volume of waste that requires disposal. The ability to run reactions at room temperature also eliminates the energy costs associated with heating or cooling large reactors, contributing to a lower carbon footprint and reduced utility bills for the manufacturing site.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of robust and commercially available reagents. Unlike air-sensitive catalysts that require specialized shipping and storage conditions, the reagents used in this process are easier to handle and source globally. This reduces the lead time for raw material procurement and minimizes the risk of production delays due to supply shortages. The scalability of the process, demonstrated by its successful application to a wide range of substrates from simple alkyl nitriles to complex heterocycles, ensures that suppliers can reliably meet increasing demand as a drug candidate progresses through the pipeline. This reliability is a key factor for procurement managers looking to secure long-term partnerships for critical API intermediates.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns well with modern regulatory standards and corporate sustainability goals. The absence of toxic heavy metals simplifies the treatment of effluent streams, reducing the cost and complexity of wastewater management. The high atom economy of the reductive deuteration step ensures that most of the reactant mass is incorporated into the final product, minimizing waste generation. Furthermore, the mild conditions reduce the risk of runaway reactions, enhancing plant safety and lowering insurance premiums. These factors combined make the process highly attractive for commercial scale-up, allowing companies to transition smoothly from gram-scale laboratory synthesis to multi-kilogram or ton-scale production without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this deuteration technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for stakeholders evaluating this method for their own manufacturing needs. Understanding these details is essential for making informed decisions about process adoption and supplier selection.
Q: What are the advantages of this deuteration method over traditional transition metal catalysis?
A: Unlike traditional methods using expensive and toxic transition metals like rubidium which require high temperature and pressure, this method utilizes divalent lanthanides under mild room temperature conditions, offering higher regioselectivity and deuteration rates without toxic by-products.
Q: Can this method be applied to complex drug molecules?
A: Yes, the patent demonstrates successful application in synthesizing deuterated versions of complex bioactive molecules such as dopamine, berberine, yohimbine, melatonin, and serotonin, proving its versatility for pharmaceutical intermediate manufacturing.
Q: What represents the typical yield and deuteration rate for this process?
A: Experimental data indicates high efficiency, with yields reaching up to 97% and deuteration rates at both alpha and beta positions exceeding 90% in optimized examples, ensuring high isotopic purity for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha Beta Deuterated Amine Supplier
As the demand for deuterated drugs continues to surge, partnering with a knowledgeable and capable CDMO is essential for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from early-stage development to full-scale manufacturing. Our team is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of alpha,beta-deuterated amine meets the highest standards required for pharmaceutical applications. We understand the nuances of isotopic labeling and are committed to delivering products with superior deuteration rates and chemical purity.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this method can optimize your budget without compromising quality. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with reliable, high-quality deuterated intermediates.
