Advanced Rhodium-Catalyzed Selective Benzylic Deuteration for Commercial Scale API Manufacturing
Introduction to Next-Generation Deuteration Technology
The strategic incorporation of deuterium into pharmaceutical molecules has emerged as a critical strategy for optimizing pharmacokinetic profiles, specifically by leveraging the kinetic isotope effect to enhance metabolic stability. Patent CN113563147B discloses a groundbreaking method for the selective deuteration of benzylic carbon-hydrogen bonds in aromatic rings, addressing a significant gap in current synthetic methodologies. This technology utilizes a sophisticated rhodium metal catalyst system to achieve eta-6 coordination activation, enabling the precise exchange of hydrogen and deuterium at the benzylic position without compromising the integrity of the aromatic ring or other sensitive functional groups. The process operates under relatively mild thermal conditions and avoids the use of harsh strong acids or bases, which are often detrimental to complex drug scaffolds. By utilizing commercially available deuterated reagents as the deuterium source, this method offers a cost-effective and universally applicable solution for the synthesis of deuterated intermediates. 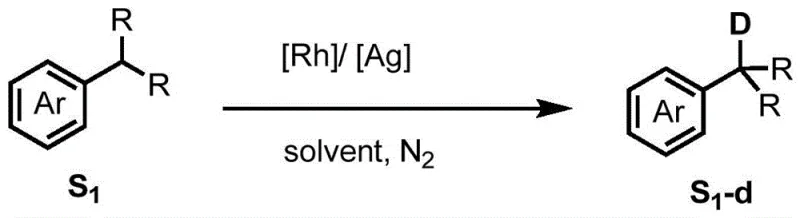 This innovation represents a substantial leap forward for reliable pharmaceutical intermediate supplier networks seeking to integrate deuterated analogs into their pipelines with high efficiency and minimal process development time.
This innovation represents a substantial leap forward for reliable pharmaceutical intermediate supplier networks seeking to integrate deuterated analogs into their pipelines with high efficiency and minimal process development time.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzylic deuterated compounds has relied heavily on two primary strategies, both of which suffer from significant drawbacks that hinder their application in modern drug discovery and manufacturing. The first approach involves starting from commercially available deuterated raw materials and constructing the target molecule through multi-step synthesis; however, this route is often cumbersome, economically inefficient due to the high cost of labeled starting materials, and results in low overall yields after multiple transformations. The second conventional method attempts direct hydrogen-deuterium exchange on non-labeled precursors, frequently employing strong bases such as potassium tert-butoxide in DMSO to abstract the benzylic proton. While conceptually simple, this base-mediated approach exhibits poor functional group compatibility, limiting its utility to very simple molecular architectures and failing completely when sensitive moieties are present. Furthermore, transition metal-catalyzed methods using palladium, ruthenium, or nickel have been explored, but they often struggle with selectivity issues, inadvertently deuterating sp2 carbon-hydrogen bonds on the aromatic ring alongside the desired benzylic position, thereby complicating purification and isotopic purity analysis.
The Novel Approach
In stark contrast to these legacy techniques, the novel rhodium-catalyzed approach described in the patent data introduces a mechanism driven by eta-6 coordination activation that fundamentally alters the reactivity landscape of the substrate. By coordinating the rhodium center to the aromatic ring in an eta-6 fashion, the acidity of the adjacent benzylic C-H bond is dramatically enhanced, creating a thermodynamic and kinetic preference for exchange at this specific site over the aromatic protons. This intrinsic selectivity allows for the direct deuteration of complex substrates bearing diverse functional groups, including ethers, esters, amines, and halides, without the need for protecting groups. The method utilizes a combination of a pentamethylcyclopentadienyl rhodium dichloride dimer derivative and a silver salt with weakly coordinating anions, which work synergistically to generate the active cationic rhodium species in situ. This catalytic system operates efficiently in deuterated methanol or acetone, providing a streamlined, one-pot procedure that delivers high deuterium incorporation rates and excellent isolated yields, effectively overcoming the selectivity and compatibility barriers that have long plagued the field of benzylic deuteration.
Mechanistic Insights into Rh-Catalyzed Eta-6 Coordination Activation
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the rhodium catalyst, which distinguishes it from standard electrophilic or radical substitution processes. Upon activation by the silver salt, the rhodium center engages in eta-6 coordination with the aromatic pi-system of the substrate, temporarily disrupting the aromaticity and significantly increasing the electron-withdrawing character of the ring system. This electronic perturbation renders the benzylic protons substantially more acidic than they would be in the free substrate, allowing for facile deprotonation or sigma-bond metathesis with the deuterated solvent. 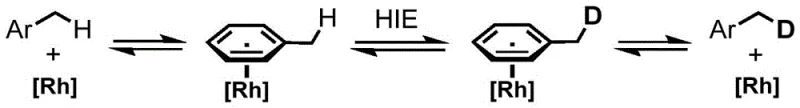 The resulting organometallic intermediate then undergoes rapid exchange with the deuterium source, followed by reductive elimination or protonolysis to release the deuterated product and regenerate the active catalyst species. This cycle ensures that the deuteration is confined strictly to the benzylic position, as the aromatic C-H bonds remain kinetically inert under these specific coordination conditions. The choice of ligand on the rhodium center is critical, with pentamethylcyclopentadienyl derivatives proving particularly effective due to their ability to stabilize the high-valent metal center while maintaining sufficient lability for substrate binding.
The resulting organometallic intermediate then undergoes rapid exchange with the deuterium source, followed by reductive elimination or protonolysis to release the deuterated product and regenerate the active catalyst species. This cycle ensures that the deuteration is confined strictly to the benzylic position, as the aromatic C-H bonds remain kinetically inert under these specific coordination conditions. The choice of ligand on the rhodium center is critical, with pentamethylcyclopentadienyl derivatives proving particularly effective due to their ability to stabilize the high-valent metal center while maintaining sufficient lability for substrate binding.  Furthermore, the presence of weakly coordinating anions from the silver salt prevents catalyst poisoning and ensures high turnover numbers, making the process robust enough for scale-up. Understanding this mechanism is vital for R&D teams aiming to predict substrate scope and optimize reaction parameters for new chemical entities, as it provides a rational basis for modifying reaction conditions to accommodate steric or electronic variations in the target molecule.
Furthermore, the presence of weakly coordinating anions from the silver salt prevents catalyst poisoning and ensures high turnover numbers, making the process robust enough for scale-up. Understanding this mechanism is vital for R&D teams aiming to predict substrate scope and optimize reaction parameters for new chemical entities, as it provides a rational basis for modifying reaction conditions to accommodate steric or electronic variations in the target molecule.
Impurity control in this process is inherently managed by the high regioselectivity of the catalytic cycle, which minimizes the formation of ring-deuterated byproducts that are common in non-selective metal-catalyzed exchanges. The mild reaction conditions, typically ranging from 80°C to 140°C, further reduce the risk of thermal degradation or rearrangement of sensitive functional groups, which is a frequent concern in base-mediated deuteration protocols. Additionally, the use of deuterated solvents like methanol-d4 or acetone-d6 serves a dual purpose as both the reaction medium and the deuterium source, simplifying the workup procedure and reducing the potential for contamination with protic impurities. For chiral benzylic centers, the reaction conditions can be tuned to preserve stereochemical integrity, as evidenced by the successful deuteration of chiral pharmaceutical intermediates with retained enantiomeric ratios. This level of control over the impurity profile is essential for meeting the stringent regulatory requirements of pharmaceutical manufacturing, where isotopic purity and chemical purity are inextricably linked to the safety and efficacy of the final drug product.
How to Synthesize Benzylic Deuterated Compounds Efficiently
The practical implementation of this deuteration technology is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment, facilitating rapid adoption by process chemistry teams. The general protocol involves charging a reaction vessel with the aromatic substrate, a catalytic amount of the rhodium precursor (typically 2.5 to 10 mol%), and a stoichiometric or excess amount of the silver salt activator under an inert nitrogen atmosphere to prevent oxidation of the metal center. Following the addition of the deuterated solvent, the mixture is heated to the specified temperature, usually between 120°C and 140°C for less reactive substrates, and stirred for a period of 24 to 48 hours to ensure complete conversion. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results with precision.
- Prepare the reaction mixture by combining the aromatic substrate, rhodium catalyst (e.g., Cp*Rh derivative), and silver salt additive under a nitrogen atmosphere.
- Add the deuterated solvent (methanol-d4 or acetone-d6) and optional inorganic base if acetone is used, ensuring precise molar ratios.
- Heat the mixture to 80-140°C for 24-48 hours, then cool, concentrate, and purify via column chromatography to isolate the deuterated product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this rhodium-catalyzed deuteration method offers transformative benefits for procurement managers and supply chain directors focused on cost reduction in pharmaceutical intermediate manufacturing and operational efficiency. The reliance on commercially available starting materials and deuterated solvents eliminates the need for custom-synthesized labeled precursors, which are often subject to long lead times and exorbitant pricing, thereby significantly lowering the raw material cost baseline. The operational simplicity of the one-pot procedure reduces labor costs and minimizes the requirement for specialized equipment, as the reaction can be conducted in standard glass-lined or stainless steel reactors without the need for cryogenic conditions or high-pressure hydrogenation setups. Furthermore, the high selectivity of the process reduces the burden on downstream purification, lowering solvent consumption and waste generation associated with extensive chromatographic separations, which translates directly into improved process mass intensity and reduced environmental compliance costs.
- Cost Reduction in Manufacturing: The elimination of multi-step synthetic routes starting from expensive deuterated building blocks results in substantial cost savings, as the direct exchange method maximizes atom economy and reduces the number of unit operations required. By avoiding the use of strong bases and harsh reagents, the process also extends the lifespan of reactor vessels and reduces maintenance costs associated with corrosion, while the high catalyst turnover ensures that the loading of the precious metal rhodium remains economically viable even at large scales. The ability to use common deuterated solvents like methanol-d4, which are more affordable and easier to recover than specialized deuterated reagents, further enhances the economic attractiveness of this route for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The broad substrate scope and functional group tolerance of this method mitigate supply chain risks by allowing for the deuteration of advanced intermediates late in the synthesis sequence, reducing the inventory holding costs of valuable labeled materials. Since the reagents involved are stable and widely sourced, the risk of supply disruption due to raw material shortages is minimized, ensuring consistent production schedules for critical deuterated drug candidates. The robustness of the catalytic system against moisture and air, relative to other organometallic methods, simplifies logistics and storage requirements, making it easier to maintain a reliable supply of deuterated intermediates for clinical and commercial needs without complex cold chain management.
- Scalability and Environmental Compliance: The mild thermal conditions and absence of hazardous strong acids or bases make this process inherently safer and easier to scale from gram to kilogram quantities without significant re-optimization. The reduced generation of hazardous waste streams aligns with green chemistry principles, facilitating smoother regulatory approvals and reducing the environmental footprint of the manufacturing site. The high yields and selectivity observed across a diverse range of substrates, including complex drug molecules, demonstrate the versatility of the platform, allowing manufacturers to consolidate multiple deuteration projects onto a single, validated production line, thereby maximizing asset utilization and operational flexibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective benzylic deuteration technology, based on the detailed experimental data and scope provided in the patent literature. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this method for their specific development programs.
Q: What is the primary advantage of this Rh-catalyzed deuteration method over traditional base-mediated methods?
A: The primary advantage is superior regioselectivity. Traditional strong base methods often lack functional group tolerance and can lead to side reactions, whereas this Rh-catalyzed eta-6 coordination mechanism specifically activates the benzylic C-H bond, leaving the aromatic ring and other sensitive functional groups intact.
Q: Can this method be applied to complex drug molecules in late-stage functionalization?
A: Yes, the patent demonstrates excellent compatibility with complex biologically active molecules, including derivatives of fingolimod, estrone, and various heterocycles, making it highly suitable for late-stage deuteration of pharmaceutical candidates without requiring protection-deprotection strategies.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction typically proceeds under a nitrogen atmosphere at temperatures ranging from 80°C to 140°C using deuterated methanol or acetone as both solvent and deuterium source, with reaction times generally between 24 to 48 hours depending on substrate reactivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Intermediates Supplier
As the demand for deuterated pharmaceuticals continues to grow, partnering with an experienced CDMO capable of executing complex isotopic labeling strategies is essential for maintaining a competitive edge in drug development. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your deuterated intermediates are manufactured with the highest standards of quality and consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting isotopic enrichment and chemical purity requirements necessary for regulatory submission, leveraging advanced analytical techniques to verify deuterium incorporation at specific sites.
We invite you to engage with our technical procurement team to discuss how this innovative rhodium-catalyzed deuteration technology can be integrated into your supply chain to optimize your development timelines and budget. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your molecule, along with access to specific COA data and route feasibility assessments tailored to your project's unique challenges. Let us help you navigate the complexities of deuterated chemistry and secure a reliable supply of high-quality intermediates for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
