Advanced Rhodium-Catalyzed Synthesis of 4-Alkoxy Phenols for High-Value Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct critical structural motifs, particularly phenolic compounds which serve as foundational building blocks for numerous active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN113443970A, which discloses a novel synthesis method for 4-alkoxy phenol compounds. This technology represents a paradigm shift from traditional multi-step functionalization strategies to a streamlined, direct C-H bond activation approach. By utilizing a dimeric rhodium acetate catalyst in conjunction with a hypervalent iodine oxidant, this method achieves high regioselective direct hydroxylation of aryl alkyl ethers. The implications for manufacturing key drugs such as Monobenzone and Pramocaine are profound, offering a route that is not only chemically elegant but also industrially robust. For R&D directors and procurement specialists alike, understanding this technology is crucial for optimizing supply chains and reducing the environmental footprint of API production.
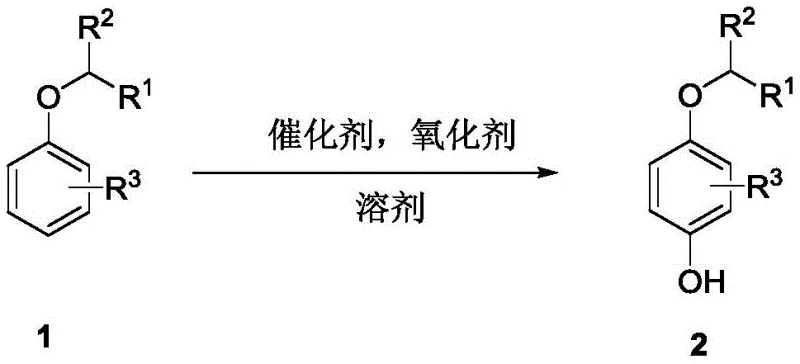
The transition from legacy synthetic methods to this novel rhodium-catalyzed protocol addresses several long-standing pain points in organic synthesis. Conventional methods for preparing 4-alkoxy phenols typically rely on the hydroxylation of halogenated aromatic hydrocarbons using transition metal catalysts like palladium or copper. These traditional routes are fraught with limitations: they require strict anhydrous and oxygen-free conditions, often necessitate harsh reagents such as strong bases at elevated temperatures, and are generally restricted to highly activated substrates like brominated or iodinated aromatics. Furthermore, the prerequisite halogenation step itself poses a challenge, as achieving high regioselectivity during the initial halogenation of complex aromatic hydrocarbons is difficult and generates significant stoichiometric waste. In contrast, the novel approach described in the patent bypasses the need for pre-functionalization entirely. By directly activating the inert carbon-hydrogen bond on the benzene ring, the process simplifies the synthetic sequence, improves atom economy, and operates under much milder conditions, typically around 80°C in trifluoroacetic anhydride.
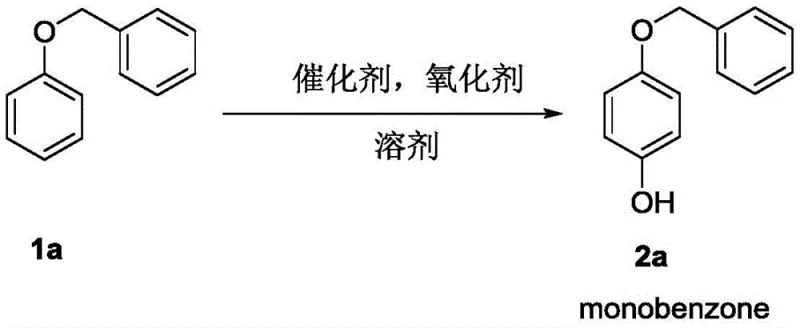
Mechanistically, this transformation relies on the unique ability of the rhodium catalyst to mediate the electrophilic activation of the aromatic ring in the presence of the hypervalent iodine oxidant, iodobenzene diethyl ester. The reaction exhibits exceptional regioselectivity, predominantly targeting the para-position relative to the alkoxy group, which is critical for producing high-purity intermediates required in drug synthesis. The catalytic system, comprising dimeric rhodium acetate and the oxidant in trifluoroacetic anhydride solvent, creates a reactive environment that facilitates the insertion of the hydroxyl group without disturbing other sensitive functional groups. This tolerance is evidenced by the successful conversion of substrates bearing esters, ketones, and various halogens (fluorine, chlorine, bromine, iodine) without significant side reactions or degradation. For the R&D team, this means a cleaner impurity profile and reduced downstream purification burdens. The mechanism avoids the formation of toxic heavy metal waste streams associated with stoichiometric copper or palladium reagents, aligning with modern green chemistry principles and simplifying the regulatory path for final drug approval.
How to Synthesize 4-Alkoxy Phenol Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory optimization and pilot plant operations. The procedure involves charging a sealed pressure-resistant tube with the aryl alkyl ether substrate, the rhodium catalyst, and the oxidant, followed by the addition of the solvent. The reaction is then heated, typically at 80°C, for a duration ranging from 1 to 10 hours depending on the specific substrate electronics. Post-reaction workup is straightforward, involving quenching with saturated sodium bicarbonate, extraction with ethyl acetate, and standard column chromatography purification. The detailed standardized synthesis steps for implementing this protocol in your facility are outlined below.
- Charge a sealed pressure tube with the aryl alkyl ether substrate, dimeric rhodium acetate catalyst, and iodobenzene diethyl ester oxidant.
- Add trifluoroacetic anhydride (TFAA) as the solvent and seal the tube tightly to prevent moisture ingress.
- Heat the reaction mixture to 80°C with magnetic stirring for 1 to 10 hours, then quench with saturated sodium bicarbonate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this rhodium-catalyzed direct hydroxylation technology offers substantial strategic advantages for procurement managers and supply chain heads. The primary benefit lies in the drastic simplification of the manufacturing process. By eliminating the preliminary halogenation step and the subsequent harsh hydroxylation conditions, the overall production timeline is significantly compressed. This reduction in unit operations translates directly into lower operational expenditures and reduced energy consumption. Moreover, the avoidance of strong bases and extreme temperatures enhances process safety, lowering the risk profile associated with large-scale manufacturing. The ability to recycle the trifluoroacetic anhydride solvent via distillation after the reaction further contributes to cost efficiency and waste minimization, making the process economically sustainable.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps removes the need for expensive halogenating agents and the disposal of halogenated waste byproducts. Additionally, the high yields reported, often exceeding 80% and reaching up to 95% for specific substrates like butyl phenyl ether derivatives, ensure maximum material throughput. This efficiency reduces the cost of goods sold (COGS) by minimizing raw material waste and maximizing the output per batch. The use of a robust catalytic system that maintains activity upon scale-up ensures that these cost benefits are retained when moving from gram-scale development to metric-ton commercial production.
- Enhanced Supply Chain Reliability: The broad substrate scope of this method means that a single versatile platform can be used to produce a wide array of 4-alkoxy phenol intermediates. This flexibility reduces dependency on multiple specialized synthetic routes and allows for more agile response to market demands. The mild reaction conditions reduce the wear and tear on reactor equipment and minimize the risk of batch failures due to thermal runaway or sensitivity issues. Consequently, lead times for high-purity pharmaceutical intermediates can be reduced, ensuring a more reliable and continuous supply of critical materials for downstream API synthesis.
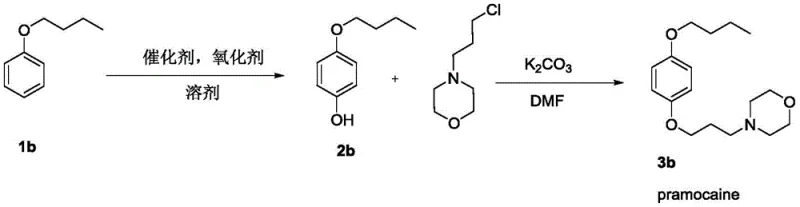
- Scalability and Environmental Compliance: The patent explicitly notes that the reaction activity does not significantly weaken upon amplification, indicating excellent scalability potential. The process generates less hazardous waste compared to traditional heavy-metal catalyzed couplings, facilitating easier compliance with increasingly stringent environmental regulations. The ability to produce key drugs like Monobenzone and Pramocaine with higher practicality and industrial application value positions this technology as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this process for integration into their supply chains.
Q: What are the key advantages of this Rhodium-catalyzed method over traditional halogenation routes?
A: This method eliminates the need for pre-functionalization (halogenation) of the substrate, avoiding harsh conditions like strong bases and high temperatures. It offers superior regioselectivity for the para-position and improves atom economy by directly activating the C-H bond.
Q: Is this process scalable for industrial production of intermediates like Monobenzone?
A: Yes, the patent data indicates that the reaction maintains high activity and yield even upon amplification to gram-level scales. The use of recoverable solvents like TFAA further supports its viability for commercial scale-up.
Q: What is the substrate scope regarding functional group tolerance?
A: The method demonstrates broad substrate applicability, tolerating various substituents including halogens (Cl, Br, I), esters, ketones, and alkyl chains, making it suitable for synthesizing diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Alkoxy Phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the rhodium-mediated direct hydroxylation described in CN113443970A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that the consistency of intermediates like 4-alkoxy phenols is critical for the efficacy and safety of the final pharmaceutical products, and our infrastructure is designed to deliver exactly that level of reliability.
We invite global partners to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Whether you require a Customized Cost-Saving Analysis for switching from traditional halogenation routes or need to evaluate the feasibility of this catalytic system for your specific molecule, our technical team is ready to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with high-performance, cost-effective chemical solutions.
