Advanced Aqueous Synthesis of Dimethyloxamonoamide for Nuclear Applications
The landscape of nuclear fuel reprocessing and high-level waste liquid treatment demands chemical intermediates of exceptional purity and reliability, particularly for the TRPO (Trialkyl Phosphine Oxide) extraction process. Patent CN100404499C introduces a groundbreaking aqueous phase synthesis method for water-soluble dimethyloxamonoamide, a critical stripping agent in nuclide decontamination. Historically, the synthesis of short-chain oxamonoamides was hindered by the stringent requirement for anhydrous conditions, as acid anhydrides typically undergo rapid hydrolysis in the presence of water. This patent overturns conventional wisdom by demonstrating that diglycolic anhydride can react directly with aqueous dimethylamine solution in the presence of pyridine, achieving high yields without the need for hazardous gas handling. For R&D directors and procurement specialists in the nuclear sector, this represents a paradigm shift towards safer, more scalable, and economically viable manufacturing protocols for specialized amide compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of methyl monoamides has been plagued by significant operational hazards and inefficiencies that pose severe challenges for industrial scale-up. The primary obstacle lies in the physical state of dimethylamine, which exists as a toxic, flammable gas at normal temperature and pressure, making its storage and transportation inherently dangerous and costly. Previous methodologies necessitated the generation of dimethylamine gas in situ by dripping aqueous solutions into hot sodium hydroxide, followed by rigorous two-stage drying processes to remove moisture before the reaction could proceed. This archaic approach not only required a massive excess of dimethylamine, often 5 to 10 times the theoretical amount, but also introduced complex engineering controls to manage gas leaks and ensure anhydrous environments. Consequently, the conventional route resulted in prohibitively high production costs, difficult synthetic operations, and substantial barriers to expanding capacity for meeting the growing demands of the nuclear waste treatment industry.
The Novel Approach
In stark contrast to the cumbersome traditional routes, the novel aqueous phase synthesis method described in the patent leverages the stability of commercial dimethylamine water solutions to streamline the entire production workflow. By utilizing diglycolic anhydride as the acylating agent and introducing pyridine as a specific promoter, the reaction successfully proceeds in a mixed solvent system containing water, which was previously thought impossible due to competitive hydrolysis. The process operates under mild low-temperature conditions, specifically maintaining the reaction mixture between 3-7°C, which effectively suppresses side reactions while facilitating the desired amidation. This innovation eliminates the need for gas generation and drying units, allowing manufacturers to utilize standard stainless steel reactors and significantly reducing the capital expenditure associated with safety infrastructure. The result is a robust, high-yield pathway that transforms a hazardous laboratory procedure into a practical, industrially feasible manufacturing process.
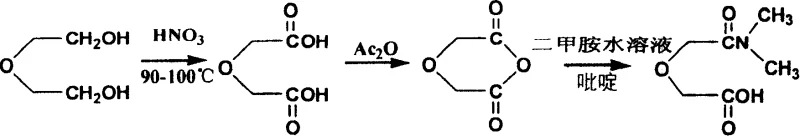
Mechanistic Insights into Aqueous Phase Amidation
The core chemical breakthrough of this technology lies in the unique interaction between the acid anhydride, the aqueous amine, and the pyridine catalyst, which alters the kinetic competition between amidation and hydrolysis. In a typical aqueous environment, the carbonyl carbon of an acid anhydride is highly susceptible to nucleophilic attack by water molecules, leading to the formation of carboxylic acids rather than the desired amide. However, the presence of pyridine likely forms a reactive acylpyridinium intermediate or activates the anhydride ring, lowering the activation energy for the nucleophilic attack by dimethylamine. This catalytic effect ensures that the amine, even when diluted in water, reacts faster with the activated anhydride than water molecules do, thereby preserving the integrity of the acyl group. Furthermore, the exothermic nature of the ring-opening reaction is carefully managed by the ice-salt bath, preventing thermal runaway that could accelerate hydrolysis or degrade the sensitive oxamonoamide structure.
Prior to the amidation step, the precursor diglycolic anhydride is synthesized through a controlled oxidation and dehydration sequence that ensures high purity of the starting material. The initial oxidation of diethylene glycol using concentrated nitric acid at 90-100°C generates diglycolic acid, which is then purified through recrystallization to remove nitrate impurities that could interfere with downstream reactions. Subsequent dehydration using acetic anhydride and a phosphoric acid initiator at 130-150°C drives the cyclization to form the anhydride ring with high efficiency. This rigorous purification protocol at the intermediate stage is critical for the final product's performance in nuclear applications, where trace metal ions or organic impurities could compromise the extraction efficiency or cause precipitation in the TRPO process. The meticulous control over each synthetic step guarantees a final product with a sharp melting point and consistent spectral characteristics, validating the structural integrity required for high-performance ligands.
How to Synthesize Dimethyloxamonoamide Efficiently
The synthesis of this high-value nuclear intermediate follows a logical three-stage progression that balances reaction kinetics with safety parameters to maximize output. The process begins with the preparation of the acid backbone, moves through the formation of the reactive anhydride, and concludes with the delicate aqueous coupling step. Each stage requires precise temperature control and stoichiometric balance to prevent the formation of byproducts such as diglycolic acid or bis-amides. For detailed operational parameters, including specific stirring rates, addition times, and workup procedures, operators should refer to the standardized technical guidelines derived from the patent data.
- Oxidize diethylene glycol using 10mol/L nitric acid at 90-100°C to produce diglycolic acid crystals.
- Dehydrate diglycolic acid using acetic anhydride and phosphoric acid catalyst at 130-150°C to form diglycolic anhydride.
- React diglycolic anhydride with 33% aqueous dimethylamine and pyridine in 1,4-dioxane at 3-7°C to obtain the final amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous synthesis method offers profound strategic benefits that extend far beyond simple yield improvements. The elimination of gaseous dimethylamine handling removes a major bottleneck in logistics and storage, as aqueous solutions are significantly easier to transport and store in bulk quantities compared to pressurized gas cylinders. This shift drastically simplifies the regulatory compliance landscape, reducing the need for specialized hazardous gas detection systems and emergency scrubbing towers, which translates directly into lower overhead costs and reduced insurance premiums for manufacturing facilities. Furthermore, the reliance on commodity chemicals like nitric acid, acetic anhydride, and pyridine ensures a stable supply chain that is less vulnerable to the fluctuations often seen with specialized reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the reaction setup and the reduction in reagent waste. By avoiding the need for extensive drying agents and the massive excess of dimethylamine required in older methods, the overall material cost per kilogram of product is significantly lowered. Additionally, the ability to run the reaction at near-ambient pressures and low temperatures reduces energy consumption associated with heating and pressurization, contributing to a leaner cost structure. The streamlined workup procedure, which involves simple distillation and crystallization, minimizes solvent usage and labor hours, further enhancing the cost-effectiveness of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply continuity is greatly improved because the key raw material, dimethylamine, is sourced as a stable aqueous solution rather than a volatile gas. This reduces the risk of supply interruptions caused by transportation restrictions on hazardous gases or failures in gas generation equipment. The robustness of the aqueous reaction conditions also means that production can be maintained consistently without the frequent downtime associated with maintaining strict anhydrous environments. Manufacturers can therefore offer more reliable delivery schedules to their clients in the nuclear sector, who depend on uninterrupted supplies of stripping agents for their continuous waste treatment operations.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns well with modern green chemistry principles by reducing the generation of hazardous waste streams. The absence of strong bases like sodium hydroxide in the reaction zone minimizes the production of saline wastewater, simplifying effluent treatment requirements. The process is inherently scalable because it does not rely on complex gas-liquid mass transfer limitations that often plague scale-up efforts in traditional amidation. This allows for seamless expansion from pilot plant batches to multi-ton commercial production runs, ensuring that the supply can grow in tandem with the increasing global demand for advanced nuclear fuel cycle technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dimethyloxamonoamide. These answers are derived directly from the mechanistic data and experimental results presented in the patent literature to provide clarity on process feasibility. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their existing supply chains or research pipelines.
Q: Why is the aqueous phase synthesis method superior for dimethyloxamonoamide?
A: Traditional methods require hazardous anhydrous dimethylamine gas and complex drying steps. This patented aqueous method allows the use of stable, commercially available dimethylamine water solution, significantly improving operational safety and simplifying the workflow.
Q: What is the role of pyridine in this reaction mechanism?
A: Pyridine acts as a crucial promoter that facilitates the nucleophilic attack of dimethylamine on the anhydride even in the presence of water, preventing preferential hydrolysis of the anhydride and ensuring high conversion rates at low temperatures.
Q: Can this process be scaled for industrial nuclear waste treatment applications?
A: Yes, the process utilizes standard reactor equipment and avoids extreme pressure or cryogenic conditions. The use of aqueous reagents and common solvents like 1,4-dioxane makes it highly adaptable for large-scale commercial production required in the nuclear industry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimethyloxamonoamide Supplier
As the global demand for efficient nuclear waste treatment solutions continues to rise, the need for high-purity stripping agents like dimethyloxamonoamide has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this specialized sector, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet the rigorous standards of the nuclear industry. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of dimethyloxamonoamide meets the exacting requirements for TRPO processes, guaranteeing optimal performance in nuclide decontamination applications.
We invite potential partners to engage with our technical team to explore how this advanced aqueous synthesis technology can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient production route. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your project's unique volume and purity needs.
