Optimizing Rosuvastatin Intermediate Production: A Technical Breakdown of Novel Synthetic Routes
The pharmaceutical industry continuously seeks robust synthetic pathways for critical statin intermediates, particularly for the production of Rosuvastatin Calcium, a potent HMG-CoA reductase inhibitor. Patent CN113185428B introduces a transformative methodology for synthesizing 4-methyl-3-oxo-valeronitrile, a pivotal building block in this therapeutic class. This innovation addresses longstanding inefficiencies in prior art by replacing hazardous cryogenic conditions with a streamlined reflux protocol. The strategic importance of this intermediate cannot be overstated, as it serves as the foundational scaffold for the final active pharmaceutical ingredient. By leveraging acetonitrile not merely as a solvent but as an active reactant, the process achieves a dual optimization of material efficiency and operational safety. This technical advancement represents a significant leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediates supplier capable of meeting stringent global quality standards.
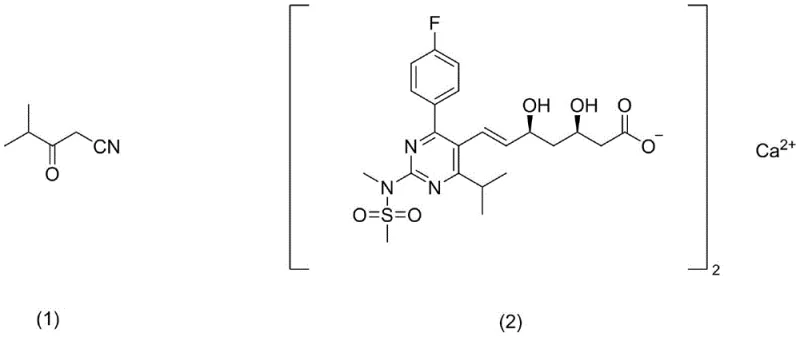
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-methyl-3-oxo-valeronitrile has been plagued by severe operational constraints that hinder industrial scalability. Traditional routes often rely on strong, hazardous bases such as lithium bis(trimethylsilyl)amide or n-butyllithium, which necessitate ultra-low temperature environments around -78°C to control reactivity. These cryogenic requirements impose a massive energy burden on manufacturing facilities and introduce significant safety risks associated with handling pyrophoric reagents on a large scale. Furthermore, alternative methods utilizing sodium hydride in tetrahydrofuran often suffer from difficult post-treatment procedures, requiring high-temperature distillation that can degrade product quality and increase processing time. The reliance on expensive solvents and the generation of complex waste streams further exacerbate the economic and environmental drawbacks of these legacy processes, making them unsuitable for modern green chemistry mandates.
The Novel Approach
In stark contrast, the disclosed invention utilizes potassium methoxide as a cost-effective and safer base, operating under mild reflux conditions between 70-90°C. This thermal shift eliminates the need for energy-intensive cooling systems, drastically simplifying the reactor setup and reducing utility costs. The reaction proceeds with high selectivity, minimizing the formation of byproducts that typically complicate purification. By employing acetonitrile as the reaction medium, the process reduces the overall solvent load, aligning with principles of atom economy. The workup involves a straightforward pH adjustment and liquid-liquid extraction, bypassing the need for complex distillation columns. This approach not only enhances the safety profile for plant operators but also ensures a more consistent supply chain by reducing dependency on specialized cryogenic infrastructure.
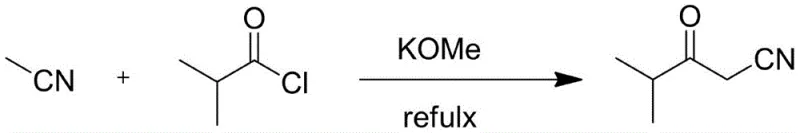
Mechanistic Insights into Potassium Methoxide Catalyzed Condensation
The core of this synthetic breakthrough lies in the efficient generation of the acetonitrile anion using potassium methoxide. Under reflux conditions, the base deprotonates the alpha-carbon of acetonitrile, creating a nucleophilic species capable of attacking the carbonyl carbon of isobutyryl chloride. This nucleophilic acyl substitution proceeds rapidly due to the enhanced leaving group ability of the chloride ion compared to ester counterparts used in older methods. The reaction kinetics are optimized by the molar ratio of reactants, typically maintained at 3:2:1 for acetonitrile, potassium methoxide, and isobutyryl chloride respectively. This stoichiometric balance ensures complete consumption of the acid chloride while preventing excessive polymerization or side reactions. The stability of the intermediate enolate is maintained throughout the 3-4 hour reaction window, allowing for high conversion rates without the degradation observed in harsher alkaline environments.
Impurity control is inherently managed through the precise regulation of the quenching phase. Upon completion, the reaction mixture is cooled to 0-5°C, and the pH is carefully adjusted to 5-6 using a 2 mol/L hydrochloric acid solution. This specific pH range is critical; it neutralizes excess base and decomposes any remaining reactive intermediates without hydrolyzing the sensitive nitrile group or the beta-keto functionality. The subsequent phase separation allows the organic product to partition efficiently into the oil phase or ethyl acetate extract. Washing steps further remove inorganic salts and polar impurities, resulting in a crude product with purity exceeding 99%. This high level of intrinsic purity reduces the burden on downstream purification units, ensuring that the final high-purity pharmaceutical intermediates meet rigorous regulatory specifications for heavy metals and residual solvents.
How to Synthesize 4-Methyl-3-Oxo-Valeronitrile Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent embodiments. The process begins with the preparation of the base mixture, followed by the controlled addition of the acylating agent to manage exothermicity. Operators must monitor the reaction progress via gas chromatography to confirm the disappearance of starting materials before initiating the quench. The extraction protocol is equally vital, requiring multiple washes to ensure the removal of potassium salts. While the general workflow is robust, specific attention to temperature gradients during the reflux and cooling phases is essential for maximizing yield. The detailed standardized synthesis steps see the guide below for exact operational parameters.
- Prepare the reaction vessel by adding acetonitrile and potassium methoxide, then heat the mixture to a controlled temperature range of 70-90°C.
- Dropwise add isobutyryl chloride under reflux conditions and maintain the reaction for 3-4 hours to ensure complete conversion.
- Cool the system to 0-5°C, adjust pH to 5-6 with hydrochloric acid, separate the oil phase, and extract the aqueous layer with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers substantial strategic benefits beyond mere technical feasibility. The elimination of cryogenic reagents and the use of commodity chemicals like potassium methoxide significantly lower the raw material cost baseline. This shift mitigates the risk of supply disruptions associated with specialized, hazardous reagents that often face strict transportation regulations. Furthermore, the simplified workup procedure reduces the cycle time per batch, enhancing overall equipment effectiveness and throughput capacity. These factors combine to create a more resilient supply chain capable of responding to fluctuating market demands for statin derivatives without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The replacement of expensive organolithium reagents with potassium methoxide results in a drastic reduction in direct material costs. Additionally, the ability to use acetonitrile as both solvent and reactant minimizes solvent purchase and recovery expenses. The avoidance of high-vacuum distillation further lowers energy consumption and maintenance costs for processing equipment. These cumulative efficiencies translate into significant cost savings that can be passed down the supply chain, offering a competitive pricing advantage for the final API.
- Enhanced Supply Chain Reliability: By removing dependencies on ultra-low temperature infrastructure and hazardous pyrophoric materials, the manufacturing process becomes less susceptible to operational delays and safety shutdowns. The use of stable, commercially available reagents ensures consistent availability even during global supply tightness. This reliability is crucial for maintaining continuous production schedules and meeting just-in-time delivery commitments to downstream pharmaceutical partners, thereby strengthening the overall partnership ecosystem.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup facilitate easy scale-up from pilot plants to multi-ton commercial reactors without significant engineering redesigns. The reduction in hazardous waste generation and the use of less toxic solvents align with increasingly stringent environmental regulations. This compliance reduces the administrative burden of waste disposal and permits, ensuring long-term operational sustainability and minimizing the risk of regulatory fines or production halts due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific beneficial effects and experimental data presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer. The answers provide clarity on safety, purity, and scalability concerns that typically arise during the vendor qualification process.
Q: How does this new synthesis method improve safety compared to traditional routes?
A: Unlike conventional methods requiring pyrophoric reagents like n-butyllithium or sodium hydride at cryogenic temperatures (-78°C), this patent utilizes potassium methoxide under reflux conditions, significantly reducing operational hazards and energy consumption.
Q: What purity levels can be achieved without high-vacuum distillation?
A: The optimized workup procedure involving pH adjustment and ethyl acetate extraction allows for the direct isolation of the product with purity exceeding 99%, eliminating the need for energy-intensive high-vacuum distillation steps.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of acetonitrile as both solvent and reactant, combined with mild thermal conditions and standard extraction techniques, makes this route highly scalable for industrial batch production with consistent yield above 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methyl-3-Oxo-Valeronitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of cardiovascular therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-methyl-3-oxo-valeronitrile meets the exacting standards required for GMP manufacturing. Our commitment to process excellence means we can deliver the consistency and reliability necessary for your long-term drug development pipelines.
We invite you to collaborate with us to optimize your supply chain for statin intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By leveraging our advanced synthesis capabilities, you can secure a stable source of high-purity pharmaceutical intermediates that drive efficiency and reduce total cost of ownership for your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
