Advanced Manufacturing of 1-[3-Chloro-5-(Trifluoromethyl) Phenyl]-2,2,2-Trifluoroacetone via Diazotization
Introduction to Next-Generation Veterinary Drug Intermediates
The pharmaceutical landscape for veterinary parasiticides is undergoing a significant transformation, driven by the demand for more efficient and cost-effective synthetic routes for key active ingredients. Patent CN112778109B, published in September 2022, introduces a groundbreaking preparation method for 1-[3-chloro-5-(trifluoromethyl) phenyl]-2,2,2-trifluoroacetone and its derivatives, which serve as critical precursors for the next-generation oral anthelmintic Afoxolaner. This technology addresses long-standing inefficiencies in traditional manufacturing by replacing expensive starting materials and harsh reaction conditions with a streamlined, mild, and highly selective process. For R&D directors and procurement specialists, this innovation represents a pivotal opportunity to optimize the supply chain for high-value fluorochemical intermediates. By leveraging p-trifluoromethylaniline as a primary feedstock and utilizing a novel diazotization deamination strategy, the method not only enhances economic viability but also ensures robust scalability from laboratory bench to multi-ton commercial production.
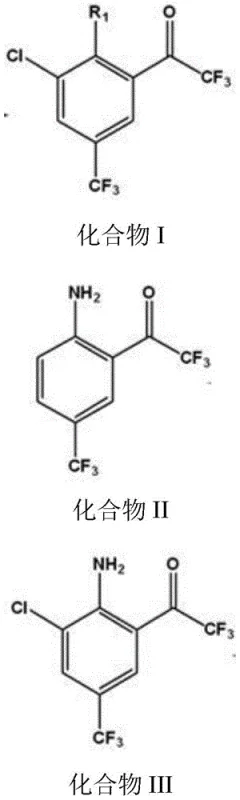
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-[3-chloro-5-(trifluoromethyl) phenyl]-2,2,2-trifluoroacetone has been dominated by the methodology disclosed in Bayer Corporation's patent US2011105532. This legacy approach relies heavily on 1-bromo-3-chloro-5-trifluorotoluene as the foundational starting material, a compound that is notoriously expensive and difficult to source in bulk quantities due to complex upstream halogenation processes. Furthermore, the core transformation involves a Grignard reagent reaction that must be conducted at cryogenic temperatures of approximately -80°C. Such extreme conditions necessitate specialized refrigeration equipment and substantial energy inputs, creating a significant bottleneck for cost reduction in API manufacturing. The operational complexity associated with maintaining such low temperatures also introduces safety risks and limits the feasibility of large-scale continuous processing, making the conventional route less attractive for modern, high-volume supply chains seeking agility and efficiency.
The Novel Approach
In stark contrast, the methodology outlined in CN112778109B revolutionizes the synthetic pathway by utilizing p-trifluoromethylaniline, pivaloyl chloride, and ethyl trifluoroacetate as readily available and economically favorable raw materials. This strategic shift eliminates the dependency on costly brominated aromatics and avoids the need for cryogenic Grignard chemistry entirely. The new process operates under mild conditions, typically ranging from ambient temperature to moderate heating (50-80°C), which drastically simplifies reactor requirements and reduces utility costs. By employing a protective group strategy followed by directed ortho-chlorination and a final diazotization deamination step, the novel approach achieves superior regioselectivity and minimizes the formation of difficult-to-remove impurities. This results in a cleaner reaction profile that facilitates easier downstream purification, ultimately delivering a high-purity pharmaceutical intermediate suitable for stringent regulatory standards without the burden of excessive processing steps.
Mechanistic Insights into Diazotization Deamination and Ortho-Chlorination
The core chemical innovation of this patent lies in the precise manipulation of electronic effects on the aromatic ring to achieve specific substitution patterns. The synthesis begins with the formation of Compound II, where the amino group of p-trifluoromethylaniline is temporarily protected and the trifluoroacetyl group is introduced. 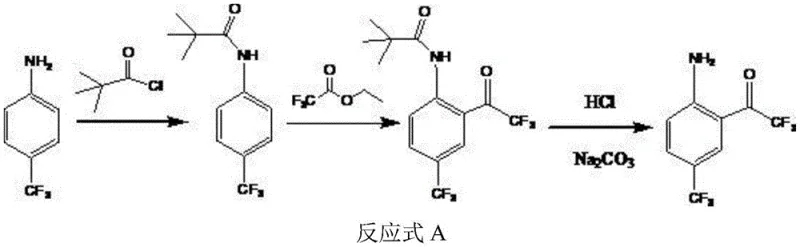 Subsequently, the amino group acts as a powerful electron-donating director, facilitating the electrophilic substitution of a chlorine atom at the ortho position during the reaction with sulfonyl chloride. This directing effect is crucial because it ensures that chlorination occurs exclusively at the desired position adjacent to the amino group, preventing the formation of meta- or para-substituted by-products that would complicate purification. The use of sulfonyl chloride as the chlorinating agent is particularly advantageous; it reacts efficiently under heated conditions in organic solvents immiscible with water, allowing for a straightforward workup where water-soluble impurities are easily separated from the organic phase containing the desired Compound III.
Subsequently, the amino group acts as a powerful electron-donating director, facilitating the electrophilic substitution of a chlorine atom at the ortho position during the reaction with sulfonyl chloride. This directing effect is crucial because it ensures that chlorination occurs exclusively at the desired position adjacent to the amino group, preventing the formation of meta- or para-substituted by-products that would complicate purification. The use of sulfonyl chloride as the chlorinating agent is particularly advantageous; it reacts efficiently under heated conditions in organic solvents immiscible with water, allowing for a straightforward workup where water-soluble impurities are easily separated from the organic phase containing the desired Compound III.
The final transformation involves a sophisticated diazotization deamination sequence that offers remarkable versatility in generating diverse derivatives. By treating Compound III with a nitrous acid reagent (such as sodium nitrite) under acidic conditions at low temperatures (below 0°C), a diazonium salt intermediate is formed. This highly reactive species can then be subjected to various substitution conditions to introduce different functional groups (R1 = H, Cl, Br, F, OH) at the position previously occupied by the amino group. For instance, using hypophosphorous acid and cuprous chloride allows for the removal of the amino group to yield hydrogen (R1=H), while cuprous bromide or hydrogen fluoride-pyridine solutions enable the introduction of bromine or fluorine atoms, respectively. This modularity is a significant asset for medicinal chemists exploring structure-activity relationships, as it allows for the rapid generation of a library of analogues from a common intermediate without redesigning the entire synthetic route, thereby accelerating drug discovery timelines.
How to Synthesize 1-[3-Chloro-5-(Trifluoromethyl) Phenyl]-2,2,2-Trifluoroacetone Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and robustness, making it ideal for transfer from pilot plants to full-scale manufacturing facilities. The process is divided into three distinct stages: the initial construction of the protected amino-ketone scaffold (Compound II), the regioselective chlorination to form Compound III, and the final diazotization step to yield the target molecule. Each stage has been optimized for solvent compatibility and temperature control to maximize yield and minimize waste. Detailed standard operating procedures regarding specific molar ratios, addition rates, and quenching protocols are essential for maintaining the high purity profiles reported in the patent examples. Operators should pay close attention to the temperature gradients during the diazotization phase, as maintaining the system below 0°C is critical for stabilizing the diazonium intermediate and preventing premature decomposition or side reactions.
- Synthesize Compound II (2-amino-5-trifluoromethyl phenyl trifluoromethyl ketone) using p-trifluoromethylaniline, pivaloyl chloride, and ethyl trifluoroacetate under mild conditions.
- Perform amino ortho-chlorination on Compound II using a chlorination reagent like sulfonyl chloride to obtain Compound III with high selectivity.
- Execute diazotization deamination on Compound III using nitrous acid reagents and specific substitution agents (e.g., hypophosphorous acid) to yield the final trifluoroacetone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond simple chemical yield improvements. The transition from cryogenic Grignard chemistry to mild thermal processes fundamentally alters the cost structure of production by eliminating the need for expensive low-temperature infrastructure and reducing energy consumption significantly. Moreover, the reliance on commodity chemicals like p-trifluoromethylaniline and sodium nitrite mitigates supply risk, as these materials are produced by multiple global vendors, ensuring continuity of supply even during market fluctuations. This diversification of the raw material base is a critical factor in building a resilient supply chain capable of withstanding geopolitical or logistical disruptions.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions (-80°C) removes a major capital expenditure barrier, allowing production in standard glass-lined or stainless steel reactors without specialized cooling jackets. Additionally, the use of sulfonyl chloride instead of chlorine gas or N-chlorosuccinimide improves atom economy and reduces the cost of goods sold by minimizing reagent waste. The simplified workup procedures, which often involve simple phase separations rather than complex chromatographic purifications, further drive down operational expenses by reducing solvent usage and labor hours per batch.
- Enhanced Supply Chain Reliability: By shifting away from proprietary or scarce brominated starting materials to widely available aniline derivatives, manufacturers can secure long-term contracts with multiple suppliers, reducing dependency on single sources. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, leading to more consistent batch-to-batch performance and fewer production delays caused by out-of-specification inputs. This reliability is paramount for meeting the strict delivery schedules required by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The patent data demonstrates successful scale-up from gram to multi-kilogram scales (Examples 31-35) with maintained yields, proving the technology's readiness for commercial deployment. The aqueous workup steps effectively sequester acidic by-products and metal salts into the water phase, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations. The overall reduction in hazardous reagent usage and energy intensity aligns with green chemistry principles, enhancing the sustainability profile of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this new synthesis method superior to the conventional Bayer process?
A: The conventional method (US2011105532) relies on expensive 1-bromo-3-chloro-5-trifluorotoluene and requires harsh Grignard reactions at -80°C. This new patent (CN112778109B) utilizes cheaper p-trifluoromethylaniline and operates under mild conditions, significantly lowering energy consumption and raw material costs while avoiding cryogenic equipment requirements.
Q: What represents the key advantage in terms of product purity and yield?
A: The process achieves exceptional purity levels, often exceeding 99%, with yields reaching up to 96.4% in the chlorination step and over 83% in the final deamination step. The use of specific solvents like toluene and precise temperature controls during diazotization minimizes side reactions and simplifies post-reaction separation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates successful scale-up experiments (Examples 31-35) where the reaction was amplified tenfold without loss of yield or purity. The use of readily available reagents like sodium nitrite and sulfonyl chloride ensures supply chain stability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-[3-Chloro-5-(Trifluoromethyl) Phenyl]-2,2,2-Trifluoroacetone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation veterinary therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify identity and assay every batch. Our facility is equipped to handle the specific thermal and safety requirements of fluorinated chemistry, providing a secure and compliant environment for the manufacture of complex pharmaceutical intermediates.
We invite global partners to collaborate with us to leverage this innovative synthesis route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development programs. Let us be your trusted partner in bringing high-performance anthelmintics to the market faster and more cost-effectively.
