Advanced Regioselective Synthesis of High-Purity Glycosyl Phosphate Intermediates for Commercial Scale-Up
Advanced Regioselective Synthesis of High-Purity Glycosyl Phosphate Intermediates for Commercial Scale-Up
The synthesis of phosphorylated aminosugars represents a critical frontier in the development of next-generation therapeutics, particularly for glycogen metabolism modifiers and novel antiviral agents. However, the chemical complexity of these molecules, characterized by multiple hydroxyl groups with similar reactivities, has historically posed severe challenges for industrial manufacturing. Patent CN112500441A introduces a groundbreaking preparation process for high-purity glycosyl phosphate that effectively resolves these long-standing bottlenecks. By employing a sophisticated strategy of sequential protection and deprotection, specifically utilizing trityl and benzyl groups, this technology enables the precise installation of phosphate moieties at the C6 position. For global pharmaceutical developers, this innovation translates into a reliable glycosyl phosphate supplier capability that guarantees exceptional chemical integrity and minimizes the presence of difficult-to-remove impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-6-phosphoaminosugars has been plagued by poor regioselectivity and difficult purification profiles. Traditional chemical routes often involve the direct phosphorylation of unprotected or partially protected sugar alcohols, which inevitably leads to a mixture of positional isomers. As illustrated in prior art, one common approach involves the use of phenyl phosphate esters. 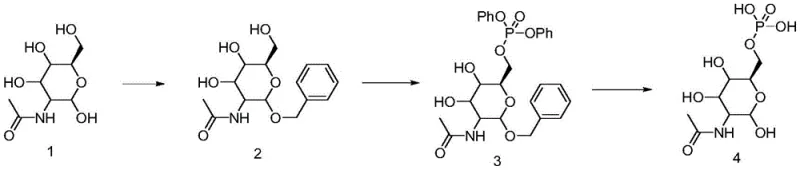 While conceptually straightforward, the phenyl phosphate intermediate presents a formidable barrier to efficient production. The catalytic hydrogenolysis required to remove the phenyl group exhibits low activity, necessitating the use of large excesses of noble metal catalysts and prolonged reaction times under elevated heat and pressure. These harsh conditions frequently compromise the stability of the sensitive sugar substrate, leading to the generation of degradation impurities that are structurally similar to the target molecule and extremely difficult to separate. Furthermore, alternative enzymatic approaches using hexokinase and pyruvate kinase complexes, while specific, introduce their own set of logistical nightmares, including complex reaction systems, safety hazards associated with sodium azide reagents, and the arduous task of separating water-soluble products from enzyme mixtures.
While conceptually straightforward, the phenyl phosphate intermediate presents a formidable barrier to efficient production. The catalytic hydrogenolysis required to remove the phenyl group exhibits low activity, necessitating the use of large excesses of noble metal catalysts and prolonged reaction times under elevated heat and pressure. These harsh conditions frequently compromise the stability of the sensitive sugar substrate, leading to the generation of degradation impurities that are structurally similar to the target molecule and extremely difficult to separate. Furthermore, alternative enzymatic approaches using hexokinase and pyruvate kinase complexes, while specific, introduce their own set of logistical nightmares, including complex reaction systems, safety hazards associated with sodium azide reagents, and the arduous task of separating water-soluble products from enzyme mixtures.
The Novel Approach
In stark contrast to these legacy methods, the novel process disclosed in the patent utilizes a meticulously designed protective group strategy to ensure absolute control over the reaction trajectory. 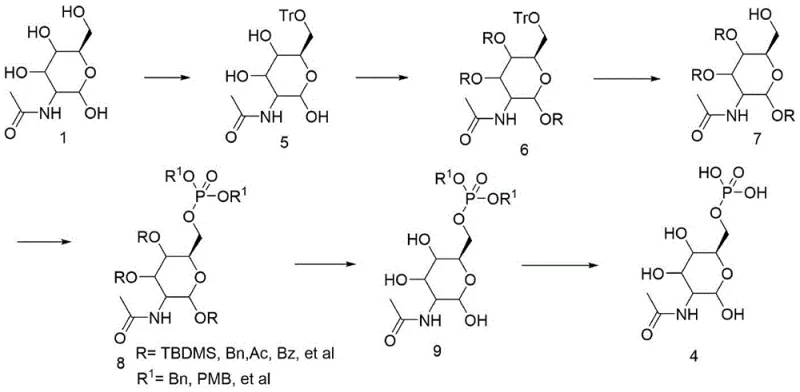 The core of this innovation lies in the initial selective protection of the C6 hydroxyl group using a trityl (Tr) moiety, followed by the protection of remaining hydroxyls with acetyl groups. This creates a robust intermediate where the C6 position can be selectively unveiled by removing the trityl group without disturbing the rest of the molecule. Crucially, the subsequent phosphorylation step employs dibenzyl phosphate rather than phenyl phosphate. This strategic substitution is a game-changer for manufacturing efficiency. The benzyl phosphate ester is inherently more labile under hydrogenolysis conditions, allowing for complete deprotection at room temperature and normal pressure. This shift not only eliminates the risk of thermal degradation but also drastically simplifies the workup procedure, ensuring that the final product emerges with high purity and minimal related impurities, thereby establishing a new standard for cost reduction in pharmaceutical intermediate manufacturing.
The core of this innovation lies in the initial selective protection of the C6 hydroxyl group using a trityl (Tr) moiety, followed by the protection of remaining hydroxyls with acetyl groups. This creates a robust intermediate where the C6 position can be selectively unveiled by removing the trityl group without disturbing the rest of the molecule. Crucially, the subsequent phosphorylation step employs dibenzyl phosphate rather than phenyl phosphate. This strategic substitution is a game-changer for manufacturing efficiency. The benzyl phosphate ester is inherently more labile under hydrogenolysis conditions, allowing for complete deprotection at room temperature and normal pressure. This shift not only eliminates the risk of thermal degradation but also drastically simplifies the workup procedure, ensuring that the final product emerges with high purity and minimal related impurities, thereby establishing a new standard for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Regioselective Protection and Phosphorylation
The success of this synthesis hinges on the precise exploitation of steric hindrance and electronic differences between the various hydroxyl groups on the aminosugar scaffold. In the first stage, the reaction of the starting amino sugar with triphenylchloromethane (TrCl) in the presence of a base like pyridine or triethylamine is highly selective for the primary hydroxyl at the C6 position. The bulky trityl group physically cannot access the secondary hydroxyls on the ring as easily as the exposed primary alcohol, driving the formation of intermediate 5 with high fidelity. Following this, the remaining secondary hydroxyls are capped with acetyl groups using acetic anhydride, locking the molecule into a fully protected state (Intermediate 6). The subsequent removal of the trityl group is achieved under mild acidic conditions (acetic acid/water), which cleaves the trityl ether without affecting the acid-stable acetyl esters or the glycosidic bond. This sequence ensures that when the phosphorylation reagent is introduced, the C6 hydroxyl is the only available nucleophile, guaranteeing regioselectivity.
Furthermore, the choice of phosphorylating agent and the final deprotection mechanism are critical for impurity control. 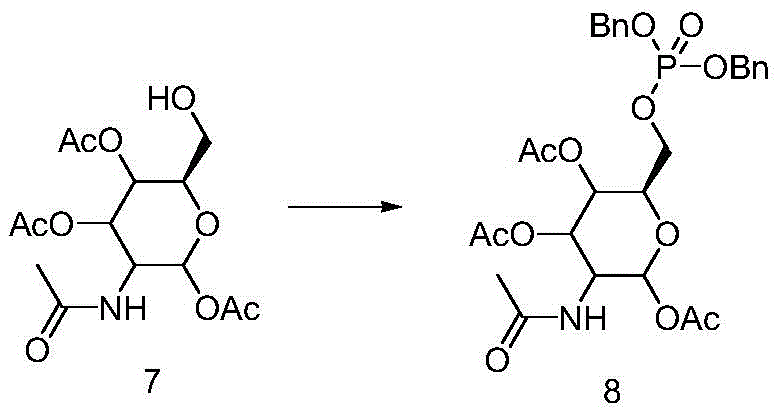 The use of dibenzyl phosphoryl chloride or tetrabenzyl pyrophosphate allows for the formation of the dibenzyl phosphate intermediate (Intermediate 8) under controlled anhydrous conditions. Unlike phenyl esters, which form strong bonds with the phosphorus atom requiring aggressive conditions to break, the benzyl-oxygen bond is susceptible to catalytic hydrogenolysis using palladium on carbon. This mechanistic advantage means that the final deprotection can proceed smoothly at ambient temperatures, preserving the stereochemical integrity of the sugar ring and preventing the formation of elimination by-products or degradation fragments. The result is a clean reaction profile where the primary impurities are easily removed via crystallization or simple extraction, leading to a final product with purity exceeding 99%.
The use of dibenzyl phosphoryl chloride or tetrabenzyl pyrophosphate allows for the formation of the dibenzyl phosphate intermediate (Intermediate 8) under controlled anhydrous conditions. Unlike phenyl esters, which form strong bonds with the phosphorus atom requiring aggressive conditions to break, the benzyl-oxygen bond is susceptible to catalytic hydrogenolysis using palladium on carbon. This mechanistic advantage means that the final deprotection can proceed smoothly at ambient temperatures, preserving the stereochemical integrity of the sugar ring and preventing the formation of elimination by-products or degradation fragments. The result is a clean reaction profile where the primary impurities are easily removed via crystallization or simple extraction, leading to a final product with purity exceeding 99%.
How to Synthesize High-Purity Glycosyl Phosphate Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and reaction conditions to maximize yield and purity. The process begins with the suspension of the aminosugar in pyridine, followed by the addition of trityl chloride at elevated temperatures to ensure complete conversion to the mono-trityl intermediate. Subsequent acetylation is performed at lower temperatures to prevent side reactions. The selective de-tritylation step utilizes a specific ratio of acetic acid to water to tune the acidity, ensuring only the trityl group is removed. Phosphorylation is then conducted under inert atmosphere to prevent moisture interference, using either dibenzyl phosphoryl chloride with an organic base or a lithium hexamethyldisilazide (LHMDS) mediated coupling with tetrabenzyl pyrophosphate. Finally, the sequential removal of acetyl and benzyl groups yields the target phosphate. For detailed operational parameters and specific molar ratios, please refer to the standardized protocol below.
- Selectively protect the C6 hydroxyl group of the amino sugar using a trityl (Tr) group to form intermediate 5, followed by acetylation of remaining hydroxyls to yield intermediate 6.
- Selectively remove the trityl protecting group using an acetic acid/water solution to regenerate the C6 hydroxyl, resulting in intermediate 7.
- Phosphorylate the C6 hydroxyl using dibenzyl phosphoryl chloride or tetrabenzyl pyrophosphate to install the benzyl phosphate group, forming intermediate 8.
- Remove acetyl protecting groups using alkaline hydrolysis to obtain intermediate 9, followed by catalytic hydrogenolysis to remove benzyl groups and yield the final high-purity product 4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers substantial strategic benefits beyond mere chemical elegance. The transition from phenyl-based to benzyl-based phosphate intermediates fundamentally alters the economic landscape of production by eliminating the need for expensive, high-pressure hydrogenation equipment and excessive catalyst loading. This process optimization directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering energy consumption and extending the lifespan of processing equipment. Moreover, the high regioselectivity inherent in the trityl protection strategy minimizes the formation of isomeric impurities, which are notoriously expensive and time-consuming to remove via preparative chromatography. By designing impurities out of the process rather than trying to filter them out at the end, manufacturers can achieve significant throughput improvements.
- Cost Reduction in Manufacturing: The replacement of phenyl phosphate with benzyl phosphate eliminates the need for harsh hydrogenolysis conditions. In traditional methods, incomplete hydrogenolysis often requires recycling batches or using massive excesses of palladium catalyst, driving up raw material costs. The new method allows for smooth deprotection at room temperature and normal pressure, significantly reducing utility costs and catalyst consumption. Additionally, the high selectivity of the initial trityl protection step reduces the burden on downstream purification, meaning less solvent and silica gel are required for column chromatography or crystallization, leading to substantial overall cost savings.
- Enhanced Supply Chain Reliability: This synthetic route relies on robust, commercially available reagents such as trityl chloride, acetic anhydride, and dibenzyl phosphoryl chloride, avoiding the supply chain volatility associated with specialized enzymes or unstable reagents like sodium azide used in older methods. The chemical stability of the intermediates, particularly the fully protected species, allows for potential storage or transport between stages if necessary, providing flexibility in production scheduling. This robustness ensures consistent batch-to-batch quality and reduces the risk of production delays caused by failed enzymatic reactions or difficult separations.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up of complex carbohydrate derivatives in mind. Steps such as the precipitation of intermediates upon adding water or non-solvents (like methyl tert-butyl ether) are highly scalable unit operations that do not require specialized equipment. Furthermore, by avoiding the use of heavy metal catalysts in excessive amounts and reducing the generation of complex waste streams associated with enzymatic buffers, the process aligns better with modern environmental, health, and safety (EHS) standards. The ability to achieve high purity (>99%) through crystallization and simple washes rather than extensive chromatography also reduces the volume of organic solvent waste generated per kilogram of product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity glycosyl phosphate synthesis. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the feasibility and advantages of this route for potential partners and licensees.
Q: Why is the benzyl phosphate group preferred over the phenyl phosphate group in this synthesis?
A: The benzyl phosphate group is significantly easier to remove via catalytic hydrogenolysis under mild conditions (room temperature and pressure) compared to the phenyl phosphate group. The phenyl ester requires harsh conditions that often lead to substrate degradation and incomplete reaction, whereas the benzyl ester allows for clean deprotection with high yields.
Q: How does this process achieve high regioselectivity at the C6 position?
A: The process utilizes the steric hindrance of the trityl chloride (TrCl) reagent. Due to the bulky nature of the trityl group and the difference in hydroxyl activity, it selectively reacts with the primary hydroxyl group at the C6 position first, allowing for precise differentiation from the secondary hydroxyls on the sugar ring.
Q: What purity levels can be achieved with this manufacturing method?
A: This optimized synthetic route is designed to minimize structural analogue impurities and degradation products. Experimental data indicates that the final glycosyl phosphate product can achieve an HPLC purity of greater than 99%, significantly reducing the burden on downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycosyl Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory bench to commercial production requires more than just a patent; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate protection and deprotection steps of this glycosyl phosphate synthesis are executed with precision and consistency. We understand that for critical pharmaceutical intermediates, stringency is non-negotiable. Therefore, our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of high-purity amino sugar phosphate meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced technology for your drug development pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can reduce lead time for high-purity glycosyl phosphates and accelerate your time to market.
