Scalable AMPPD Production via Novel Titanium Catalysis for Global Diagnostic Markets
Scalable AMPPD Production via Novel Titanium Catalysis for Global Diagnostic Markets
The landscape of chemiluminescence immunoassay diagnostics is constantly evolving, driven by the critical need for high-sensitivity substrates like AMPPD (4-methoxy-4-(3-phenylphosphonic)spiro[1,2-dioxetane-3,2'-adamantane]disodium salt). A pivotal advancement in this field is detailed in Chinese Patent CN102030779A, which discloses a robust preparation method for this essential luminophore. This patent addresses the longstanding bottlenecks in AMPPD manufacturing by introducing a novel catalytic system that replaces hazardous and complex traditional routes with a safer, more efficient methodology. The core innovation lies in the formation of the key intermediate, substituted phenylmethoxy-methylene adamantane, through a streamlined olefination reaction catalyzed by Titanium Tetrachloride (TiCl4) in combination with common metal reductants. This technological breakthrough not only enhances the purity profile of the final product but also fundamentally reshapes the economic feasibility of producing high-performance diagnostic reagents on a commercial scale.
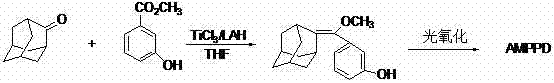
For R&D directors and process chemists, understanding the mechanistic shift provided by this patent is crucial for evaluating potential technology transfers. The disclosed method circumvents the limitations of earlier syntheses that relied on unstable intermediates or dangerous reagents, offering a pathway that is both chemically elegant and industrially pragmatic. By leveraging the reducing power of metals like Zinc, Tin, or Lithium Aluminum Hydride in the presence of TiCl4, the process generates low-valent titanium species in situ that effectively mediate the coupling of 2-adamantanone and 3-hydroxybenzoate derivatives. This approach ensures a high-yielding conversion to the methylene-bridged intermediate, which is subsequently converted to AMPPD via standard photooxidation. The implications for supply chain stability are profound, as the reliance on bespoke or highly regulated precursors is minimized in favor of bulk commodity chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of AMPPD has been plagued by significant technical and safety hurdles that hindered its widespread adoption and kept market prices artificially high. Prior art, including various United States patents and earlier Chinese filings, often depended on Grignard reagents or complex silyl-protected intermediates, necessitating up to seven distinct reaction steps to reach the final target. These multi-step sequences inherently suffer from cumulative yield losses, where the efficiency of each individual transformation compounds to result in a disappointing overall output. Furthermore, the use of Grignard reagents introduces severe operational risks, requiring strictly anhydrous conditions and specialized handling equipment to prevent violent exothermic reactions. Perhaps most critically, previous titanium-mediated approaches utilized anhydrous titanium trichloride (TiCl3), a substance known for its pyrophoric nature and tendency to spontaneously combust upon exposure to air or moisture. This extreme hazard profile not only endangered personnel but also imposed rigorous and costly engineering controls on manufacturing facilities, effectively barring many producers from entering the market.
The Novel Approach
In stark contrast to these perilous legacy methods, the technology outlined in CN102030779A presents a paradigm shift towards safety and simplicity without compromising chemical efficacy. The novel approach utilizes a catalytic system composed of Titanium Tetrachloride (TiCl4) paired with readily available reductants such as metallic Zinc (Zn), metallic Tin (Sn), or Lithium Aluminum Hydride (LAH). This substitution is transformative because TiCl4, while moisture-sensitive, does not possess the spontaneous combustion characteristics of TiCl3, thereby drastically lowering the danger coefficient of the entire operation. The reaction proceeds smoothly in tetrahydrofuran (THF) under nitrogen protection, where the in situ generation of the active low-valent titanium species facilitates the direct coupling of 2-adamantanone and methyl 3-hydroxybenzoate. As illustrated in the reaction scheme below, this single-step formation of the key olefinic intermediate bypasses the need for protecting group manipulations and extensive purification sequences required by older routes.
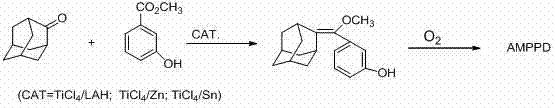
The versatility of this new catalytic system allows manufacturers to select the reductant that best aligns with their specific cost structures and safety protocols, whether opting for the mildness of Zinc or the potency of LAH. The resulting intermediate is obtained in high yield as a white solid with a sharp melting point, indicating excellent purity that simplifies downstream processing. By condensing the synthetic pathway and eliminating the most hazardous reagents, this method paves the way for the reliable mass production of AMPPD, ensuring a consistent supply of this critical component for the global diagnostics industry. The transition from a seven-step hazardous process to a streamlined, safer catalytic cycle represents a significant maturation of the manufacturing technology for chemiluminescent materials.
Mechanistic Insights into TiCl4-Mediated Reductive Coupling
To fully appreciate the technical sophistication of this patent, one must delve into the mechanistic underpinnings of the Titanium Tetrachloride-mediated reductive coupling. The reaction operates on principles similar to the McMurry coupling but is adapted here for the specific steric and electronic requirements of the adamantane scaffold. When TiCl4 is mixed with a reducing metal like Zinc or LAH in THF, it undergoes reduction to form low-valent titanium species, often described as Ti(0) or Ti(II) clusters. These electron-rich species act as powerful oxygen scavengers, coordinating with the carbonyl oxygen of the 2-adamantanone and the ester carbonyl of the hydroxybenzoate derivative. This coordination weakens the carbon-oxygen bonds, facilitating the elimination of oxygen and the subsequent formation of the carbon-carbon double bond that links the adamantane cage to the aromatic ring. The presence of the methoxy group and the phenolic hydroxyl requires careful management of stoichiometry and reaction conditions to prevent side reactions, yet the patented protocol demonstrates remarkable tolerance.
From an impurity control perspective, this mechanism offers distinct advantages over nucleophilic additions like Grignard reactions. In Grignard processes, over-addition or incomplete reaction can lead to difficult-to-remove alcohol byproducts or unreacted starting materials that persist through multiple steps. In the TiCl4/Zn or TiCl4/Sn system, the driving force of the reductive coupling pushes the equilibrium strongly towards the olefin product, minimizing the formation of saturated alcohol impurities. Furthermore, the use of triethylamine as an additive in the workup or reaction phase helps to neutralize acidic byproducts and stabilize the reaction mixture, preventing the degradation of the sensitive enol ether linkage. The high purity of the isolated intermediate, evidenced by the clean NMR spectra reported in the embodiments, suggests that the catalytic cycle is highly selective. This selectivity is paramount for pharmaceutical and diagnostic applications, where trace metal contaminants or organic impurities can interfere with the enzymatic activity or luminescent properties of the final AMPPD substrate.
How to Synthesize AMPPD Key Intermediate Efficiently
The practical execution of this synthesis requires strict adherence to anhydrous and oxygen-free conditions to maintain the activity of the low-valent titanium catalyst. The process begins with the activation of the catalyst system, followed by the controlled addition of substrates to manage the exotherm and ensure complete conversion. Detailed operational parameters, including temperature ramps and quenching protocols, are critical for maximizing yield and safety. For a comprehensive, step-by-step guide on executing this reaction in a pilot or production setting, please refer to the standardized protocol below.
- Prepare the low-valent titanium catalyst system by reacting anhydrous TiCl4 with a reductant such as Zinc powder, Lithium Aluminum Hydride, or Tin in THF under inert atmosphere.
- Introduce 2-Adamantanone and Methyl 3-hydroxybenzoate to the activated catalyst mixture and reflux to facilitate the olefination reaction forming the key methylene bridge.
- Quench the reaction carefully with water, extract the organic phase, and purify the resulting white solid intermediate via silica gel chromatography before final photooxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN102030779A translates directly into tangible operational benefits and risk mitigation. The shift away from pyrophoric titanium trichloride and complex Grignard reagents eliminates the need for specialized storage facilities and expensive safety infrastructure, leading to a substantial reduction in overhead costs. Moreover, the reliance on commodity chemicals like Zinc powder and Titanium Tetrachloride, which are produced on a massive global scale, ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with niche fine chemical intermediates. This stability is crucial for maintaining continuous production schedules for diagnostic kits, where interruptions can have significant downstream impacts on healthcare providers.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route offers profound economic advantages by collapsing multiple reaction steps into a single, high-yielding transformation. By eliminating the need for protecting group strategies and the associated reagents and solvents, the overall material cost per kilogram of product is significantly decreased. Additionally, the simplified work-up procedure, which involves basic extraction and crystallization rather than complex chromatographic separations at every stage, reduces labor hours and solvent consumption. This efficiency gain allows for a more competitive pricing structure for the final AMPPD product, enabling diagnostic companies to improve their margins or pass savings on to end-users without sacrificing quality.
- Enhanced Supply Chain Reliability: Sourcing raw materials for chemical synthesis is often a bottleneck, particularly when those materials are hazardous or produced by limited suppliers. The new method utilizes reductants like Zinc and Tin, which are abundant industrial metals with robust global supply networks. This abundance mitigates the risk of raw material shortages that could halt production lines. Furthermore, the reduced hazard profile of the reagents simplifies logistics and transportation, as the materials do not require the same level of regulatory compliance and special handling as pyrophoric substances. This ease of transport ensures that manufacturing sites can maintain adequate inventory levels with greater flexibility and lower logistical costs.
- Scalability and Environmental Compliance: Scaling a chemical process from the bench to the tonne scale often reveals hidden inefficiencies and safety hazards, but this novel catalytic system is inherently designed for enlargement. The use of heterogeneous catalysts or easily manageable slurries facilitates heat transfer and mixing in large reactors, addressing one of the primary challenges in scale-up. From an environmental standpoint, the reduction in step count inherently lowers the E-factor (mass of waste per mass of product), as fewer solvents and reagents are consumed and discarded. The avoidance of heavy metal catalysts that require complex removal steps also simplifies wastewater treatment, ensuring that the manufacturing process aligns with increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this AMPPD synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this route for commercial production.
Q: Why is the TiCl4/Zn catalyst system preferred over traditional Grignard reagents for AMPPD synthesis?
A: Traditional Grignard routes involve hazardous reagents and complex multi-step sequences (up to 7 steps) with lower overall yields. The TiCl4/Zn system described in CN102030779A utilizes common, large-scale industrial chemicals that are significantly safer to handle, eliminating the pyrophoric risks associated with older titanium trichloride methods while simplifying the purification process.
Q: What are the primary safety advantages of this novel catalytic method?
A: The patent highlights that previous methods using anhydrous titanium trichloride (TiCl3) posed severe safety risks due to spontaneous combustion upon contact with air or moisture. The new method employs Titanium Tetrachloride (TiCl4) combined with stable metals like Zinc or Tin, which drastically reduces the danger coefficient, making the process suitable for large-scale industrial production without specialized explosion-proof infrastructure.
Q: How does this synthesis route impact the cost of AMPPD for immunoassay applications?
A: By reducing the number of synthetic steps and utilizing inexpensive, commercially available catalysts instead of exotic or highly hazardous reagents, the overall manufacturing cost is substantially lowered. Furthermore, the higher yield of the key intermediate and the simplified work-up procedure contribute to a more economically viable supply chain for high-purity chemiluminescence materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable AMPPD Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust manufacturing capabilities. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry described in CN102030779A can be realized at the volumes your business demands. Our state-of-the-art facilities are equipped to handle the specific requirements of low-valent titanium chemistry, including rigorous inert atmosphere controls and advanced purification suites. We are committed to delivering high-purity AMPPD and its intermediates that meet stringent purity specifications, verified by our rigorous QC labs to ensure consistency batch after batch.
We invite you to collaborate with us to leverage this advanced synthesis technology for your diagnostic product lines. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our AMPPD intermediates and to discuss route feasibility assessments for your next project. Together, we can secure a sustainable and cost-effective supply of critical chemiluminescence materials for the global healthcare market.
