Advanced AgOTf-Catalyzed Synthesis of 1-(N-Substituted Amino)isoquinolines for Commercial Scale-Up
Advanced AgOTf-Catalyzed Synthesis of 1-(N-Substituted Amino)isoquinolines for Commercial Scale-Up
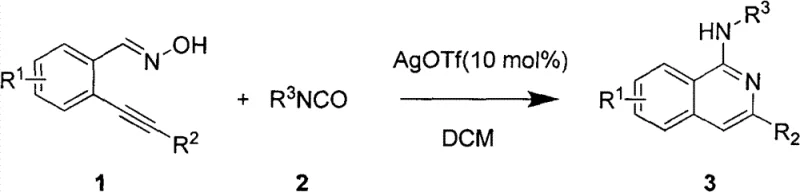
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that possess significant biological activity. Patent CN102503887B introduces a groundbreaking methodology for the preparation of 1-(N-substituted amino)isoquinoline compounds, a class of molecules renowned for their potent anti-tumor and pharmacological properties. This innovation addresses critical bottlenecks in traditional synthesis by utilizing a Lewis acid-catalyzed cyclization strategy that operates under exceptionally mild conditions. By leveraging ortho-alkynyl benzaldoximes and isocyanates as key building blocks, this process eliminates the need for hazardous reagents and extreme reaction parameters, thereby establishing a new standard for efficiency in fine chemical manufacturing. For global procurement teams and R&D directors, this technology represents a pivotal shift towards safer, more sustainable, and cost-effective production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-(N-substituted amino)isoquinoline derivatives has relied heavily on the nucleophilic substitution of corresponding halo-isoquinoline compounds. This conventional pathway is fraught with significant operational challenges and safety hazards that complicate large-scale manufacturing. Typically, the amination step requires the use of highly basic conditions and elevated temperatures, which can lead to thermal degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, the preparation of the requisite halo-isoquinoline precursors often necessitates the use of phosphorus oxychloride (POCl3), an extremely corrosive and hazardous reagent that demands specialized handling equipment and rigorous waste treatment protocols. These factors collectively contribute to increased production costs, extended lead times, and substantial environmental burdens, making the traditional route less attractive for modern, green chemistry-focused supply chains.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102503887B offers a streamlined, one-pot synthetic strategy that fundamentally reshapes the production landscape. This novel approach utilizes the reaction between ortho-alkynyl benzaldoximes and isocyanates, catalyzed by a Lewis acid system, to achieve concurrent intramolecular rearrangement and ring-closure. The process proceeds efficiently at room temperature in a neutral environment, completely obviating the need for strong bases or corrosive chlorinating agents. This mildness not only preserves the integrity of diverse functional groups but also drastically simplifies the workup procedure, allowing for direct purification via column chromatography. The versatility of this method is evidenced by its compatibility with a wide array of substrates, enabling the rapid generation of structurally diverse libraries essential for drug discovery programs.
Mechanistic Insights into AgOTf-Catalyzed Cyclization
The core of this technological advancement lies in the precise activation of the alkyne moiety by the silver triflate (AgOTf) catalyst. As a potent Lewis acid, AgOTf coordinates with the triple bond of the ortho-alkynyl benzaldoxime, increasing its electrophilicity and facilitating the nucleophilic attack by the nitrogen atom of the isocyanate. This interaction triggers a cascade of electronic rearrangements that drive the intramolecular cyclization, ultimately forming the isoquinoline core with high regioselectivity. The mechanism ensures that the reaction proceeds through a well-defined transition state, minimizing the formation of regioisomers and other structural byproducts that typically plague heterocyclic synthesis. Understanding this catalytic cycle is crucial for R&D directors aiming to optimize reaction parameters for specific substrate classes, as the electronic nature of the substituents can be fine-tuned to enhance reaction kinetics without compromising yield.
From an impurity control perspective, the neutrality of the reaction medium plays a pivotal role in ensuring high product purity. Traditional basic conditions often promote hydrolysis or polymerization side reactions, especially when dealing with sensitive ester or amide functionalities present in the isocyanate component. By maintaining a neutral pH throughout the transformation, this novel method effectively suppresses these degradation pathways, resulting in a cleaner crude reaction mixture. This inherent selectivity reduces the burden on downstream purification processes, such as crystallization or chromatography, thereby improving the overall mass balance of the manufacturing process. For quality assurance teams, this translates to a more consistent impurity profile and a higher likelihood of meeting stringent pharmacopeial specifications for active pharmaceutical ingredients (APIs) and their intermediates.
How to Synthesize 1-(N-Substituted Amino)isoquinoline Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to facilitate easy adoption in both laboratory and pilot plant settings. The protocol involves dissolving the ortho-alkynyl benzaldoxime and the chosen isocyanate in a common organic solvent, typically methylene dichloride, to ensure homogeneity. Following this, a catalytic amount of AgOTf is introduced to initiate the cyclization at ambient temperature. The reaction is allowed to stir for a defined period, usually around 24 hours, to ensure complete conversion of the starting materials. Upon completion, the solvent is removed under reduced pressure, and the residue is subjected to standard purification techniques. This operational simplicity makes it an ideal candidate for rapid scale-up, reducing the technical barriers often associated with complex heterocyclic chemistry.
- Dissolve ortho-alkynyl benzaldoxime and isocyanate in an organic solvent such as methylene dichloride (DCM) to form a homogeneous reaction mixture.
- Add the Lewis acid catalyst, specifically Silver Triflate (AgOTf), at a molar ratio of approximately 10 mol% relative to the substrate.
- Stir the reaction mixture at room temperature for approximately 24 hours until completion, then concentrate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this AgOTf-catalyzed route offers compelling economic and logistical advantages over incumbent technologies. The elimination of hazardous reagents like POCl3 and the avoidance of high-temperature, high-pressure conditions significantly lower the capital expenditure required for reactor infrastructure and safety systems. Furthermore, the mild reaction conditions reduce energy consumption, aligning with corporate sustainability goals and reducing the overall carbon footprint of the manufacturing process. The robustness of the method across various substrates ensures a stable supply of diverse intermediates, mitigating the risk of production delays caused by substrate-specific optimization issues. These factors combine to create a more resilient and cost-efficient supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic sequence and the reduction in auxiliary material costs. By removing the need for expensive noble metal catalysts often used in cross-coupling alternatives and avoiding the costly disposal of corrosive waste streams, the overall cost of goods sold (COGS) is substantially decreased. The high yields reported in the patent embodiments, ranging significantly above 80% for various derivatives, indicate excellent atom economy and minimal raw material wastage. Additionally, the ability to run the reaction at room temperature eliminates the energy costs associated with heating or cooling large-scale reactors, further contributing to long-term operational savings.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, namely ortho-alkynyl benzaldoximes and isocyanates, are generally commercially available or easily accessible from established chemical suppliers. This accessibility reduces dependency on single-source vendors for exotic precursors, thereby diversifying the supply base and enhancing continuity. The short reaction time and simple workup procedure also mean that batch turnover rates can be optimized, allowing manufacturers to fulfill orders with shorter lead times. For supply chain planners, this reliability is critical for maintaining inventory levels and ensuring uninterrupted production schedules for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the absence of exothermic hazards and the use of common solvents like dichloromethane, which are well-understood in industrial settings. The neutral nature of the reaction minimizes the generation of acidic or basic wastewater, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. The high purity of the crude product reduces the solvent load required for purification, further minimizing the environmental impact. This alignment with green chemistry principles not only future-proofs the manufacturing process against regulatory changes but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios. Understanding these nuances is essential for making informed decisions about process feasibility and resource allocation.
Q: What are the optimal reaction conditions for this isoquinoline synthesis?
A: The patent specifies using Silver Triflate (AgOTf) as the Lewis acid catalyst in methylene dichloride (DCM) at room temperature. The optimal molar ratio of substrate to catalyst to isocyanate is 1:0.1:1.1.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate scope. It tolerates electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, and trifluoromethyl on the aromatic rings.
Q: How does this route compare to traditional halo-isoquinoline amination?
A: Unlike traditional methods requiring harsh bases, high temperatures, or corrosive reagents like POCl3 for precursor synthesis, this novel approach operates under neutral, mild conditions with high yields and fewer side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(N-Substituted Amino)isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the AgOTf-catalyzed synthesis described in CN102503887B for the production of high-quality pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-volume supply without compromising on quality. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 1-(N-substituted amino)isoquinoline compounds we produce. We are committed to leveraging this innovative chemistry to deliver cost-effective solutions that accelerate your drug development timelines.
We invite you to collaborate with us to explore how this advanced synthetic route can optimize your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this method can improve your margins. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary candidates. Let us be your trusted partner in navigating the complexities of modern heterocyclic chemistry.
