Advanced Asymmetric Hydrogenation Technology for Scalable Diphenylalanine Intermediate Production
The pharmaceutical industry's relentless pursuit of metabolically stable peptide therapeutics has placed unnatural alpha-amino acids, specifically 3,3-diarylalanines, at the forefront of modern drug design. Patent CN101175715B introduces a groundbreaking methodology for the asymmetric hydrogenation of diphenylalanine derivatives, addressing a critical bottleneck in the synthesis of these high-value building blocks. This technology leverages a chiral diphosphine rhodium homogeneous hydrogenation catalyst to achieve exceptional enantioselectivity and catalytic activity, overcoming the historical limitations associated with tetrasubstituted alkene substrates. For R&D directors and process chemists, this patent represents a paradigm shift from inefficient stoichiometric resolutions to elegant, atom-economical catalytic processes. The ability to access single enantiomers of 3,3-diarylalanines with high optical purity is essential for developing next-generation thrombin inhibitors and HIV protease inhibitors, making this intellectual property a cornerstone for reliable pharmaceutical intermediate supplier strategies aiming to secure robust supply chains for complex API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3-diarylalanines has been plagued by significant inefficiencies that hinder cost-effective manufacturing. Traditional routes, such as those reported by Josien et al., rely heavily on the use of stoichiometric chiral auxiliaries coupled with low-temperature alkylation steps, which are not only operationally cumbersome but also generate substantial chemical waste. Furthermore, classical resolution methods, while conceptually simple, are inherently flawed due to their maximum theoretical yield of 50%, necessitating the disposal or recycling of the unwanted enantiomer. This intrinsic limitation drastically increases the cost of goods sold (COGS) and complicates waste management protocols, posing severe challenges for supply chain heads focused on sustainability and efficiency. Additionally, existing catalytic methods using standard ligands like DuPhos often fail to deliver sufficient activity when confronted with the steric bulk of tetrasubstituted enamides, leading to prolonged reaction times and the need for excessive catalyst loadings that are economically unviable for large-scale production.
The Novel Approach
The novel approach disclosed in the patent revolutionizes this landscape by employing a specialized chiral-diphosphine rhodium catalyst system featuring the PhanePhos ligand. Unlike conventional trisubstituted enamides, the tetrasubstituted enamide precursors used in this method present a formidable steric challenge that typically results in low hydrogenation rates. However, the unique structural rigidity and electronic properties of the PhanePhos ligand enable the rhodium center to effectively coordinate and reduce these bulky substrates with remarkable speed and precision. This innovation allows for significantly reduced catalyst loadings and shorter processing times, directly translating to enhanced throughput and lower operational costs. By shifting the synthetic strategy to this highly active asymmetric hydrogenation, manufacturers can bypass the yield ceilings of resolution methods and the harsh conditions of auxiliary-based syntheses, establishing a streamlined pathway for cost reduction in API manufacturing that aligns perfectly with green chemistry principles.
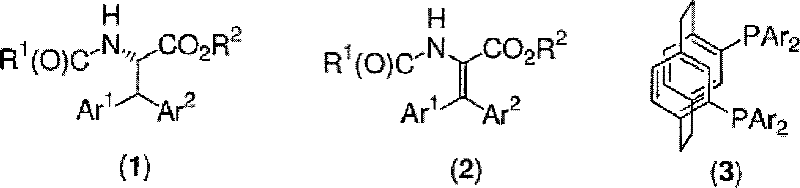
Mechanistic Insights into Rh-PhanePhos Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the sophisticated interplay between the rhodium metal center and the PhanePhos ligand architecture. The PhanePhos ligand, characterized by its [2.2]paracyclophane backbone, imposes a specific chiral pocket that dictates the facial selectivity of hydrogen addition to the prochiral double bond. In the context of 3,3-diarylalanine synthesis, the substrate possesses two bulky aryl groups at the beta-position, creating significant steric congestion around the reactive alkene. Standard ligands often fail to differentiate the enantiotopic faces of such hindered alkenes effectively, or they bind too weakly to facilitate rapid turnover. The PhanePhos-rhodium complex, however, maintains a robust coordination sphere that stabilizes the catalytic cycle while allowing the substrate to approach in a geometry that favors the formation of the desired enantiomer. This mechanistic advantage ensures that even with the steric demand of the diphenyl substitution, the reaction proceeds with high turnover frequencies, minimizing the residence time required in the reactor and maximizing the space-time yield of the process.
Furthermore, the control of impurity profiles is intrinsically linked to the high enantioselectivity of this catalytic system. In asymmetric hydrogenations, low selectivity often leads to the formation of the opposite enantiomer, which acts as a difficult-to-remove impurity that can compromise the safety and efficacy of the final drug product. The PhanePhos system described in the patent consistently delivers enantiomeric excess (ee) values exceeding 90%, and often reaching higher levels depending on the specific substrate substitution. This high level of stereocontrol simplifies downstream purification, as the need for extensive chromatographic separation or recrystallization cycles is markedly reduced. For quality assurance teams, this means a more consistent and predictable impurity profile, reducing the risk of batch failures and ensuring that the high-purity 3,3-diarylalanine derivatives meet the stringent specifications required for clinical and commercial applications without requiring costly additional processing steps.
How to Synthesize 3,3-Diarylalanine Derivatives Efficiently
The practical implementation of this synthesis involves dissolving the tetrasubstituted enamide substrate in a protic solvent, preferably methanol, which facilitates proton transfer during the catalytic cycle. The reaction is conducted under a hydrogen atmosphere, typically at moderate pressures of 5-15 bar, which balances safety concerns with the kinetic requirements for efficient hydrogen uptake. Temperature control is maintained between 20°C and 60°C to optimize the reaction rate without compromising the stability of the catalyst or the product. The detailed standardized synthesis steps, including precise molar ratios, catalyst activation procedures, and workup protocols, are outlined below to guide process development teams in replicating this high-efficiency route.
- Prepare the reaction vessel by charging the tetrasubstituted enamide substrate and the chiral Rh-PhanePhos pre-catalyst under inert atmosphere.
- Dissolve the mixture in a suitable alcoholic solvent such as methanol and pressurize the reactor with hydrogen gas to 5-15 bar.
- Maintain the reaction temperature between 20-60°C with stirring until conversion is complete, followed by workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this PhanePhos-catalyzed hydrogenation process offers transformative economic and logistical benefits. By replacing stoichiometric chiral auxiliaries or resolution steps with a catalytic asymmetric process, the overall material efficiency of the synthesis is drastically improved. This shift eliminates the need to purchase and dispose of expensive chiral resolving agents, thereby reducing the raw material bill and minimizing hazardous waste generation. The high activity of the catalyst system means that less precious metal is required per kilogram of product, mitigating the financial risk associated with fluctuating rhodium prices. Moreover, the simplified workflow reduces the number of unit operations, leading to lower utility consumption and labor costs, which collectively contribute to substantial cost savings in the manufacturing of these critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition to this catalytic method fundamentally alters the cost structure of producing 3,3-diarylalanines. By achieving high conversion rates with low catalyst loadings, the process minimizes the consumption of expensive noble metals and ligands. The elimination of resolution steps effectively doubles the theoretical yield compared to traditional racemic synthesis followed by separation, meaning that the same amount of starting material produces twice the amount of usable product. This inherent efficiency gain drives down the cost per kilogram significantly, allowing for more competitive pricing in the global market for API intermediates while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex multi-step syntheses that rely on scarce or hard-to-source reagents. This hydrogenation method utilizes readily available enamide precursors and standard hydrogen gas, reducing dependency on specialized chiral pool materials that may face supply constraints. The robustness of the PhanePhos catalyst system ensures consistent batch-to-batch performance, reducing the likelihood of production delays caused by failed reactions or off-spec material. This reliability is crucial for maintaining just-in-time inventory levels and meeting the tight delivery schedules demanded by multinational pharmaceutical clients who require uninterrupted supply of key building blocks for their drug pipelines.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, particularly with exothermic reactions or sensitive catalysts. The homogeneous nature of this rhodium-catalyzed hydrogenation is well-suited for scale-up in standard high-pressure reactors, facilitating the commercial scale-up of complex amino acids without requiring exotic equipment. Furthermore, the use of alcohol solvents like methanol and the reduction of waste streams align with increasingly strict environmental regulations. By minimizing solvent usage and avoiding the generation of stoichiometric salt waste associated with resolution methods, this process supports corporate sustainability goals and reduces the regulatory burden associated with waste disposal and environmental compliance audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the patent specifications and experimental data, providing clarity on the operational parameters and expected outcomes of the PhanePhos-catalyzed process. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is the PhanePhos ligand preferred over DuPhos for this specific reaction?
A: The PhanePhos ligand provides a unique steric environment that accommodates the bulky tetrasubstituted alkene substrate, offering superior activity and enantioselectivity compared to standard bis(phosphacycle) ligands like DuPhos which struggle with steric hindrance in this specific transformation.
Q: What are the typical reaction conditions for this asymmetric hydrogenation?
A: The process typically operates under mild conditions, utilizing methanol as the solvent, hydrogen pressures between 5-15 bar, and temperatures ranging from 20°C to 60°C, ensuring safety and ease of scale-up.
Q: Can this method be scaled for industrial API production?
A: Yes, the homogeneous nature of the rhodium catalyst and the use of standard hydrogenation equipment make this method highly amenable to commercial scale-up, addressing the supply chain needs for large-volume unnatural amino acid production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Diarylalanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality unnatural amino acids in the development of life-saving therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3,3-diarylalanine derivative performs consistently in your downstream synthesis. Our facility is equipped to handle the specific requirements of homogeneous catalysis, including high-pressure hydrogenation capabilities and advanced analytical tools for enantiomeric excess determination.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of Patent CN101175715B can optimize your supply chain and reduce your overall manufacturing costs. Let us be your partner in bringing innovative peptide therapies to market faster and more economically.
