Revolutionizing Adamantane Derivative Production: A Deep Dive into Zinc-Mediated Alkylation for Commercial Scale-Up
Revolutionizing Adamantane Derivative Production: A Deep Dive into Zinc-Mediated Alkylation for Commercial Scale-Up
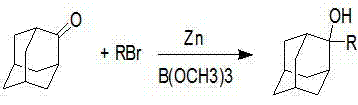
The landscape of fine chemical synthesis is constantly evolving, driven by the relentless pursuit of efficiency, sustainability, and cost-effectiveness. A pivotal advancement in this domain is detailed in patent CN102276421B, which discloses a robust method for preparing 2-substituted-2-adamantane alcohol compounds. This technology represents a significant departure from conventional organometallic approaches, utilizing a zinc-mediated reaction system that operates under remarkably mild conditions without the need for rigorous air isolation. For R&D directors and procurement strategists alike, this innovation offers a compelling pathway to secure high-purity intermediates essential for photo-resist monomers, photochromic compounds, and advanced pharmaceutical applications. By leveraging borate esters and zinc powder, this process not only enhances yield but also drastically simplifies the downstream purification workflow, positioning it as a cornerstone for modern industrial chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of 2-adamantanone has been plagued by significant technical hurdles that hinder efficient large-scale production. Traditional methodologies predominantly rely on organomagnesium (Grignard) or organoaluminum compounds, which suffer from inherent selectivity issues. As documented in prior art such as Tetrahedron Lett., 31, 3151 (1990), these reagents frequently induce preferential reduction reactions rather than the desired alkylation, resulting in a substantial formation of reduced byproducts like 2-adamantanol instead of the target alkylated alcohol. In some comparative studies, the selectivity for the alkylated product was as low as 25%, rendering the process economically unviable due to excessive raw material waste and complex separation requirements. Furthermore, alternative routes utilizing organolithium reagents, while offering better selectivity, introduce severe operational constraints, including the necessity for cryogenic temperatures around -78°C and strictly anhydrous, oxygen-free environments. These harsh conditions demand specialized equipment and drive up energy consumption and safety risks, making them ill-suited for cost-effective commercial manufacturing.
The Novel Approach
In stark contrast, the novel approach outlined in CN102276421B introduces a transformative zinc-mediated protocol that effectively circumvents these longstanding challenges. By employing a combination of 2-adamantanone, a borate ester, and a halogenated compound in the presence of zinc powder, the reaction achieves high conversion rates with exceptional selectivity. This method operates at moderate temperatures ranging from 0°C to 80°C, eliminating the need for energy-intensive cryogenic cooling. The reaction mechanism inherently suppresses the reduction side pathways that plague Grignard chemistry, ensuring that the majority of the starting ketone is converted into the valuable 2-substituted-2-adamantane alcohol. Moreover, the process demonstrates remarkable tolerance to reaction conditions, requiring no strict air isolation, which significantly lowers the barrier for implementation in standard chemical reactors. This shift from sensitive organometallics to a more robust zinc-borate system marks a critical evolution in synthetic strategy, enabling safer and more reliable production workflows.
Mechanistic Insights into Zinc-Mediated Barbier-Type Alkylation
The core of this technological breakthrough lies in the unique mechanistic interplay between zinc, the borate ester, and the halogenated substrate. Unlike traditional Grignard formations where the organometallic species is pre-formed, this process likely proceeds via a Barbier-type mechanism where the organozinc species is generated in situ. The zinc powder activates the carbon-halogen bond of the alkyl halide, forming a reactive organozinc intermediate that is subsequently transmetallated or coordinated by the borate ester. This coordination enhances the nucleophilicity of the alkyl group while simultaneously modulating its reactivity to favor addition to the carbonyl carbon of the adamantanone over single-electron transfer reduction pathways. The presence of the borate ester is crucial, acting as a Lewis acid activator that stabilizes the transition state and directs the stereochemical outcome, although the rigid adamantane cage largely dictates the geometry. This synergistic effect ensures that the reaction proceeds with high fidelity, minimizing the formation of pinacol coupling byproducts or reduced alcohols that typically contaminate crude reaction mixtures in metal-mediated reductions.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity intermediates required in sensitive applications like semiconductor photo-resists. The suppression of reduction byproducts means that the crude reaction mixture is significantly cleaner, reducing the burden on downstream purification units. In conventional processes, separating the reduced alcohol (2-adamantanol) from the alkylated product often requires tedious chromatography or multiple recrystallizations, which erodes overall yield. Here, the selectivity is intrinsic to the catalytic system, allowing for simple aqueous workups to remove inorganic zinc salts and boron residues. The use of n-butyl ether as a solvent further aids in impurity management; its immiscibility with water allows for efficient extraction of organic products while leaving polar inorganic impurities in the aqueous phase. This results in a final product with purity levels exceeding 99% as confirmed by gas chromatography analysis in the patent examples, meeting the stringent specifications demanded by the electronics and pharmaceutical industries.
How to Synthesize 2-Substituted-2-Adamantane Alcohol Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and solvent selection to maximize the benefits of the zinc-mediated system. The process begins by dissolving the 2-adamantanone substrate and the borate ester in a suitable organic solvent, with n-butyl ether being the preferred choice due to its favorable physical properties. Zinc powder is then introduced to the mixture, followed by the controlled addition of the halogenated compound. The reaction is exothermic and self-sustaining once initiated, proceeding smoothly at elevated temperatures without external cooling. Following the reaction period, typically lasting between 3 to 6 hours depending on the specific halide used, the mixture is quenched with a saturated ammonium chloride solution to decompose any remaining organometallic species. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Mix 2-adamantanone, borate ester (e.g., trimethyl borate), and organic solvent (preferably n-butyl ether) in a reactor under agitation.
- Add zinc powder to the mixture, followed by the dropwise addition of the halogenated compound (e.g., methyl iodide or alkyl bromide) at temperatures between 0°C and 80°C.
- Upon completion, quench with saturated ammonium chloride, separate layers, wash the organic phase, and purify the product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this zinc-mediated synthesis offers profound strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing expensive and hazardous organolithium or Grignard reagents with commodity-grade zinc powder and stable borate esters, manufacturers can significantly reduce raw material procurement costs and mitigate supply risks associated with specialized reagents. Furthermore, the elimination of cryogenic requirements translates to lower energy consumption and reduced capital expenditure on specialized low-temperature reactors, directly impacting the bottom line through operational expenditure (OpEx) savings. The robustness of the process also enhances supply continuity, as it is less susceptible to batch failures caused by minor fluctuations in moisture or oxygen levels, ensuring consistent delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The economic implications of this process are substantial, primarily driven by the replacement of high-cost reagents and the optimization of solvent usage. Traditional organolithium routes require stoichiometric amounts of expensive lithium metal and specialized solvents like THF or hexanes, which are costly to recover. In contrast, the zinc-mediated method utilizes inexpensive zinc powder and n-butyl ether, a solvent known for its high recovery rate and ease of separation from water. The ability to recycle the solvent efficiently minimizes waste disposal costs and reduces the overall volume of raw materials required per kilogram of product. Additionally, the high yield and selectivity mean that less starting material is wasted on byproducts, further driving down the cost of goods sold (COGS) and improving margin potential for high-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically improved by the mild operating conditions and the stability of the reagents involved. Organolithium reagents are notoriously unstable and require cold chain logistics for transport and storage, introducing potential points of failure in the supply network. Zinc powder and alkyl halides, however, are stable solids and liquids that can be stored under ambient conditions, simplifying inventory management and reducing logistics costs. The process tolerance to air and moisture also means that production can proceed with fewer interruptions due to environmental controls, leading to more predictable lead times. This reliability is essential for maintaining just-in-time inventory systems and meeting the tight delivery windows demanded by global pharmaceutical and electronic chemical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of extreme thermal or pressure hazards. The reaction generates minimal hazardous waste compared to traditional methods, as the inorganic byproducts (zinc salts) are non-toxic and easily managed. The use of n-butyl ether significantly reduces the organic load in wastewater, facilitating compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and effluent treatment. This environmental compatibility not only reduces regulatory risk but also aligns with the sustainability goals of major multinational corporations, making the supply chain more attractive to eco-conscious partners and investors looking for green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc-mediated synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in patent CN102276421B, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific intermediate manufacturing needs.
Q: Why is the zinc-mediated method superior to traditional Grignard reagents for adamantane alkylation?
A: Traditional Grignard reagents often cause preferential reduction reactions, leading to low yields of the desired alkylated product (often below 30%). The zinc-mediated method described in CN102276421B suppresses this reduction side reaction, achieving yields exceeding 90% while operating under much milder conditions without strict air isolation.
Q: What are the specific advantages of using n-butyl ether as the solvent in this process?
A: N-butyl ether offers excellent phase separation properties with water, which significantly simplifies the workup process. Furthermore, its higher boiling point facilitates high solvent recovery rates, reducing both raw material costs and the volume of organic contaminants in wastewater, aligning with green chemistry principles.
Q: Can this synthesis method be scaled for industrial production of photo-resist monomers?
A: Yes, the process is highly suitable for industrial scale-up. It eliminates the need for cryogenic conditions (-78°C) required by organolithium routes and avoids the instability issues of Grignard reagents. The simple purification steps (washing, filtration, recrystallization) ensure consistent high purity (99%+) essential for electronic and optical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted-2-Adamantane Alcohol Supplier
The technological potential of zinc-mediated alkylation for adamantane derivatives is immense, offering a clear path to high-quality, cost-effective intermediates for the global market. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 2-substituted-2-adamantane alcohol meets the exacting standards required for photo-resist and pharmaceutical applications. We understand the critical nature of supply chain consistency and are committed to delivering products that empower your R&D and production teams.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics and our proven track record in fine chemical manufacturing.
