Advanced Synthesis of Posaconazole Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antifungal agents, and patent CN108084049B presents a significant breakthrough in the preparation of posaconazole intermediates. This specific intellectual property details a novel five-step synthesis route that transforms L-ethyl lactate into a key formyl hydrazine derivative, known as Compound 2, which serves as a pivotal building block for the final antifungal drug. Unlike traditional methods that struggle with liquid intermediates and complex purification, this invention leverages a strategic protection-deprotection sequence combined with TEMPO-mediated oxidation to ensure high stereochemical control. For R&D directors and procurement specialists, this patent represents a shift towards more manufacturable chemistry, offering a reliable posaconazole intermediate supplier pathway that mitigates the risks associated with low-yield resolution processes. The technical depth of this approach lies in its ability to convert oily, hard-to-handle substances into crystalline solids, thereby enhancing the overall quality control of the supply chain.
The strategic value of this patent extends beyond mere chemical novelty; it addresses the fundamental bottlenecks in antifungal API manufacturing. By establishing a route that avoids the need for chiral resolution in the later stages, the process inherently improves the optical purity of the final product without the substantial material loss typically associated with recrystallization of racemates. This is particularly critical for the production of high-purity posaconazole intermediate materials where regulatory standards demand stringent impurity profiles. The methodology described ensures that the chiral center introduced early in the synthesis is preserved and effectively induces the formation of the second chiral center, resulting in a diastereomerically enriched product. This level of precision is essential for partners seeking cost reduction in antifungal API manufacturing, as it minimizes the waste of expensive starting materials and reduces the environmental burden of solvent-intensive purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of posaconazole intermediates has been plagued by significant technical hurdles that compromise both efficiency and cost-effectiveness. As illustrated in prior art such as World Patent WO96/33178, conventional routes often rely on the resolution of racemic mixtures, a process that theoretically caps the maximum yield at 50% and requires extensive recycling of the unwanted enantiomer. 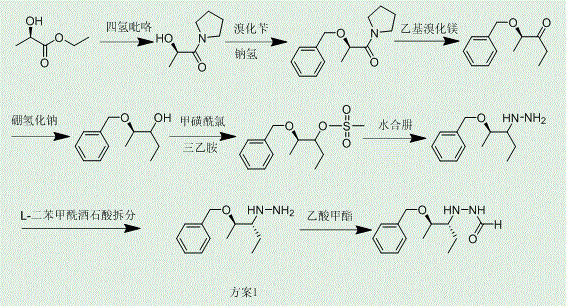 . Furthermore, many intermediates in these traditional pathways exist as oils at room temperature, making them exceptionally difficult to purify using standard crystallization techniques. This physical state necessitates the use of column chromatography, a method that is notoriously difficult to scale up for commercial scale-up of complex pharmaceutical intermediates due to high solvent consumption and low throughput. The accumulation of solvent residues and the inability to effectively remove trace impurities in liquid phases often lead to downstream reaction failures, affecting the quality of the final posaconazole product.
. Furthermore, many intermediates in these traditional pathways exist as oils at room temperature, making them exceptionally difficult to purify using standard crystallization techniques. This physical state necessitates the use of column chromatography, a method that is notoriously difficult to scale up for commercial scale-up of complex pharmaceutical intermediates due to high solvent consumption and low throughput. The accumulation of solvent residues and the inability to effectively remove trace impurities in liquid phases often lead to downstream reaction failures, affecting the quality of the final posaconazole product.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent introduces a paradigm shift by focusing on the physical state of the intermediates to drive purification efficiency. The new method strategically employs a bulky protecting group, 4-chloro-benzhydrol, which not only shields the hydroxyl group but also provides significant steric hindrance that aids in stereochemical induction. This design choice ensures that key intermediates, specifically Compound 5 and the final Compound 2, are solids at room temperature, allowing for purification via simple and scalable crystallization. This transition from liquid to solid intermediates eliminates the need for chromatographic separation, thereby drastically simplifying the workflow and enhancing process stability. For supply chain heads, this means reducing lead time for high-purity antifungal intermediates, as the operational complexity is significantly lowered, and the risk of batch failure due to purification issues is minimized. The robustness of this solid-phase handling capability is a key differentiator that supports consistent commercial production.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Grignard Alkylation
The core chemical innovation within this synthesis lies in the oxidation step utilizing TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) and sodium hypochlorite, which converts the alcohol intermediate into a reactive aldehyde with high selectivity. This catalytic system is preferred over traditional chromium-based oxidants due to its environmental profile and compatibility with sensitive functional groups present in the molecule. The mechanism involves the formation of an oxoammonium ion which abstracts a hydride from the alcohol, generating the carbonyl group while being regenerated by the hypochlorite co-oxidant. Crucially, the subsequent isolation of this aldehyde as a bisulfite adduct (Compound 5) serves as a purification checkpoint, trapping the aldehyde in a stable crystalline form that can be washed free of organic impurities. This step is vital for maintaining the integrity of the carbon skeleton before the final carbon-carbon bond-forming reaction, ensuring that the subsequent Grignard addition proceeds with minimal side reactions.
The final stage of the synthesis involves the addition of an ethyl Grignard reagent to the hydrazone derivative, a step that establishes the second chiral center critical for biological activity. The presence of the adjacent chiral center from the L-lactate starting material exerts a strong diastereofacial bias, directing the approach of the nucleophile to favor the desired stereoisomer. 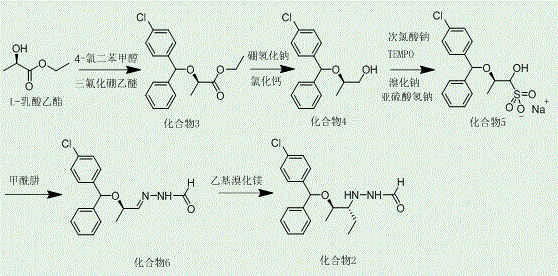 . This substrate-controlled asymmetric induction is highly efficient, negating the need for external chiral catalysts or resolution agents in the final step. The resulting Compound 2 is obtained as a solid with high optical purity, which can be further enhanced through recrystallization. This mechanistic elegance ensures that the process is not only chemically sound but also commercially viable, as it maximizes atom economy and minimizes the generation of difficult-to-separate diastereomeric impurities that could compromise the safety profile of the final drug substance.
. This substrate-controlled asymmetric induction is highly efficient, negating the need for external chiral catalysts or resolution agents in the final step. The resulting Compound 2 is obtained as a solid with high optical purity, which can be further enhanced through recrystallization. This mechanistic elegance ensures that the process is not only chemically sound but also commercially viable, as it maximizes atom economy and minimizes the generation of difficult-to-separate diastereomeric impurities that could compromise the safety profile of the final drug substance.
How to Synthesize Posaconazole Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions, particularly temperature control during the etherification and Grignard steps to prevent side reactions. The process begins with the protection of L-lactate, followed by reduction and the critical TEMPO oxidation, culminating in the condensation and alkylation steps that yield the target molecule. Each stage has been optimized to balance reaction rate with selectivity, ensuring that the intermediates remain stable throughout the sequence. For technical teams looking to adopt this methodology, the detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, solvent choices, and workup procedures required to achieve the reported yields and purity levels. Adhering to these parameters is essential for replicating the high quality and consistency demonstrated in the patent examples.
- Protect L-lactate hydroxyl using 4-chloro-benzhydrol and Lewis acid to form Compound 3.
- Reduce the ester group in Compound 3 to hydroxyl using metal hydride to obtain Compound 4.
- Oxidize Compound 4 to aldehyde using TEMPO and sodium hypochlorite, then isolate as solid sulfonate Compound 5.
- Condense Compound 5 with formylhydrazine to form Compound 6, followed by Grignard alkylation to yield target Compound 2.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The primary benefit is the substantial cost savings derived from the use of cheap and easily available raw materials, such as L-ethyl lactate and 4-chlorobenzhydrol, which are commodity chemicals with stable supply chains. By eliminating the need for expensive chiral resolution reagents and reducing the reliance on chromatographic purification, the overall cost of goods sold (COGS) is significantly lowered. This economic efficiency is further amplified by the high yield of the process, which ensures that less starting material is wasted, thereby maximizing the output per batch and improving the return on investment for manufacturing campaigns.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and expensive resolution agents translates directly into lower raw material costs. Furthermore, the ability to purify intermediates via crystallization rather than column chromatography drastically reduces solvent consumption and waste disposal costs. This streamlined approach minimizes the operational overhead associated with complex purification workflows, leading to a more competitive pricing structure for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of robust, non-proprietary reagents ensures that the supply chain is not vulnerable to single-source bottlenecks. Since the intermediates are solids, they possess better storage stability and are easier to transport compared to oily liquids, reducing the risk of degradation during logistics. This physical stability enhances supply continuity, allowing for larger batch sizes and longer shelf-life, which is critical for maintaining inventory levels and meeting the demanding production schedules of downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like crystallization and filtration that are well-understood in industrial settings. The avoidance of hazardous oxidants and the reduction in solvent waste align with green chemistry principles, facilitating easier regulatory approval and environmental compliance. This scalability ensures that the manufacturing capacity can be expanded from pilot scale to multi-ton commercial production seamlessly, supporting long-term growth and market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent data and are intended to clarify the operational benefits and technical feasibility for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into existing manufacturing portfolios.
Q: Why is the new synthesis route superior to conventional methods for posaconazole intermediates?
A: The new route avoids difficult chromatographic purification and resolution steps found in older methods. By generating solid intermediates like Compound 5 and Compound 2, it allows for purification via simple crystallization, significantly improving optical purity and chemical stability.
Q: How does this process impact the cost of goods for antifungal API manufacturing?
A: The process utilizes cheap and easily available raw materials such as L-ethyl lactate. Furthermore, the elimination of expensive resolution reagents and the ability to use crystallization instead of column chromatography drastically reduces operational costs and solvent consumption.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. The use of robust reaction conditions, such as TEMPO-catalyzed oxidation and standard Grignard reactions, combined with the isolation of stable solid intermediates, ensures high process stability and safety suitable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Posaconazole Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent methodologies like CN108084049B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of posaconazole intermediate meets the highest global standards. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical companies seeking to secure their supply chains for critical antifungal medications.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the potential impact of this technology on your bottom line. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable posaconazole intermediate supplier dedicated to driving innovation and efficiency in the pharmaceutical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
