Scalable Synthesis of Ortho-Amino Trifluoroacetophenone for Advanced Pharmaceutical Manufacturing
Scalable Synthesis of Ortho-Amino Trifluoroacetophenone for Advanced Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands robust and scalable synthetic routes for critical intermediates, particularly those serving as precursors for quinolone antibiotics. Patent CN113651707A introduces a groundbreaking preparation method for ortho-amino trifluoroacetophenone and its derivatives, addressing long-standing challenges in regioselectivity and yield. This innovative three-step protocol utilizes an amino protection strategy followed by directed ortho-metalation and subsequent deprotection, offering a superior alternative to conventional nitration pathways. By leveraging readily available raw materials and mild reaction conditions, this technology enables the production of high-purity intermediates essential for modern drug synthesis. The process demonstrates exceptional adaptability for large-scale manufacturing, ensuring consistent quality and supply continuity for global pharmaceutical partners seeking reliable sourcing solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-amino substituted trifluoroacetophenones has relied heavily on the direct nitration of benzene ring-substituted trifluoroacetophenones followed by reduction. However, this traditional approach is plagued by significant technical hurdles that hinder industrial application. The strong electron-withdrawing effect of the trifluoroacetyl group makes the aromatic ring highly deactivated towards electrophilic substitution, necessitating harsh nitration conditions that often lead to over-nitration or decomposition. Furthermore, controlling the nitration site is exceptionally difficult, resulting in complex mixtures of isomers that are arduous and costly to separate. These purification challenges drastically reduce the overall yield and compromise the purity profile required for pharmaceutical-grade intermediates. Consequently, the reliance on scarce starting materials and the generation of substantial hazardous waste render this legacy method economically and environmentally unsustainable for modern production needs.
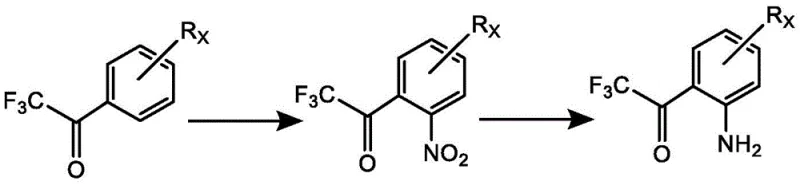
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a strategic sequence of amino protection, acylation, and deprotection to bypass the limitations of electrophilic aromatic substitution. By first converting the amino group into a stable amide, the process effectively masks the reactive nitrogen, preventing interference during the subsequent organometallic step. This protection allows for the precise introduction of the trifluoroacetyl group via directed ortho-lithiation, a technique renowned for its high regioselectivity. The reaction conditions are remarkably mild, avoiding the extreme temperatures and corrosive reagents associated with nitration. Moreover, the use of commercially available aniline derivatives as starting materials ensures a stable supply chain, while the streamlined workflow minimizes unit operations. This paradigm shift not only enhances the chemical efficiency but also aligns with green chemistry principles by reducing waste generation and energy consumption throughout the manufacturing lifecycle.
Mechanistic Insights into Directed Ortho-Metalation and Protection Strategy
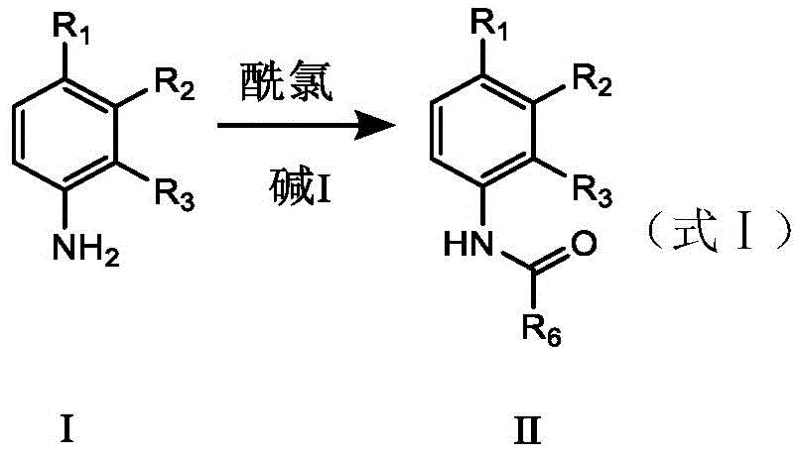
The core of this synthetic breakthrough lies in the sophisticated interplay between the protecting group and the organolithium reagent. In the initial step, the aniline derivative reacts with an acyl chloride in the presence of a base to form an amide intermediate. This transformation is critical as the resulting amide carbonyl oxygen acts as a Lewis basic site, coordinating with the lithium cation of the n-butyllithium reagent. This coordination directs the lithiation specifically to the ortho-position relative to the nitrogen, ensuring that the subsequent acylation occurs exactly where needed. The addition of TMEDA further enhances this process by depolymerizing the n-butyllithium aggregates, thereby increasing the nucleophilicity and reactivity of the lithiated species. Such precise control over the reaction trajectory eliminates the formation of meta- or para-substituted byproducts, which are common pitfalls in less controlled synthetic routes.
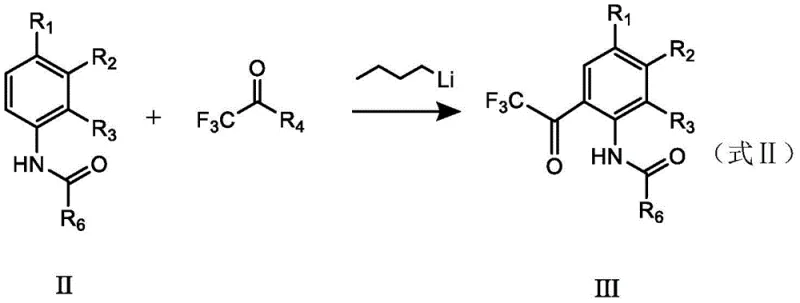
Following the lithiation, the introduction of the trifluoroacetyl moiety is achieved through reaction with a trifluoroacetic ester. The choice of ester is deliberate; the alcohol byproduct generated during this acylation is inert under the reaction conditions and does not interfere with the sensitive organolithium intermediate. This compatibility allows the reaction to proceed in a one-pot manner without the need for intermediate isolation, significantly simplifying the process flow. The final deprotection step involves acidic hydrolysis, which cleaves the amide bond to regenerate the free amino group. Crucially, the hydrolysis conditions are optimized to be mild enough to preserve the integrity of the trifluoroacetyl group while efficiently removing the protecting group. This mechanistic elegance ensures that the final product is obtained with exceptional purity, often exceeding 99%, thereby reducing the burden on downstream purification processes.
How to Synthesize Ortho-Amino Trifluoroacetophenone Efficiently
The practical implementation of this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize yield and safety. The process begins with the dissolution of the aniline substrate in an organic solvent such as diethyl ether, followed by the controlled addition of acyl chloride under basic conditions at low temperatures to prevent side reactions. Once the protected intermediate is secured, the system is cooled further for the lithiation step, where n-butyllithium is added slowly to maintain thermal stability. The subsequent addition of the trifluoroacetylating agent completes the carbon skeleton construction before a final acidic workup releases the target molecule. For a detailed breakdown of the specific operational parameters, reagent ratios, and safety protocols required for execution, please refer to the standardized guide below.
- Protect the amino group of the aniline derivative using acyl chloride and base to prevent side reactions during lithiation.
- Perform directed ortho-metalation using n-butyllithium and TMEDA, followed by reaction with a trifluoroacetyl compound.
- Hydrolyze the protected intermediate under acidic conditions to remove the protecting group and isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from nitration-based chemistry eliminates the need for specialized corrosion-resistant equipment and extensive waste treatment facilities associated with mixed acid handling. By utilizing common aniline derivatives that are often byproducts of other large-scale chemical processes, the method leverages existing market abundance to stabilize raw material costs. The high selectivity of the reaction means that less material is lost to byproducts, effectively increasing the mass efficiency of the production line. Furthermore, the ability to perform the acylation and quenching in a continuous sequence reduces the number of isolation steps, lowering labor costs and shortening the overall production cycle time. These factors combine to create a manufacturing process that is not only cost-effective but also highly responsive to fluctuating market demands.
- Cost Reduction in Manufacturing: The elimination of expensive separation processes and the high atom economy of the lithiation step contribute to substantial cost savings. By avoiding the formation of difficult-to-remove isomers, the need for resource-intensive chromatographic purification is minimized, allowing for simpler crystallization or distillation techniques. Additionally, the recovery and reuse of solvents like diethyl ether and ethyl acetate further drive down operational expenditures. The use of standard reagents rather than exotic catalysts ensures that the cost of goods sold remains competitive even at smaller batch sizes. This economic efficiency makes the technology viable for both pilot-scale development and full commercial production.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the starting materials are derived from widely produced industrial chemicals rather than niche intermediates. The robustness of the reaction conditions reduces the risk of batch failures due to minor variations in raw material quality or environmental factors. This reliability translates into more predictable lead times and the ability to maintain consistent inventory levels for downstream customers. Manufacturers can confidently commit to long-term supply agreements knowing that the production process is resilient to common supply chain disruptions. The scalability of the method ensures that volume requirements can be met without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with thermal hazards managed through controlled addition rates and efficient cooling systems. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing sites. Solvent recovery systems can be easily integrated to minimize volatile organic compound emissions, supporting sustainability goals. The mild nature of the hydrolysis step avoids the generation of heavy metal waste often associated with reduction catalysts. This environmentally friendly profile enhances the corporate social responsibility standing of the supply chain and facilitates easier permitting for new production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation. Understanding these nuances is vital for R&D teams evaluating the feasibility of adopting this route for their specific API projects. The answers provide clarity on yield expectations, safety protocols, and the versatility of the method for producing various derivatives.
Q: Why is the traditional nitration-reduction route unsuitable for industrial scale?
A: Traditional nitration of trifluoroacetophenones suffers from poor regioselectivity due to the strong electron-withdrawing nature of the carbonyl group, leading to difficult separations and lower yields compared to the directed lithiation approach.
Q: How does the amino protection strategy improve reaction yield?
A: Converting the free amine to an amide prevents unwanted side reactions with organolithium reagents and acts as a directing group, ensuring precise ortho-substitution and significantly enhancing overall process efficiency.
Q: What are the key safety considerations for the lithiation step?
A: The lithiation step requires strict temperature control below 5°C during reagent addition and the use of inert atmospheres to manage the reactivity of n-butyllithium and prevent thermal runaway.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Amino Trifluoroacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ortho-amino trifluoroacetophenone meets the highest industry standards. Our commitment to excellence extends beyond mere compliance; we actively collaborate with clients to optimize processes for cost and performance. By partnering with us, you gain access to a supply chain that is both robust and adaptable, capable of supporting your growth from clinical trials to full market launch.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific applications. Request a Customized Cost-Saving Analysis today to understand the potential economic impact on your project. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your requirements. Let us help you secure a competitive advantage through superior chemical manufacturing and reliable supply chain partnerships.
