Advanced Production of 3,5-Dibromo-o-Aminobenzaldehyde: Transforming Nitro Waste into High-Value Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking sustainable and cost-effective pathways to synthesize critical intermediates, particularly for high-volume drugs like Ambroxol Hydrochloride. Patent CN113444004A introduces a groundbreaking production process for 3,5-dibromo-o-aminobenzaldehyde, a pivotal precursor in the synthesis of this widely used mucolytic agent. Unlike conventional approaches that rely on expensive, fresh starting materials, this innovative methodology leverages waste valorization by utilizing o-nitro-dibromo-benzyl, a byproduct typically generated during the industrial production of o-nitrobenzaldehyde. By transforming this nitro waste into a high-value intermediate, the process addresses both economic inefficiencies and environmental concerns associated with chemical manufacturing. The core of this technology lies in a robust sequence involving iron-mediated reduction, precise acidic bromination, and a final conversion step that ensures high purity and yield. This approach not only mitigates the disposal costs of hazardous nitro waste but also establishes a more resilient supply chain for reliable pharmaceutical intermediate suppliers looking to optimize their production portfolios.
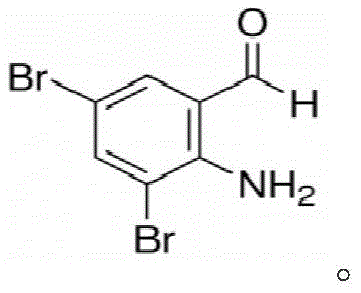
The strategic importance of 3,5-dibromo-o-aminobenzaldehyde cannot be overstated, given its role as the foundational building block for Ambroxol Hydrochloride, a drug approved globally for treating respiratory tract diseases. The molecular architecture, characterized by the specific positioning of bromine atoms relative to the amino and aldehyde groups, dictates the subsequent condensation and hydrogenation reactions required to form the final active pharmaceutical ingredient. As depicted in the structural analysis, the precise arrangement of functional groups is critical for downstream reactivity. Traditional synthesis routes often struggle with regioselectivity and the high cost of halogenated precursors. However, the method disclosed in this patent circumvents these issues by starting with a dibrominated nitro compound that is already structurally primed for the target molecule. This eliminates the need for difficult late-stage bromination steps that often lead to complex impurity profiles, thereby streamlining the overall manufacturing workflow and enhancing the feasibility of commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 3,5-dibromo-o-aminobenzaldehyde was plagued by significant technical and economic hurdles. One prominent historical method, described by von Danqi, utilized o-aminobenzaldehyde as the starting material, subjecting it to direct bromination. While conceptually straightforward, this route suffers from severe environmental drawbacks due to the generation of substantial quantities of iron sludge during the reduction phases, creating a heavy burden for waste treatment facilities. Furthermore, controlling the degree of bromination on the electron-rich aniline ring is notoriously difficult, often resulting in over-brominated byproducts that are challenging to separate. Another approach, documented by Zhang Weidong, employed methyl 2-aminobenzoate and specialized tribromo-reagents. Although this method achieved the target structure, it relied on expensive and less readily available raw materials, driving up the overall production cost. The multi-step nature of these legacy processes, involving harsh conditions and complex purification sequences, rendered them less attractive for modern, green chemistry-focused manufacturing environments.
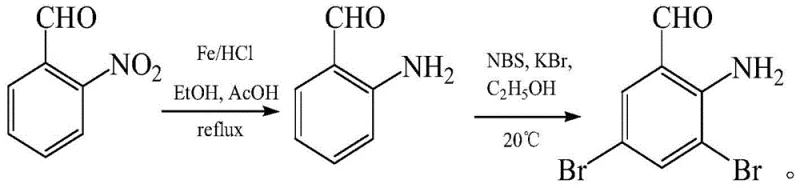
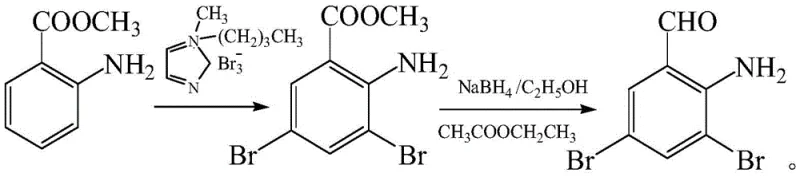
The Novel Approach
In stark contrast to these legacy techniques, the process outlined in Patent CN113444004A represents a paradigm shift towards circular economy principles in fine chemical synthesis. By identifying o-nitro-dibromo-benzyl—a common waste stream in o-nitrobenzaldehyde plants—as the primary feedstock, the inventors have effectively turned a liability into an asset. This novel approach bypasses the need for expensive fresh bromination reagents and simplifies the synthetic trajectory. The process begins with a clean reduction of the nitro group to an amine using active iron powder under controlled acidic conditions, followed by a highly selective bromination step that leverages the existing bromine atoms on the ring. This strategy not only reduces the consumption of virgin raw materials but also minimizes the formation of hazardous byproducts. The result is a streamlined workflow that offers cost reduction in pharmaceutical intermediates manufacturing while simultaneously adhering to stricter environmental regulations regarding waste discharge and resource utilization.
Mechanistic Insights into Iron-Mediated Reduction and Controlled Bromination
The heart of this synthesis lies in the precise orchestration of reduction and electrophilic substitution reactions. The initial step involves the reduction of o-nitro-dibromo-benzyl to o-amino-dibromo-benzyl using active iron powder and hydrochloric acid. This classic Bechamp reduction is optimized here by conducting the reaction at elevated temperatures between 70°C and 90°C, ensuring complete conversion of the nitro group while minimizing side reactions. Crucially, the reaction mixture is subsequently neutralized to a pH of 7-9 using sodium carbonate, which facilitates the separation of the organic amine phase from the aqueous iron sludge. This pH control is vital for preventing the formation of emulsions and ensuring high recovery rates of the amine intermediate. The use of extracting agents such as toluene or xylene further enhances the efficiency of this separation, allowing for the isolation of a high-purity o-amino-dibromo-benzyl solution ready for the subsequent functionalization steps.
Following the reduction, the process moves to a critical bromination and oxidation phase conducted under stringent low-temperature conditions (5-10°C). The o-amino-dibromo-benzyl sulfuric acid solution is treated with hydrobromic acid, followed by the dropwise addition of bromine and/or an oxidant like hydrogen peroxide. This low-temperature regime is essential for controlling the reactivity of the amino group and preventing poly-bromination or oxidative degradation of the sensitive aldehyde precursor. The patent specifies maintaining the monobromo impurity content at less than or equal to 0.2%, a testament to the high selectivity of this method. The mechanism likely involves the formation of a reactive bromonium species in situ, which selectively targets the remaining open positions on the aromatic ring. The subsequent addition of chloroform allows for the efficient extraction of the 3,5-dibromo-o-aminobenzyl dibromide intermediate, setting the stage for the final hydrolysis and cyclization steps that yield the target aldehyde with exceptional purity.
How to Synthesize 3,5-Dibromo-o-Aminobenzaldehyde Efficiently
The synthesis protocol detailed in the patent provides a robust framework for producing high-purity 3,5-dibromo-o-aminobenzaldehyde suitable for pharmaceutical applications. The procedure integrates waste management with chemical synthesis, requiring careful attention to temperature gradients and stoichiometric ratios during the bromination phase. Operators must ensure that the reduction step is driven to completion to avoid carrying over nitro impurities, which could interfere with downstream reactions. The extraction and acidification steps are equally critical, serving to purify the intermediate amine before it undergoes the final transformation. For a comprehensive understanding of the specific operational parameters, including exact molar ratios and stirring times, please refer to the standardized synthesis guide below which outlines the critical path for successful execution.
- Reduce o-nitro-dibromo-benzyl using active iron powder and hydrochloric acid at 70-90°C, followed by pH adjustment and extraction to isolate o-amino-dibromo-benzyl.
- Acidify the extract with sulfuric acid at low temperature (5-10°C) and perform selective bromination using bromine and/or oxidants while controlling monobromo impurities below 0.2%.
- React the dibromide intermediate with dimethylamine and sodium bicarbonate, then concentrate, decolorize, and crystallize to obtain the final aldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this waste-valorization technology offers compelling strategic advantages beyond simple technical feasibility. The most significant benefit is the drastic reduction in raw material costs achieved by substituting expensive, dedicated brominated precursors with low-cost nitro waste streams. This shift fundamentally alters the cost structure of the intermediate, providing a buffer against market volatility in halogen prices. Furthermore, by integrating this process, manufacturers can reduce their dependency on external suppliers of specialized reagents, thereby enhancing supply chain security. The ability to source feedstock from internal waste streams or local nitrobenzaldehyde producers creates a closed-loop system that insulates the production line from global logistics disruptions and raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like methyl 2-aminobenzoate or pure o-aminobenzaldehyde results in substantial cost savings. By utilizing o-nitro-dibromo-benzyl, which is often considered a disposal burden, the process converts a cost center (waste treatment) into a profit center (raw material). Additionally, the simplified purification steps reduce the consumption of solvents and energy, further driving down the operational expenditure per kilogram of finished product. This economic efficiency makes the final API more competitive in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing raw materials from established o-nitrobenzaldehyde production lines ensures a consistent and reliable supply of feedstock. Unlike niche reagents that may face availability issues, nitro waste is a ubiquitous byproduct in the fine chemical industry. This abundance guarantees long-term supply continuity, allowing manufacturers to plan production schedules with greater confidence. Moreover, the robustness of the iron reduction chemistry means that the process is less susceptible to minor fluctuations in reagent quality, ensuring stable output even when feedstock specifications vary slightly.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial constraints in mind. The avoidance of heavy metal catalysts and the minimization of iron sludge generation simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations. This 'green' profile not only reduces regulatory risk but also aligns with the sustainability goals of major pharmaceutical buyers who prioritize eco-friendly supply chains. The ease of scaling from pilot batches to multi-ton production runs facilitates rapid market entry and capacity expansion to meet growing global demand for Ambroxol.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,5-dibromo-o-aminobenzaldehyde using this patented waste-valorization method. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on impurity control, yield optimization, and regulatory compliance. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: What is the primary advantage of the synthesis method in Patent CN113444004A?
A: The primary advantage is the utilization of o-nitro-dibromo-benzyl, a waste byproduct from o-nitrobenzaldehyde production, as the starting material. This transforms a disposal liability into a valuable resource, significantly reducing raw material costs and environmental impact compared to traditional methods that require expensive fresh reagents.
Q: How does this process control impurities during bromination?
A: The process employs strict temperature control (5-10°C) during the acidification and bromination steps. Furthermore, it utilizes a controlled dropwise addition of bromine and/or oxidants (like hydrogen peroxide) in the presence of hydrobromic acid, specifically monitoring the reaction to ensure monobromo impurity levels remain below 0.2% before proceeding to the next stage.
Q: Is this method suitable for large-scale industrial production of Ambroxol intermediates?
A: Yes, the method is explicitly designed for industrial scalability. It avoids the generation of massive amounts of iron sludge associated with older reduction methods and simplifies the purification process through efficient extraction and crystallization steps, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dibromo-o-Aminobenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in Patent CN113444004A for the global Ambroxol supply chain. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this waste-valorization process are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 3,5-dibromo-o-aminobenzaldehyde meets the exacting standards required for pharmaceutical synthesis, delivering a product with minimal impurity profiles and consistent performance in downstream reactions.
We invite forward-thinking pharmaceutical companies and chemical distributors to collaborate with us to leverage this advanced technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to evaluate how this innovative, sustainable production method can enhance your supply chain resilience and drive down costs for your Ambroxol Hydrochloride programs.
