Industrial Scale Asymmetric Synthesis of D-(-)-Pantolactone for High-Purity Vitamin B5 Manufacturing
Industrial Scale Asymmetric Synthesis of D-(-)-Pantolactone for High-Purity Vitamin B5 Manufacturing
The global demand for Vitamin B5 (Calcium Pantothenate) continues to surge across pharmaceutical, nutritional, and feed additive sectors, driving an urgent need for more efficient and sustainable synthetic routes for its key chiral precursor, D-(-)-pantolactone. Patent CN109705065B introduces a groundbreaking asymmetric catalytic synthesis method that fundamentally shifts the production paradigm away from hazardous cyanide-based chemistry toward a safer, phase-transfer catalyzed approach. This technology leverages chiral quaternary ammonium salts derived from Cinchona alkaloids to facilitate a one-pot reaction between hydroxypivalaldehyde and chloroform under concentrated alkaline conditions. For R&D directors and procurement strategists, this innovation represents a critical opportunity to secure a reliable D-(-)-pantolactone supplier capable of delivering high-purity intermediates while mitigating the severe environmental and safety liabilities associated with legacy manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pantolactone has been plagued by significant safety and efficiency bottlenecks that compromise supply chain stability and operational expenditure. The most prevalent traditional route involves the Strecker reaction, which necessitates the use of hydrocyanic acid (HCN), a substance of extreme toxicity that requires rigorous containment protocols, specialized waste treatment facilities, and poses catastrophic risks in the event of leakage. Furthermore, alternative asymmetric hydrogenation methods relying on precious transition metal catalysts, such as rhodium or ruthenium complexes, introduce substantial raw material costs and create complex downstream purification challenges due to the difficulty of removing trace heavy metal residues to meet stringent pharmaceutical specifications. These conventional pathways often suffer from moderate enantioselectivity or require harsh reaction conditions that limit their scalability and economic viability for mass production.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN109705065B utilizes a chiral phase transfer catalysis (PTC) system that operates under remarkably mild and safe conditions. By employing readily available chloroform and concentrated alkali solutions instead of toxic cyanides, the process inherently enhances workplace safety and simplifies regulatory compliance. The reaction proceeds via a one-pot mechanism where the chiral quaternary ammonium salt facilitates the generation of a reactive dichlorocarbene intermediate, which then undergoes asymmetric addition to hydroxypivalaldehyde. This approach not only achieves impressive yields reaching up to 88.3% but also delivers exceptional enantiomeric excess values of up to 98.0% ee, effectively eliminating the need for costly resolution steps to separate unwanted isomers. 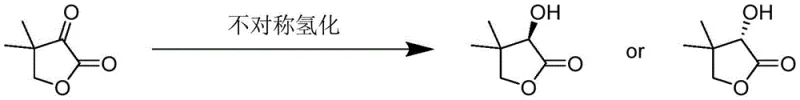
Mechanistic Insights into Chiral Phase Transfer Catalysis
The core of this technological breakthrough lies in the sophisticated interplay between the chiral catalyst and the reactive intermediates generated in the biphasic system. Under strong alkaline conditions, chloroform is deprotonated to form a trichloromethyl anion, which subsequently eliminates a chloride ion to generate the highly electrophilic dichlorocarbene species. The chiral quaternary ammonium salt acts as a molecular shuttle, transporting these anionic species from the aqueous phase into the organic phase where the hydroxypivalaldehyde substrate resides. Crucially, the bulky chiral environment surrounding the nitrogen center of the catalyst imposes strict stereochemical control during the carbene addition step, ensuring the formation of the desired D-configuration with high fidelity. This mechanistic precision allows for the direct construction of the chiral center without the need for subsequent enzymatic resolution or chemical separation, streamlining the entire synthetic sequence.
Structural optimization of the catalyst plays a pivotal role in maximizing both activity and selectivity. The patent details a series of Cinchona alkaloid-derived quaternary ammonium salts where variations in the substituents at the R1, R2, and R3 positions significantly influence the outcome. For instance, catalysts featuring specific methoxy or hydroxyl groups on the quinoline ring, combined with bulky aromatic groups like naphthyl or phenanthryl at the quaternary nitrogen, create a rigid chiral pocket that effectively discriminates between the prochiral faces of the aldehyde substrate. 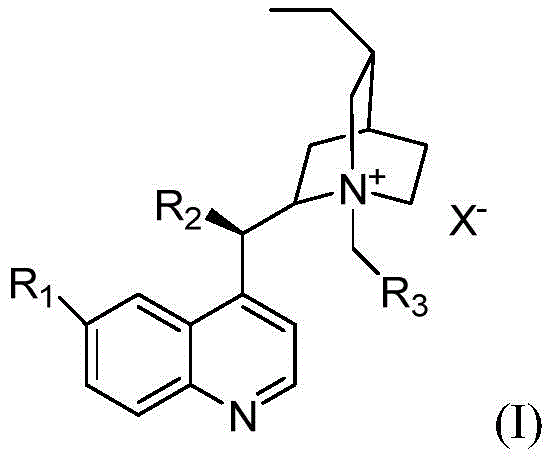
Furthermore, the impurity profile of the final product is inherently cleaner due to the absence of metal catalysts. In traditional hydrogenation routes, trace amounts of rhodium or ruthenium can persist through multiple purification stages, posing toxicity risks for final drug products. By utilizing an organocatalytic phase transfer system, this method completely bypasses the introduction of heavy metals, thereby simplifying the purification workflow and ensuring that the resulting high-purity D-(-)-pantolactone meets the most rigorous quality standards required for pharmaceutical and food-grade applications without the need for expensive metal scavenging resins.
How to Synthesize D-(-)-Pantolactone Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and robustness, making it highly attractive for process chemists aiming to transfer technology from the laboratory to the pilot plant. The procedure involves a straightforward one-pot protocol where hydroxypivalaldehyde, chloroform, and the selected chiral catalyst are charged into a reactor, followed by the controlled addition of concentrated alkali solution. The reaction temperature is maintained within a moderate range of 25°C to 60°C, avoiding the energy-intensive heating or cryogenic cooling often required by other asymmetric methods. Detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup procedures, are outlined in the comprehensive guide below to ensure reproducible high-yield results.
- Prepare the reaction mixture by combining hydroxypivalaldehyde, chloroform, and a specific chiral quaternary ammonium salt catalyst (1-20 mol%) in a reactor.
- Slowly add concentrated alkali solution (30-50% NaOH or KOH) while maintaining the temperature between 25°C and 60°C to generate the reactive dichlorocarbene intermediate.
- After reaction completion (2-30 hours), adjust pH to 0.8-1.2 with acid, extract with chloroform, and purify to obtain D-(-)-pantolactone with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this asymmetric phase transfer catalysis technology offers transformative benefits that extend far beyond simple chemical efficiency. The elimination of hydrocyanic acid from the supply chain removes a major logistical and regulatory burden, as sourcing, storing, and disposing of cyanide compounds involve exorbitant costs and strict governmental oversight. By switching to commodity reagents like chloroform and caustic soda, manufacturers can drastically simplify their raw material procurement strategy, reducing dependency on specialized hazardous chemical suppliers and mitigating the risk of supply disruptions caused by safety incidents or regulatory crackdowns on toxic substances.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of expensive noble metal catalysts with inexpensive organocatalysts and the removal of downstream metal removal units. Traditional routes often require dedicated filtration columns loaded with scavengers to reduce metal content to ppm levels, adding significant capital and operational expenditure. This new method inherently produces metal-free products, allowing for a streamlined isolation process that reduces solvent consumption, waste generation, and overall processing time, leading to substantial cost savings in the final manufacturing of Vitamin B5 intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply continuity. Because the process operates at ambient to moderate temperatures and utilizes stable reagents, it is less susceptible to the fluctuations and sensitivities that often plague sensitive organometallic reactions. This stability ensures consistent batch-to-batch quality and high throughput, enabling suppliers to maintain reliable inventory levels and meet tight delivery schedules for global pharmaceutical clients. The simplified safety profile also means that production facilities face fewer shutdowns for safety audits or maintenance related to hazardous material handling.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with modern green chemistry principles. The avoidance of cyanide waste streams significantly lowers the cost and complexity of wastewater treatment, a critical factor for chemical plants operating under increasingly strict environmental regulations. The high atom economy and reduced waste generation facilitate easier scale-up from kilogram to multi-ton scales, ensuring that the commercial scale-up of complex pharmaceutical intermediates can proceed without encountering the bottlenecks typically associated with hazardous waste disposal and permitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards and what specific advantages it offers for large-scale production.
Q: How does this method improve safety compared to traditional Strecker synthesis?
A: Traditional methods often utilize highly toxic hydrocyanic acid (HCN) for the Strecker reaction. This patented process replaces HCN with chloroform and concentrated alkali, significantly reducing occupational health risks and eliminating the need for specialized cyanide handling infrastructure.
Q: What represents the primary cost advantage of this phase transfer catalysis route?
A: The process avoids expensive transition metal catalysts like rhodium or ruthenium and eliminates costly downstream heavy metal removal steps. Additionally, the use of commodity chemicals like chloroform and caustic soda drives down raw material costs substantially.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights suitability for industrial production. The one-pot nature of the reaction, mild temperature requirements (25-60°C), and high yields (up to 88.3%) with excellent enantioselectivity (up to 98.0% ee) make it robust for scaling from pilot to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-(-)-Pantolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is paramount for the future of the fine chemical industry. Our team of expert process chemists has extensively evaluated the technology described in patent CN109705065B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this advanced phase transfer catalysis method in our state-of-the-art facilities, ensuring that every batch of D-(-)-pantolactone we deliver adheres to stringent purity specifications and is validated by our rigorous QC labs to guarantee consistency and quality for your downstream Vitamin B5 synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can optimize your production costs while securing a sustainable and compliant source of this critical pharmaceutical intermediate.
