Advanced One-Pot Synthesis of Nucleoside Dimer Phosphoramidites for Oligonucleotide Drugs
The rapid expansion of the oligonucleotide therapeutic market has placed unprecedented demand on the supply chain for high-quality building blocks, specifically nucleoside dimer phosphoramidites. Patent CN114230624A discloses a groundbreaking synthetic methodology that addresses critical bottlenecks in the production of these essential intermediates. This technology introduces a streamlined one-pot strategy that integrates coupling, sulfuration, and desilylation reactions, fundamentally altering the economic and operational landscape of oligonucleotide precursor manufacturing. By utilizing modified nucleoside monomers and phosphoramidites as raw materials, the process achieves superior purity profiles while drastically simplifying the operational workflow. For R&D directors and procurement specialists seeking a reliable nucleoside dimer phosphoramidite supplier, this innovation represents a significant leap forward in process chemistry.

The implementation of this novel route allows for the efficient construction of phosphorothioate linkages, which are vital for the stability and efficacy of antisense and siRNA drugs. Unlike traditional methods that often suffer from low atom economy and complex workup procedures, this approach leverages mild reaction conditions and highly selective reagents. The ability to synthesize stable intermediates like compound M-V with high selectivity ensures that downstream solid-phase synthesis processes, such as those performed on controlled pore glass (CPG) supports, proceed with minimal failure sequences. This technical advancement is not merely a laboratory curiosity but a robust industrial solution designed to meet the rigorous quality standards of the pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nucleoside dimer phosphoramidites has been plagued by inefficiencies inherent in multi-step batch processes. Conventional routes typically require the isolation and purification of intermediates after each chemical transformation, leading to substantial material loss and increased solvent consumption. A major pain point in traditional phosphonylation strategies is the reliance on excessive amounts of phosphorus-chlorine (P-Cl) reagents, often requiring 12.5 to 16 equivalents to drive the reaction to completion. This overuse not only inflates raw material costs but also generates significant volumes of phosphorus-containing waste, complicating environmental compliance and waste treatment protocols. Furthermore, the harsh conditions often associated with these older methods can lead to degradation of sensitive protecting groups, resulting in complex impurity profiles that are difficult to separate.
The Novel Approach
The methodology described in the patent data offers a transformative alternative by employing a one-pot coupling, sulfuration, and deprotection sequence. This approach eliminates the need for intermediate isolation, thereby preserving yield and reducing the overall processing time. Crucially, the new method switches from expensive and wasteful P-Cl reagents to more economical P-Amidite reagents, reducing the stoichiometric requirement to just 3 equivalents. This shift alone represents a massive reduction in reagent costs and waste generation. The process operates under mild temperatures, typically between 20-25°C, which enhances safety and reduces energy consumption. By integrating these steps, the novel approach achieves yields improving from approximately 69.5 mol% in conventional methods to over 75.19 mol%, demonstrating clear superiority in both efficiency and output quality for cost reduction in oligonucleotide manufacturing.
Mechanistic Insights into One-Pot Coupling and Sulfuration
The core of this synthetic breakthrough lies in the precise orchestration of three distinct chemical transformations within a single reaction vessel. The process initiates with the activation of the phosphoramidite moiety using agents such as tetrazole, 4,5-dicyanoimidazole, or 5-ethylthiotetrazole. This activation facilitates the nucleophilic attack of the 5'-hydroxyl group of the incoming nucleoside on the phosphorus center, forming the critical P-O bond of the dimer intermediate M-III. The choice of activator is critical for minimizing side reactions and ensuring high coupling efficiency. Following the coupling, the trivalent phosphite triester is immediately subjected to sulfuration using specialized reagents like 3-amino-1,2,4-dithia-5-azolethione. This step converts the unstable P(III) species into a stable P(V) phosphorothioate linkage, which is essential for the biological activity of the final oligonucleotide drug.
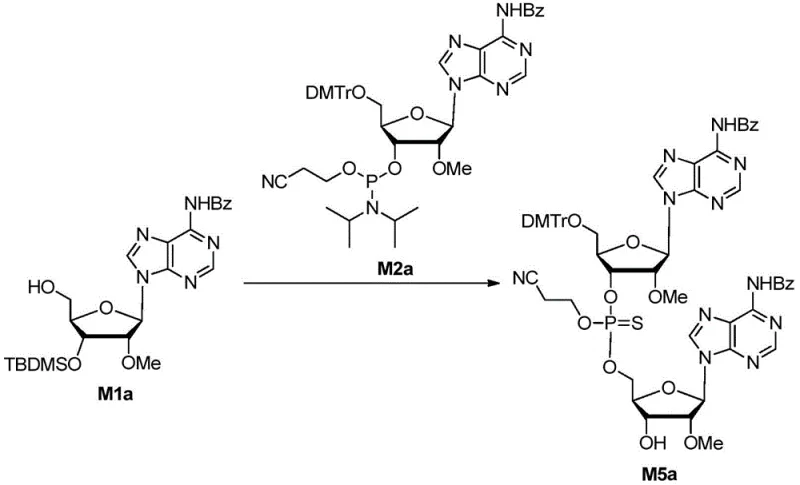
Following sulfuration, the protocol employs a desilylation step using fluoride reagents such as tetrabutylammonium fluoride (TBAF) or pyridinium hydrogen fluoride. This step selectively removes the tert-butyldimethylsilyl (TBDMS) protecting group from the 3'-position without disturbing other acid-labile protecting groups like the dimethoxytrityl (DMTr) group. The mechanistic elegance of this sequence ensures that the final intermediate M-V possesses a free 3'-hydroxyl group ready for the final phosphonylation. Impurity control is maintained through careful monitoring via Ultra Performance Liquid Chromatography (UPLC), ensuring that the conversion is complete before proceeding to the next stage. This rigorous control over the reaction pathway prevents the accumulation of deletion sequences or incomplete sulfurization byproducts, which are common failure modes in oligonucleotide synthesis.
How to Synthesize Nucleoside Dimer Phosphoramidite Efficiently
The synthesis of these complex molecules requires strict adherence to the optimized one-pot protocol to ensure maximum yield and purity. The process begins with the dissolution of the silylated nucleoside and the phosphoramidite monomer in a suitable solvent system such as acetonitrile or dichloromethane. Detailed standardized operating procedures regarding reagent addition rates, temperature control, and quenching protocols are essential for reproducibility. The following guide outlines the critical phases of this advanced manufacturing process, highlighting the specific conditions required to achieve the high purity levels demanded by regulatory bodies.
- Mix protected nucleoside monomer M-I with phosphoramidite M-II in the presence of an activator like tetrazole to form the coupled intermediate M-III.
- Perform in-situ sulfuration of the trivalent phosphine in M-III using a sulfurizing agent to generate the phosphorothioate linkage in intermediate M-IV.
- Execute desilylation using a fluoride reagent to remove the protecting group, followed by final phosphonylation to yield the target dimer phosphoramidite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits that extend beyond simple yield improvements. The reduction in process steps directly translates to a shorter manufacturing cycle time, allowing for faster response to market demands and reduced inventory holding costs. By eliminating the need for multiple chromatographic purifications between steps, the process significantly lowers the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical production. This streamlined workflow enhances the overall reliability of the supply chain by reducing the number of potential failure points where batch losses could occur.
- Cost Reduction in Manufacturing: The shift from using 12.5 to 16 equivalents of P-Cl reagents to merely 3 equivalents of P-Amidite reagents results in a drastic reduction in raw material expenditure. Additionally, the elimination of intermediate isolation steps reduces labor costs and utility consumption associated with drying and concentrating multiple fractions. The qualitative impact on the bottom line is substantial, as the process minimizes waste disposal fees associated with phosphorus-containing byproducts, aligning with green chemistry principles.
- Enhanced Supply Chain Reliability: The starting materials, such as compounds M-I and M-II, are conventional nucleoside monomers that are readily available from established suppliers. This availability mitigates the risk of raw material shortages that can plague the production of exotic intermediates. The robustness of the one-pot method means that production schedules are less susceptible to delays caused by complex purification bottlenecks, ensuring a steady flow of high-purity nucleoside dimer phosphoramidites to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions (10-80°C, preferably 20-25°C) make this process inherently safer and easier to scale from kilogram to tonne quantities. The reduced solvent usage and lower waste generation simplify the environmental permitting process and reduce the burden on wastewater treatment facilities. This scalability ensures that the supply can grow in tandem with the increasing global demand for oligonucleotide therapies without compromising on quality or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these nucleoside dimer phosphoramidites. The answers are derived directly from the patented methodology and reflect the current state-of-the-art in oligonucleotide intermediate synthesis. Understanding these details is crucial for partners looking to integrate these building blocks into their own drug development pipelines.
Q: What are the key advantages of the one-pot synthesis method for nucleoside dimers?
A: The one-pot method significantly simplifies the process by combining coupling, sulfuration, and deprotection steps without isolating intermediates. This reduces solvent usage, minimizes waste generation, and improves overall yield compared to traditional multi-step isolation processes.
Q: How does this method improve cost efficiency in oligonucleotide manufacturing?
A: Cost efficiency is achieved by reducing the equivalents of phosphorus reagents required (from 12.5-16 equivalents of P-Cl reagents down to 3 equivalents of P-Amidite reagents) and eliminating costly chromatographic purification steps between intermediate stages.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the method utilizes mild reaction conditions (typically 20-25°C) and readily available starting materials. The simplified workflow and reduced purification requirements make it highly suitable for scaling up to commercial quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nucleoside Dimer Phosphoramidite Supplier
At NINGBO INNO PHARMCHEM, we recognize that the success of next-generation genetic medicines depends on the quality and availability of their foundational components. Our technical team has extensively evaluated the synthetic pathways described in CN114230624A and possesses the expertise to implement these advanced one-pot methodologies at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and impurity profiles of every batch using advanced UPLC and MS techniques.
We invite you to collaborate with us to leverage these cost-effective and high-yield synthetic routes for your oligonucleotide programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of chemical excellence and reliability.
