Advanced Manufacturing of Selamectin: A Cost-Effective IBX Oxidation Route for Veterinary APIs
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with environmental sustainability, particularly for high-volume veterinary active pharmaceutical ingredients (APIs). Patent CN112830945A introduces a transformative preparation method for macrolide compounds, specifically targeting the synthesis of Selamectin, a critical antiparasitic agent. This innovation departs from traditional oxidative protocols by employing 2-iodoxybenzoic acid (IBX) as a superior oxidant, effectively resolving longstanding issues related to filtration bottlenecks and wastewater management. For R&D directors and process chemists, this patent represents a significant leap forward in optimizing the oxidation step from compound T00 to T01, ensuring that the subsequent reduction and oximation steps proceed with minimal impurity carryover. The strategic integration of this novel oxidation protocol not only enhances the overall yield but also simplifies the downstream processing requirements, making it an attractive candidate for modern, green chemistry manufacturing standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Selamectin and related macrolides has relied heavily on manganese dioxide (MnO2) or other heterogeneous oxidants for the critical oxidation step. While chemically effective, these traditional reagents introduce severe operational inefficiencies at an industrial scale. The primary drawback lies in the physical properties of the spent oxidant; manganese dioxide residues are notoriously difficult to filter, often clogging filter media and extending process times significantly. This leads to increased labor costs and equipment downtime. Furthermore, the fine particulate nature of MnO2 frequently results in leakage into the filtrate, necessitating complex purification steps to remove metal contaminants, which ultimately increases the volume of hazardous wastewater requiring treatment. These factors collectively inflate the cost of goods sold (COGS) and complicate regulatory compliance regarding heavy metal residuals in the final API.
The Novel Approach
In stark contrast, the methodology disclosed in CN112830945A utilizes 2-iodoxybenzoic acid (IBX) to drive the oxidation of the starting material T00 to the intermediate T01. This switch fundamentally alters the physical dynamics of the reaction work-up. IBX and its reduced byproducts exhibit solubility characteristics that allow for rapid and clean filtration, eliminating the chronic clogging issues associated with manganese dioxide. Crucially, the insoluble substances recovered after filtration are primarily unreacted or reduced IBX species, which can be easily re-oxidized and recycled back into the process. This closed-loop capability drastically reduces raw material consumption and minimizes the environmental footprint of the manufacturing process. By streamlining the isolation of intermediate T01, the novel approach sets a cleaner foundation for the subsequent hydrogenation and oximation steps, ensuring a higher quality crude product entering the final purification stages.
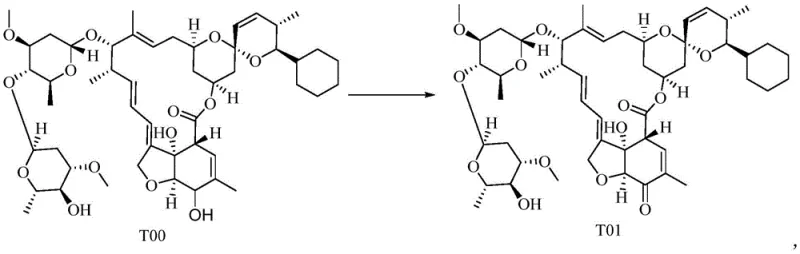
Mechanistic Insights into IBX-Catalyzed Oxidation and Subsequent Reduction
The core of this synthetic advancement lies in the selective oxidation mechanism facilitated by IBX in a polar aprotic solvent system, typically acetone. Unlike radical-based oxidations that can lead to non-selective degradation of the sensitive macrolide backbone, IBX operates through a well-defined ligand exchange mechanism that targets specific hydroxyl groups with high chemoselectivity. This precision is vital for preserving the integrity of the complex macrocyclic ring structure found in T00. The reaction conditions are mild, typically maintained between 20°C and 35°C, which further suppresses the formation of thermal degradation byproducts. For the R&D team, understanding this mechanism is key to controlling the impurity profile; the high conversion rate (>99% residual T00 consumption) ensures that the downstream hydrogenation catalyst is not poisoned by unreacted starting materials, thereby maintaining high catalytic efficiency throughout the multi-step sequence.
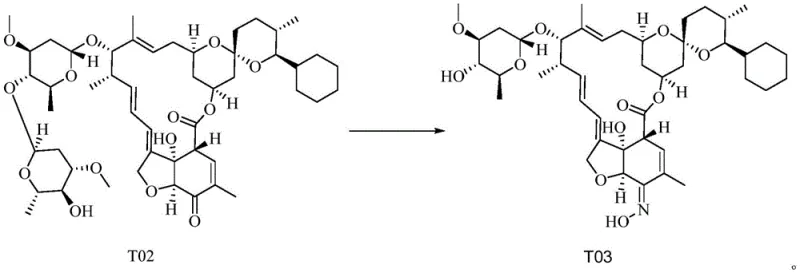
Following the oxidation, the process flows seamlessly into a catalytic hydrogenation step to convert T01 to T02, utilizing a rhodium-based catalyst under moderate hydrogen pressure. This reduction is critical for establishing the correct stereochemistry and saturation required for the biological activity of the final product. The subsequent oximation and desugarization reaction transforms T02 into the target Selamectin (T03) through a nucleophilic attack by hydroxylamine, accompanied by the cleavage of the sugar moiety. The synergy between these steps is optimized by the high purity of the incoming intermediates. By avoiding the accumulation of metal impurities from the oxidation step, the rhodium catalyst in the reduction phase maintains its activity for longer cycles, and the final crystallization of T03 yields a product with exceptional purity, often exceeding 98% after a single recrystallization sequence.
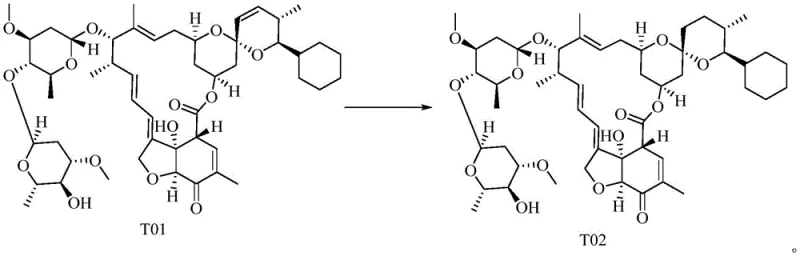
How to Synthesize Selamectin Efficiently
The synthesis of Selamectin via this patented route is designed for operational simplicity and robustness, making it highly suitable for technology transfer from laboratory to pilot plant. The process begins with the dissolution of the macrolide precursor T00 in acetone, followed by the controlled addition of IBX. Reaction progress is monitored via HPLC to ensure complete consumption of the starting material before filtration. The filtrate containing T01 is then subjected directly to hydrogenation without extensive purification, leveraging the cleanliness of the IBX oxidation. Finally, the crude T02 undergoes oximation in an isopropanol-water system, followed by a targeted crystallization protocol using toluene and methanol to isolate the final API. Detailed standardized operating procedures for each unit operation, including specific temperature ramps and agitation rates, are essential for maximizing yield and consistency.
- Oxidize compound T00 using 2-iodoxybenzoic acid (IBX) in acetone at 20-35°C to obtain intermediate T01, followed by filtration to remove insoluble residues.
- Subject compound T01 to catalytic hydrogenation using tris(triphenylphosphine)rhodium chloride under 1.0-3.0 MPa hydrogen pressure in acetone to generate compound T02.
- React compound T02 with hydroxylamine hydrochloride in isopropanol/water mixture at 25-50°C for oximation and desugarization, followed by crystallization to isolate pure Selamectin (T03).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this IBX-based synthesis route offers tangible economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage is the drastic simplification of the waste management infrastructure. By eliminating the need for handling large volumes of manganese-containing sludge, facilities can reduce their environmental compliance costs and avoid the regulatory hurdles associated with heavy metal disposal. Additionally, the ability to recycle the oxidant creates a more stable cost structure, insulating the manufacturing process from volatility in raw material pricing. This stability is crucial for long-term supply contracts where fixed pricing models are often required by major veterinary pharmaceutical clients.
- Cost Reduction in Manufacturing: The implementation of this process leads to substantial cost savings primarily through the reduction of processing time and the elimination of expensive purification steps. Since the IBX oxidation allows for direct filtration and potential recycling of the oxidant, the consumption of auxiliary chemicals is significantly lowered. Furthermore, the high purity of the intermediates reduces the load on the final crystallization step, meaning less solvent is required for recrystallization and less product is lost to mother liquors. This cumulative effect results in a lower cost per kilogram of the final API, enhancing the competitiveness of the supplier in the global market.
- Enhanced Supply Chain Reliability: Operational reliability is markedly improved due to the robustness of the filtration and work-up procedures. Traditional methods prone to filter clogging often result in unpredictable batch cycle times, leading to delays in delivery schedules. The streamlined workflow described in this patent ensures consistent batch turnover rates, allowing manufacturers to meet tight delivery windows with greater confidence. Moreover, the use of common, commercially available solvents like acetone and isopropanol reduces the risk of supply chain disruptions associated with specialized or hazardous reagents, ensuring continuous production capabilities even during market fluctuations.
- Scalability and Environmental Compliance: From a scale-up perspective, this route is exceptionally favorable because it mitigates the engineering challenges associated with solid-liquid separations on a large scale. The ease of filtration translates directly to smaller reactor occupancy times and higher throughput in existing manufacturing assets. Environmentally, the process aligns with modern green chemistry principles by reducing the E-factor (mass of waste per mass of product). The reduction in wastewater toxicity and volume simplifies the permitting process for new manufacturing lines and supports corporate sustainability goals, which are increasingly becoming a prerequisite for partnerships with top-tier multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Selamectin synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is critical for stakeholders evaluating the feasibility of adopting this new standard for veterinary API production.
Q: Why is IBX preferred over Manganese Dioxide for Selamectin oxidation?
A: IBX (2-iodoxybenzoic acid) offers superior process control compared to manganese dioxide. While MnO2 often leads to difficult filtration, prolonged processing times, and significant wastewater generation due to fine particulate leakage, IBX allows for rapid filtration of insoluble residues. Furthermore, the filtered IBX residue can be re-oxidized and reused, significantly lowering raw material costs and environmental impact.
Q: What are the purity specifications achievable with this new route?
A: The patented method emphasizes high purity and low impurity content through optimized reaction conditions and simplified work-up procedures. By avoiding multi-step purification treatments required in conventional methods and utilizing efficient crystallization techniques (using toluene and methanol), the process consistently yields Selamectin with purity levels exceeding 98%, minimizing the need for repeated recrystallization.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial implementation. It addresses key scalability issues such as filtration difficulty and wastewater treatment. The use of common solvents like acetone and isopropanol, combined with the ability to recycle the oxidant and perform telescoped reactions without isolating intermediates, makes it highly adaptable for commercial scale-up from pilot plants to multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selamectin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the IBX oxidation strategy are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Selamectin meets the highest international standards, providing our clients with the assurance needed for regulatory filings and market entry. We are committed to delivering high-purity veterinary intermediates that drive efficiency in your supply chain.
We invite you to collaborate with us to leverage this innovative technology for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your margins. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this critical veterinary active ingredient.
