Optimizing Efavirenz Production: A Technical Breakthrough in Chiral Intermediate Synthesis
Optimizing Efavirenz Production: A Technical Breakthrough in Chiral Intermediate Synthesis
The pharmaceutical industry continuously seeks robust, cost-effective pathways for the manufacture of critical antiretroviral agents, and the synthesis of Efavirenz remains a focal point of process chemistry innovation. Patent CN111548317A, published in August 2020, introduces a transformative methodology for producing Efavirenz and its key chiral intermediates, specifically addressing the economic and safety bottlenecks associated with traditional organozinc chemistry. This technical disclosure outlines a novel route that substitutes expensive and hazardous dialkyl zinc reagents with anhydrous zinc chloride, achieving a remarkable improvement in single-step yield from 86% to 93%. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and economically viable manufacturing of non-nucleoside reverse transcriptase inhibitors (NNRTIs). The following analysis dissects the chemical ingenuity of this patent and evaluates its profound implications for procurement strategy and commercial scalability in the competitive API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of the chiral alcohol intermediate required for Efavirenz has relied heavily on the addition of cyclopropylacetylide to a trifluoromethyl ketone in the presence of chiral amino alcohols and dialkyl zinc species. While effective, this conventional approach suffers from severe economic and operational drawbacks that hinder optimal production efficiency. The reliance on diethyl zinc or dimethyl zinc introduces substantial raw material costs, as these organometallic reagents are significantly more expensive than simple inorganic zinc salts. Furthermore, dialkyl zinc compounds are notoriously pyrophoric and moisture-sensitive, necessitating rigorous exclusion of air and water, specialized handling equipment, and complex safety protocols that inflate operational expenditures. These factors collectively create a fragile supply chain vulnerable to raw material price volatility and logistical delays, making the traditional route less attractive for high-volume commercial manufacturing where margin compression is a constant pressure.
The Novel Approach
The methodology disclosed in patent CN111548317A elegantly circumvents these limitations by engineering a catalytic system based on anhydrous zinc chloride rather than dialkyl zinc. As illustrated in the reaction scheme below, the process generates the active chiral zinc species in situ through the reaction of zinc chloride with sodium alkoxides derived from metallic sodium and trifluoroethanol. 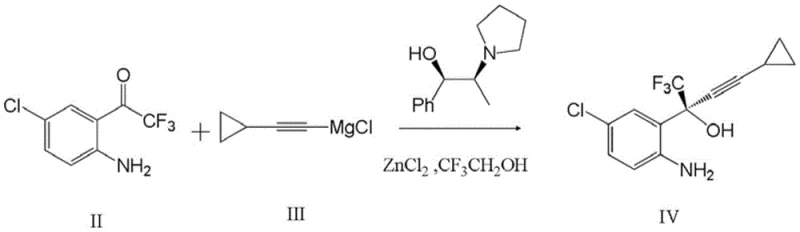 This strategic substitution not only drastically lowers the cost of goods sold (COGS) by utilizing commodity-grade zinc salts but also enhances the safety profile of the reaction by eliminating the need to handle bulk quantities of pyrophoric liquids. The patent data explicitly confirms that this modification does not compromise stereocontrol; on the contrary, it boosts the isolated yield of the critical intermediate (2S)-2-(2-amino-5-chlorophenyl)-4-cyclopropyl-1,1,1-trifluoromethyl-3-butyn-2-ol to 93%, demonstrating superior atom economy and process efficiency compared to prior art methods.
This strategic substitution not only drastically lowers the cost of goods sold (COGS) by utilizing commodity-grade zinc salts but also enhances the safety profile of the reaction by eliminating the need to handle bulk quantities of pyrophoric liquids. The patent data explicitly confirms that this modification does not compromise stereocontrol; on the contrary, it boosts the isolated yield of the critical intermediate (2S)-2-(2-amino-5-chlorophenyl)-4-cyclopropyl-1,1,1-trifluoromethyl-3-butyn-2-ol to 93%, demonstrating superior atom economy and process efficiency compared to prior art methods.
Mechanistic Insights into Zinc-Mediated Asymmetric Addition
The core of this technological advancement lies in the precise generation of a chiral zinc alkoxide complex that directs the stereoselective addition of the acetylide nucleophile. In this mechanism, metallic sodium reacts with trifluoroethanol to generate sodium trifluoroethoxide, which subsequently interacts with anhydrous zinc chloride and the chiral ligand (1R,2S)-1-phenyl-2-(pyrrolidinyl)-1-propanol. This sequence forms a highly organized chiral environment around the zinc center, which is crucial for inducing the desired (S)-configuration at the quaternary carbon center. The cyclopropyl ethynyl magnesium chloride, prepared separately from cyclopropylacetylene and n-butylmagnesium chloride, acts as the transmetallation partner, transferring the alkynyl group to the chiral zinc species before attacking the electrophilic ketone substrate. This intricate interplay ensures that the steric bulk of the ligand effectively shields one face of the carbonyl group, thereby enforcing high enantioselectivity without the need for stoichiometric amounts of expensive chiral auxiliaries.
Following the formation of the chiral alcohol intermediate, the synthesis proceeds to the final cyclization step to form the benzoxazinone ring characteristic of Efavirenz. 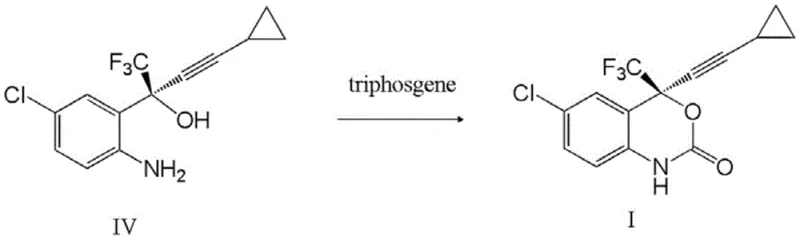 This transformation involves the reaction of the amino-alcohol intermediate with triphosgene in the presence of a base such as potassium bicarbonate. The mechanism entails the formation of a chloroformate intermediate which subsequently undergoes intramolecular nucleophilic attack by the adjacent amine group, closing the ring to yield the target heterocycle. The patent emphasizes the use of methyl tert-butyl ether (MTBE) and isopropyl ether as solvents during this stage, which facilitates easy phase separation and product isolation. The rigorous control of reaction conditions, including temperature management during the exothermic addition of triphosgene, ensures minimal formation of side products, resulting in a crude product that requires only simple recrystallization to achieve pharmaceutical-grade purity.
This transformation involves the reaction of the amino-alcohol intermediate with triphosgene in the presence of a base such as potassium bicarbonate. The mechanism entails the formation of a chloroformate intermediate which subsequently undergoes intramolecular nucleophilic attack by the adjacent amine group, closing the ring to yield the target heterocycle. The patent emphasizes the use of methyl tert-butyl ether (MTBE) and isopropyl ether as solvents during this stage, which facilitates easy phase separation and product isolation. The rigorous control of reaction conditions, including temperature management during the exothermic addition of triphosgene, ensures minimal formation of side products, resulting in a crude product that requires only simple recrystallization to achieve pharmaceutical-grade purity.
How to Synthesize Efavirenz Efficiently
The synthesis protocol detailed in the patent offers a clear, step-by-step roadmap for replicating this high-yield process in a GMP-compliant environment. The procedure begins with the low-temperature preparation of the Grignard reagent, followed by the careful assembly of the chiral zinc catalyst system under inert atmosphere. Critical parameters such as the dropwise addition rates and internal temperature controls (maintained below 10°C during exothermic steps) are essential for maximizing yield and safety. The subsequent workup involves aqueous washes with citric acid and brine to remove metal salts, followed by concentration and recrystallization from n-hexane and MTBE mixtures. For a comprehensive understanding of the specific operational parameters, reagent ratios, and safety precautions required for execution, please refer to the standardized synthesis guide provided below.
- Preparation of cyclopropyl ethynyl magnesium chloride by reacting cyclopropylacetylene with n-butylmagnesium chloride at low temperature.
- Formation of the chiral zinc complex using anhydrous zinc chloride, sodium metal, trifluoroethanol, and a chiral amino alcohol ligand.
- Addition of the ketone substrate to the chiral zinc complex followed by cyclization with triphosgene to yield Efavirenz.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc chloride-based synthesis route presents a compelling value proposition centered on cost stability and operational resilience. By shifting away from volatile organozinc reagents, manufacturers can decouple their production costs from the fluctuating prices of specialty organometallics, leading to a more predictable financial model. The use of anhydrous zinc chloride, a widely available and inexpensive commodity chemical, significantly reduces the direct material costs associated with the most critical step of the synthesis. Furthermore, the elimination of pyrophoric reagents simplifies warehouse storage requirements and reduces the need for specialized fire suppression systems, indirectly lowering facility overheads. These cumulative savings enhance the overall competitiveness of the final API in the global market, allowing for more aggressive pricing strategies without sacrificing margin.
- Cost Reduction in Manufacturing: The substitution of diethyl zinc with anhydrous zinc chloride represents a fundamental shift in cost structure, removing one of the most expensive line items from the bill of materials. Since zinc chloride is produced on a massive industrial scale for various applications, its price point is orders of magnitude lower than that of custom-synthesized dialkyl zinc compounds. Additionally, the improvement in yield from 86% to 93% means that less raw material is wasted per kilogram of final product, effectively amplifying the cost savings through improved material efficiency. This dual benefit of cheaper inputs and higher output creates a substantial buffer against market inflation, ensuring long-term economic viability for the manufacturing process.
- Enhanced Supply Chain Reliability: Reliance on niche reagents like diethyl zinc often exposes supply chains to single-source risks and logistical bottlenecks, whereas zinc chloride and metallic sodium are standard inventory items for most chemical suppliers. This ubiquity ensures that production schedules are less likely to be disrupted by raw material shortages or shipping delays. Moreover, the stability of zinc chloride allows for longer shelf life and easier transportation compared to moisture-sensitive organozinc solutions, reducing the complexity of inbound logistics. For supply chain heads, this translates to a more robust and flexible procurement strategy that can easily scale up to meet surging demand for antiretroviral therapies without the fear of critical reagent stockouts.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with modern green chemistry principles by reducing the hazard profile of the synthesis. The absence of pyrophoric liquids minimizes the risk of catastrophic fire incidents, lowering insurance premiums and regulatory compliance burdens. The waste stream generated is primarily inorganic zinc salts, which are easier to treat and dispose of compared to complex organometallic residues. This simplified waste management profile facilitates smoother environmental permitting for scale-up activities, enabling faster transition from pilot plant to commercial production scales. Consequently, manufacturers can accelerate time-to-market for generic versions of Efavirenz while maintaining a strong corporate social responsibility posture regarding worker safety and environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN111548317A, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement officers negotiating supply contracts. The answers reflect the specific advantages of the zinc chloride methodology over traditional approaches, highlighting its suitability for large-scale pharmaceutical manufacturing.
Q: What is the primary advantage of using Zinc Chloride over Diethyl Zinc in Efavirenz synthesis?
A: The primary advantage is significant cost reduction and improved safety. Anhydrous zinc chloride is substantially cheaper and less pyrophoric than diethyl zinc, while the patent data indicates an yield increase from 86% to 93%.
Q: How does this method impact the purity of the final Efavirenz API?
A: The method utilizes a highly stereoselective chiral ligand system ((1R,2S)-1-phenyl-2-(pyrrolidinyl)-1-propanol) which ensures high enantiomeric excess. The subsequent recrystallization steps described in the patent further enhance purity to meet stringent pharmaceutical standards.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route is designed for scalability. By replacing sensitive organozinc reagents with stable zinc salts and utilizing common solvents like THF and MTBE, the process mitigates safety risks associated with large-scale exothermic reactions, facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Efavirenz Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111548317A are fully realized in practical application. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Efavirenz intermediate meets the highest global pharmacopeial standards. Our commitment to quality assurance ensures that the improved yields and cost efficiencies promised by this new chemistry are delivered consistently, providing our clients with a reliable source of high-purity pharmaceutical intermediates.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-optimized synthesis technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this superior manufacturing process into your portfolio. Together, we can drive down the cost of essential medicines while maintaining the uncompromising quality required for patient safety.
