Advanced Synthesis of 3,5-Dibromo-4-Fluoropyridine for Commercial Scale-Up of Complex Heterocyclic Compounds
Introduction to Patent CN102898360A and Strategic Value
The chemical landscape for halogenated pyridines has long been dominated by complex multi-step syntheses that often suffer from poor regioselectivity and prohibitive costs. Patent CN102898360A, published in early 2013, introduces a transformative methodology for the production of 3,5-dibromo-4-fluoropyridine, a critical building block extensively utilized in the development of advanced agrochemicals and pharmaceutical agents. This intellectual property outlines a robust two-step sequence starting from the inexpensive and commercially abundant 4-aminopyridine, effectively bypassing the historical bottlenecks associated with introducing fluorine atoms into highly substituted pyridine rings. For R&D Directors and Procurement Managers alike, this patent represents a pivotal shift towards more economical and reliable sourcing strategies for high-value heterocyclic intermediates. By leveraging a radical bromination followed by a diazotization-fluorination sequence, the technology ensures consistent quality while significantly simplifying the operational complexity typically required for such transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of this specific invention, the synthesis of 3,5-dibromo-4-fluoropyridine was notably absent from standard literature, indicating a significant gap in accessible chemical technology for this specific substitution pattern. Traditional approaches to synthesizing poly-halogenated pyridines often rely on direct halogenation of pyridine or its N-oxide derivatives, which frequently result in mixtures of isomers that are notoriously difficult and expensive to separate. Furthermore, existing methods for introducing fluorine at the 4-position often require harsh conditions or exotic reagents that pose severe safety risks and environmental liabilities on an industrial scale. The lack of a defined, high-yielding pathway meant that supply chains for downstream drug candidates relying on this scaffold were inherently fragile and subject to volatile pricing structures driven by scarcity. Consequently, manufacturers faced substantial challenges in securing reliable [precise industry noun] supplier partnerships that could guarantee both volume and purity without exorbitant lead times.
The Novel Approach
The novel approach detailed in the patent data fundamentally alters the economic equation by utilizing a highly selective radical bromination strategy followed by a classic Balz-Schiemann type fluorination. Instead of struggling with unselective electrophilic substitutions, the process exploits the directing effects of the amino group to install bromine atoms precisely at the 3 and 5 positions before converting the amino group into fluorine. This strategic sequencing eliminates the formation of unwanted regioisomers at the source, thereby drastically reducing the burden on downstream purification units. The use of N-bromosuccinimide (NBS) as a brominating agent in the presence of a radical initiator allows for mild reaction conditions that preserve the integrity of the pyridine ring. This methodological innovation not only enhances the overall yield but also aligns perfectly with the industry's growing demand for cost reduction in [corresponding field noun] manufacturing by minimizing waste generation and solvent consumption.
Mechanistic Insights into Radical Bromination and Diazotization Fluorination
The core of this synthetic breakthrough lies in the precise control of radical chemistry during the initial bromination step. By employing azobisisobutyronitrile (AIBN) as a radical initiator in a non-polar solvent like carbon tetrachloride, the reaction generates bromine radicals that selectively attack the electron-rich positions ortho to the amino group on the pyridine ring. This mechanism ensures that the 3 and 5 positions are brominated efficiently while preventing over-bromination or substitution at other sites, a common pitfall in ionic bromination reactions. The subsequent conversion of the 3,5-dibromo-4-aminopyridine intermediate into the final fluoro-product involves the formation of a diazonium salt using sodium nitrite and hydrochloric acid at low temperatures. This unstable intermediate is then treated with fluoroboric acid, which facilitates the replacement of the diazonium group with a fluorine atom upon thermal decomposition. This sequence is chemically elegant because it transforms a readily available amine into a highly valued fluoride without requiring expensive transition metal catalysts or high-pressure equipment.
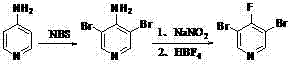
From an impurity control perspective, this mechanism offers distinct advantages for maintaining high-purity [precise industry noun] standards. The radical nature of the first step, combined with the specific stoichiometry of NBS (typically 1:2 to 1:2.5 molar ratio), limits the formation of mono-brominated byproducts or tribrominated species. Furthermore, the diazotization step is conducted under strictly controlled temperature conditions (0 to 5°C) to prevent the premature decomposition of the diazonium salt, which could lead to phenol formation or other hydrolysis byproducts. The final thermal decomposition at 50°C is optimized to ensure complete conversion to the fluoride while minimizing polymerization or tar formation. For Quality Control teams, this predictable mechanistic pathway translates into a cleaner crude profile, allowing for simpler recrystallization processes using solvents like n-hexane to achieve final purities exceeding 98%, which is critical for regulatory compliance in pharmaceutical applications.
How to Synthesize 3,5-Dibromo-4-Fluoropyridine Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and safety. The process begins with the suspension of 4-aminopyridine in carbon tetrachloride, followed by the controlled addition of NBS and AIBN, ensuring that the exothermic nature of the radical initiation is managed effectively. Once the bromination is complete, the intermediate is isolated and directly subjected to diazotization in an aqueous acidic medium, avoiding the need for extensive drying or purification between steps. This telescoped approach reduces processing time and solvent usage, contributing to the overall efficiency of the manufacturing campaign. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results with high fidelity.
- Perform radical bromination of 4-aminopyridine using NBS and AIBN in carbon tetrachloride at 50°C to obtain 3,5-dibromo-4-aminopyridine.
- Conduct diazotization of the intermediate with sodium nitrite and hydrochloric acid under controlled low temperatures.
- Add fluoroboric acid to the diazonium salt solution, heat to 50°C, and isolate the final 3,5-dibromo-4-fluoropyridine product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this synthesis route offers compelling qualitative advantages that directly impact the bottom line and operational resilience. The shift away from scarce or proprietary starting materials to commodity chemicals like 4-aminopyridine and NBS significantly de-risks the supply chain, ensuring that production schedules are not held hostage by the availability of exotic reagents. Moreover, the elimination of transition metal catalysts removes the need for costly and time-consuming heavy metal scavenging steps, which are often a bottleneck in API intermediate manufacturing. This simplification of the workflow not only accelerates batch turnover but also reduces the consumption of specialized adsorbents and filtration media, leading to substantial cost savings in raw material and operational expenditures.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy of the bromination step and the use of inexpensive inorganic reagents for the fluorination. By avoiding precious metal catalysts such as palladium or copper, which are prone to price volatility and require rigorous recovery systems, the process inherently lowers the variable cost per kilogram. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that the theoretical yield is closely approached in practice. This efficiency translates into a more competitive pricing structure for the final [precise industry noun], allowing downstream manufacturers to optimize their own cost models without compromising on quality specifications.
- Enhanced Supply Chain Reliability: The reliance on globally available bulk chemicals ensures a robust and continuous supply chain that is less susceptible to geopolitical disruptions or single-source supplier failures. Since 4-aminopyridine and sodium nitrite are produced at massive scales for various industries, securing long-term contracts for these inputs is straightforward and cost-effective. This stability allows for better forecasting and inventory management, reducing the need for safety stock and freeing up working capital. Furthermore, the simplicity of the reaction conditions means that the technology can be easily transferred between different manufacturing sites, providing redundancy and flexibility in the event of unplanned downtime at a primary facility.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations and moderate temperature ranges that do not require specialized high-pressure or cryogenic equipment. This ease of scale-up facilitates the rapid transition from kilogram to ton-scale production, meeting the demands of growing market segments without significant capital investment. From an environmental standpoint, the use of recyclable solvents and the generation of manageable inorganic byproducts simplify waste treatment protocols. The absence of persistent organic pollutants or heavy metal residues in the waste stream aligns with increasingly stringent global environmental regulations, reducing the liability and disposal costs associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 3,5-dibromo-4-fluoropyridine, based on the detailed specifications found in the patent literature. These insights are intended to clarify the feasibility of implementing this technology within existing manufacturing frameworks and to highlight the specific benefits for product development teams. Understanding these nuances is essential for making informed decisions about sourcing strategies and process optimization initiatives.
Q: What is the primary advantage of this synthesis route for 3,5-dibromo-4-fluoropyridine?
A: The primary advantage is the use of readily available 4-aminopyridine as a starting material and the avoidance of complex transition metal catalysts, leading to a simpler purification process and higher overall yields compared to traditional halogenation methods.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes specific radical initiators like AIBN to control regioselectivity during bromination, minimizing isomeric impurities, followed by a rigorous recrystallization step that achieves purity levels exceeding 98%.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction conditions operate at moderate temperatures (20°C to 50°C) and use common solvents like carbon tetrachloride and hydrochloric acid, making the thermal management and safety protocols straightforward for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dibromo-4-Fluoropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the advancement of modern therapeutics and crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,5-dibromo-4-fluoropyridine meets the exacting standards required for GMP environments. Our commitment to excellence extends beyond mere compliance; we actively collaborate with clients to optimize processes for maximum efficiency and sustainability.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific development goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into tangible value for your organization. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that you have all the necessary information to move your projects forward with confidence and speed.
