Revolutionizing Cephalosporin Side Chain Production: Advanced Aminothiazolyloximate Synthesis
Revolutionizing Cephalosporin Side Chain Production: Advanced Aminothiazolyloximate Synthesis
The pharmaceutical industry constantly seeks robust, scalable, and safe pathways for critical antibiotic intermediates, particularly for third-generation cephalosporins. Patent CN101805311B introduces a transformative synthetic method for 2-(2-amino-4-thiazolyl)-2-(Z)-methoxyimino acetic acid, commonly known as aminothiazolyloximate or AE-active ester. This compound serves as the pivotal side chain for potent antibiotics like Cefixime and Cefpodoxime, offering broad-spectrum efficacy against resistant pathogens. The disclosed technology addresses historical bottlenecks in heterocyclic synthesis by replacing hazardous gaseous reagents with safer solid alternatives and optimizing reaction kinetics through homogeneous phase engineering. By shifting from traditional chlorine gas chlorination to a triphosgene-based system and implementing a novel oximating agent system, this process ensures superior process control and environmental compliance.
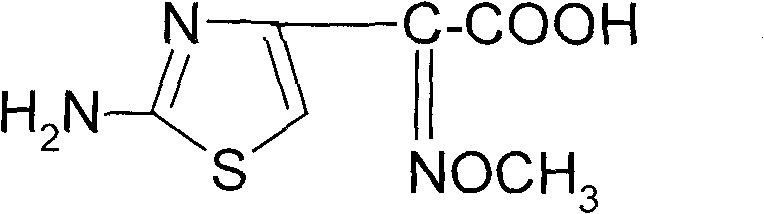
For global procurement teams and R&D directors, the implications of this patent extend beyond mere chemical curiosity; it represents a viable pathway for cost reduction in API intermediate manufacturing. The method achieves a finished product yield of not less than 90.5% with a purity exceeding 99.06%, demonstrating exceptional efficiency. As a reliable aminothiazolyloximate supplier, understanding these mechanistic advantages allows us to offer clients a supply chain that is not only economically competitive but also inherently safer and more sustainable. The transition to solid-state reagents and homogeneous reactions minimizes waste generation and simplifies downstream processing, directly impacting the bottom line for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aminothiazolyloximate has been plagued by significant safety hazards and inefficiencies inherent to older methodologies. Traditional routes often relied heavily on methyl acetoacetate or 4-chloroacetyl acetoacetic ester as starting materials, subjected to nitrosation using sodium nitrite and sulfuric acid. A critical flaw in these legacy processes is the heterogeneous nature of the nitrosation reaction, which creates mass transfer barriers that slow down reaction kinetics and prolong production cycles. Furthermore, the chlorination step traditionally utilized elemental chlorine gas, a highly toxic and corrosive substance that poses severe risks to operator health and environmental safety. Any leakage of chlorine gas can have deleterious impacts, requiring expensive containment infrastructure and rigorous safety protocols that drive up operational costs. Additionally, the uncontrollable nature of gas-liquid reactions often leads to inconsistent chlorination levels, resulting in lower yields and difficult-to-remove impurities that compromise the quality of the final beta-lactam antibiotic.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally reengineers the synthesis to overcome these structural weaknesses through chemical substitution and phase optimization. Instead of relying on dangerous chlorine gas, the new method employs triphosgene (bis(trichloromethyl) carbonate) as a solid chlorinating agent. Triphosgene decomposes in situ to generate phosgene equivalents in a controlled manner, allowing for precise stoichiometry and eliminating the risks associated with handling high-pressure toxic gases. Simultaneously, the nitrosation step is revolutionized by using a sodium nitrite and glacial acetic acid system, which facilitates a homogeneous reaction environment. This shift ensures that reactants are uniformly distributed at the molecular level, drastically accelerating the reaction rate and shortening the production cycle. The result is a streamlined process that not only enhances safety profiles but also delivers consistent, high-yield outputs suitable for commercial scale-up of complex heterocyclic intermediates.
Mechanistic Insights into Triphosgene-Mediated Chlorination and Homogeneous Oximation
The core of this technological advancement lies in the precise manipulation of reaction mechanisms to favor the desired Z-isomer while minimizing byproduct formation. In the homogeneous oximation step, the interaction between sodium nitrite and glacial acetic acid generates nitrous acid in situ within a single liquid phase. This eliminates the interfacial resistance found in solid-liquid or gas-liquid systems, allowing the nitrosation of methyl acetoacetate to proceed rapidly at low temperatures between 0°C and 5°C. The subsequent methylation reaction utilizes dimethyl sulfate under phase-transfer catalysis, ensuring efficient conversion to the methoxyimino derivative. The critical chlorination step involves the activation of the methoxyimino ketone by triphosgene in the presence of an organic base such as DMF or pyridine. The base catalyzes the decomposition of triphosgene, releasing reactive chlorinating species that selectively substitute the alpha-hydrogen without degrading the sensitive oxime functionality. This controlled release mechanism prevents over-chlorination and thermal runaways, which are common pitfalls in exothermic halogenation reactions.
Impurity control is rigorously maintained throughout the six-step sequence, particularly during the cyclization and hydrolysis phases. The cyclization of the chlorinated intermediate with thiourea and sodium acetate is conducted under mild conditions (20°C to 40°C) to promote the formation of the thiazole ring while preserving the stereochemical integrity of the methoxyimino group. The use of phase-transfer catalysts in this step further enhances the reaction efficiency, ensuring complete conversion of the chloro-intermediate. During hydrolysis, the ester group is cleaved using sodium hydroxide solution, followed by careful pH adjustment to precipitate the crude acid. The final refining step involves refluxing in alcohol solvents, which effectively removes residual organic impurities and isomers. This multi-stage purification strategy guarantees a final product with a melting point of 182.1°C to 183.9°C and purity levels surpassing 99.06%, meeting the stringent specifications required for high-purity cephalosporin side chains.
How to Synthesize Aminothiazolyloximate Efficiently
The synthesis of this critical antibiotic intermediate requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process begins with the preparation of the oxime precursor under homogeneous conditions, followed by methylation and the crucial triphosgene-mediated chlorination. Each step demands precise temperature control and stoichiometric balance to prevent the formation of degradation products. For detailed operational procedures, including specific solvent ratios, catalyst loading, and workup protocols, operators should refer to the standardized guidelines below. These steps represent the culmination of extensive process development aimed at maximizing throughput while minimizing environmental impact.
- Perform homogeneous oximation of methyl acetoacetate using sodium nitrite and glacial acetic acid at 0-5°C to form 2-hydroxamic ethyl acetoacetate.
- Execute methylation using dimethyl sulfate under phase-transfer catalysis at pH 8-10 to yield 2-methoxyimino ethyl acetoacetate.
- Conduct chlorination using solid triphosgene in organic solvent with an organic base at 10-20°C, replacing hazardous chlorine gas.
- Complete cyclization with thiourea and sodium acetate, followed by hydrolysis and final recrystallization to obtain the pure acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthetic route offers profound benefits for supply chain resilience and total cost of ownership. The elimination of chlorine gas from the process removes the need for specialized storage tanks, leak detection systems, and emergency scrubbing units, leading to substantial capital expenditure savings. Furthermore, the use of solid triphosgene simplifies logistics and inventory management, as it can be transported and stored with far fewer regulatory hurdles than compressed toxic gases. This shift significantly reduces the administrative burden and insurance costs associated with hazardous material handling. The improved reaction kinetics and higher yields directly translate to better asset utilization, allowing manufacturing facilities to produce more product in less time without expanding physical footprint. These efficiencies are critical for maintaining competitive pricing in the volatile market for beta-lactam intermediates.
- Cost Reduction in Manufacturing: The transition to a homogeneous oximation system and solid-state chlorination drastically simplifies the production workflow, removing the need for complex gas-handling infrastructure. By avoiding the use of expensive and hazardous chlorine gas, facilities can eliminate the costs associated with gas scrubbing and neutralization of acidic waste streams. The higher yields reported, with the ester intermediate exceeding 95.4% and the final acid product surpassing 90.5%, mean that less raw material is wasted per unit of output. This material efficiency, combined with reduced energy consumption due to shorter reaction times, results in significant operational cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Relying on solid reagents like triphosgene and sodium nitrite mitigates the risk of supply disruptions often associated with regulated hazardous gases. The robustness of the homogeneous reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. This reliability is essential for pharmaceutical manufacturers who require uninterrupted supplies of key starting materials to meet their own production schedules. The simplified process also allows for greater flexibility in scaling production volumes up or down in response to market demand, ensuring that procurement managers can secure stable long-term contracts without fear of capacity constraints.
- Scalability and Environmental Compliance: The process is inherently designed for green chemistry principles, minimizing the generation of toxic byproducts and hazardous waste. The replacement of chlorine gas eliminates the risk of atmospheric emissions, aligning with increasingly strict environmental regulations globally. The use of recyclable solvents and the ability to recover unreacted materials further enhance the sustainability profile of the manufacturing process. This environmental stewardship not only reduces disposal costs but also strengthens the brand reputation of suppliers as responsible partners. The scalability of the method from laboratory to industrial scale is proven by the high yields and purity, making it an ideal candidate for multi-ton annual production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of aminothiazolyloximate. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific antibiotic formulation pipelines.
Q: Why is triphosgene preferred over chlorine gas in this synthesis?
A: Triphosgene is a solid reagent that is significantly safer to store and transport compared to toxic chlorine gas. It allows for precise control of the chlorination reaction, reducing side reactions and improving operator safety while maintaining high yields above 95%.
Q: What is the achieved purity of the final aminothiazolyloximate product?
A: The optimized synthetic method described in patent CN101805311B achieves a finished product purity of not less than 99.06%, with a melting point range of 182.1°C to 183.9°C, meeting stringent requirements for third-generation cephalosporin production.
Q: How does the homogeneous oximation process improve efficiency?
A: By utilizing a sodium nitrite and glacial acetic acid system, the reaction becomes homogeneous rather than heterogeneous. This eliminates mass transfer limitations, significantly shortens the reaction cycle, and improves the overall yield of the oxime intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminothiazolyloximate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the side chain dictates the efficacy of the final antibiotic. Our technical team has thoroughly analyzed the advancements in aminothiazolyloximate synthesis to ensure our manufacturing capabilities align with the highest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver consistent quality. Our commitment to stringent purity specifications and rigorous QC labs ensures that every batch of AE-active ester meets the exacting requirements for third-generation cephalosporin synthesis, providing our partners with peace of mind and regulatory confidence.
We invite pharmaceutical manufacturers and procurement specialists to collaborate with us to optimize their supply chains. By leveraging our expertise in heterocyclic chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced synthesis capabilities can enhance your production efficiency and reduce your overall manufacturing costs while ensuring a secure and reliable supply of this critical pharmaceutical intermediate.
