Revolutionizing Valacyclovir Production: Enzymatic Catalysis for Commercial Scale-Up and Cost Efficiency
The pharmaceutical industry is constantly seeking greener, more efficient pathways for synthesizing critical antiviral agents, and patent CN112176011B presents a groundbreaking solution for the production of valacyclovir hydrochloride. This patent discloses a novel enzymatic catalysis method that transforms acyclovir, a potent but poorly bioavailable antiviral agent, into its L-valine ester prodrug, valacyclovir hydrochloride. Unlike traditional chemical synthesis routes that rely on harsh conditions and toxic reagents, this innovation utilizes papain, a readily available protease, to catalyze the direct esterification of acyclovir with L-valine hydrochloride in an aqueous environment. This shift represents a significant technological leap, addressing long-standing issues regarding safety, cost, and environmental impact in the manufacturing of high-purity pharmaceutical intermediates. By replacing complex multi-step protection and deprotection sequences with a single enzymatic step, the process not only streamlines production but also ensures a superior impurity profile, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their portfolios.
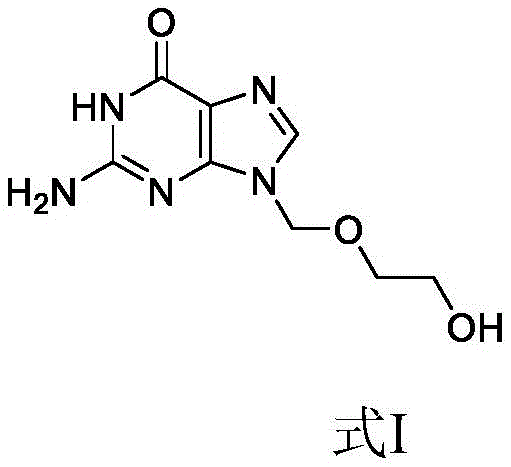
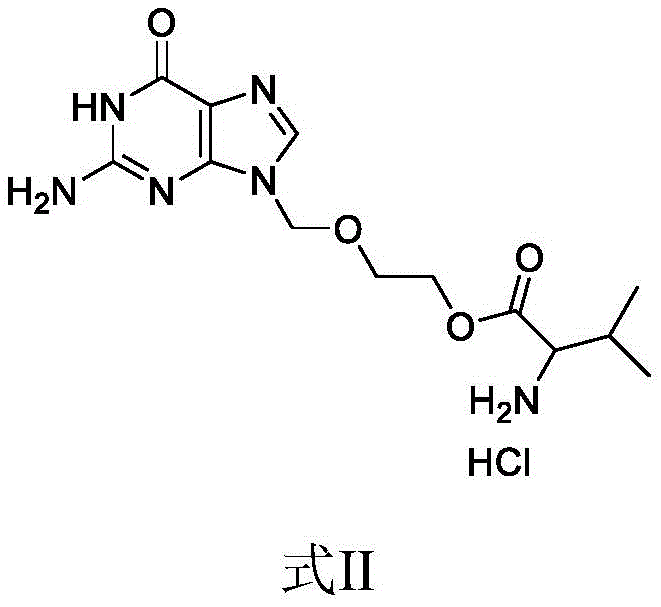
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of valacyclovir hydrochloride has been plagued by significant operational and economic challenges inherent to conventional chemical methodologies. Prior art, such as that described in US4957924, typically involves the use of Cbz-protected valine coupled with acyclovir using reagents like DCC and DMAP, followed by a hazardous hydrogenation step using palladium on carbon to remove the protecting group. This approach introduces severe safety risks due to the handling of hydrogen gas and necessitates the use of expensive noble metal catalysts, which drives up raw material costs substantially. Furthermore, alternative routes reported in documents like WO03041647 utilize EDC and Boc-protection strategies, which, while avoiding hydrogenation, suffer from the high cost of coupling reagents and the instability of ester bonds during acidic deprotection. These traditional pathways often result in complex post-treatment procedures, low overall yields, and the generation of substantial organic waste, creating a heavy burden on waste treatment facilities and complicating regulatory compliance for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the method disclosed in patent CN112176011B offers a streamlined, one-step enzymatic synthesis that fundamentally alters the production landscape for this critical antiviral intermediate. By employing papain as a biocatalyst in purified water, the process achieves direct esterification between acyclovir and L-valine hydrochloride under mild conditions, completely bypassing the need for protecting groups like Cbz or Boc. This elimination of protection-deprotection cycles drastically shortens the synthetic route, reducing both the time and the number of unit operations required to reach the final product. The use of water as the primary reaction medium not only aligns with green chemistry principles but also simplifies the isolation of the product through pH adjustment and anti-solvent crystallization. This novel approach effectively mitigates the safety hazards associated with high-pressure hydrogenation and removes the dependency on volatile organic solvents, thereby offering a robust and sustainable alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Papain-Catalyzed Esterification
The core of this technological advancement lies in the specific catalytic activity of papain, a cysteine protease that facilitates the formation of the ester bond between the hydroxyl group of acyclovir and the carboxyl group of L-valine. In the aqueous reaction system, papain acts as a highly selective biocatalyst, activating the carboxylic acid of the amino acid salt to react with the alcohol functionality of the nucleoside analogue. The reaction mechanism proceeds under mild thermal conditions, typically ranging from 10°C to 80°C, which preserves the structural integrity of the sensitive purine ring and prevents the degradation often seen in harsher chemical environments. The enzyme's specificity ensures that the reaction occurs predominantly at the desired primary hydroxyl position, minimizing the formation of regio-isomers and other structural impurities that are common in non-enzymatic esterifications. This high degree of selectivity is crucial for maintaining the stringent purity specifications required for pharmaceutical-grade materials, as it reduces the load on downstream purification processes.
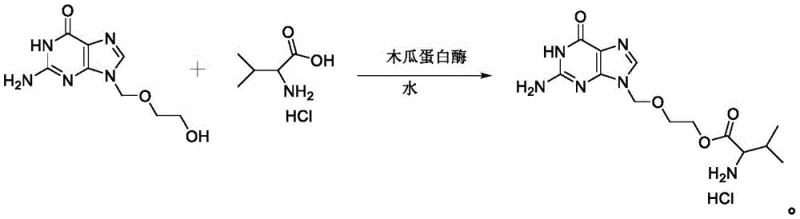
Furthermore, the impurity control mechanism in this enzymatic process is inherently superior due to the absence of reactive chemical coupling agents that typically generate persistent byproducts. In traditional DCC-mediated couplings, the formation of dicyclohexylurea (DCU) creates a difficult-to-remove solid waste that can trap product and lower yields. In the papain-catalyzed system, the only byproducts are water and potentially minor amounts of hydrolyzed starting materials, which are easily separated during the crystallization and washing steps. The process described involves cooling the reaction mixture to 0-10°C and adjusting the pH to 3.5-5.0 using dilute hydrochloric acid, which promotes the precipitation of the valacyclovir hydrochloride salt while keeping soluble impurities in the mother liquor. Subsequent recrystallization from methanol and acetone further refines the product, consistently achieving purity levels exceeding 99.5%, demonstrating the efficacy of the enzymatic route in delivering high-quality active pharmaceutical ingredients.
How to Synthesize Valacyclovir Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for manufacturing valacyclovir hydrochloride with high efficiency and minimal environmental footprint. The process begins by charging a reactor with acyclovir, L-valine hydrochloride, and purified water, followed by the addition of papain enzyme to initiate the biocatalytic transformation. Detailed operational parameters, including specific molar ratios, temperature gradients, and crystallization techniques, are critical for maximizing yield and ensuring batch-to-batch consistency. For a comprehensive understanding of the precise experimental conditions and step-by-step execution required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Combine acyclovir, L-valine hydrochloride, and purified water in a reactor, then add papain enzyme to initiate the esterification reaction under mild stirring.
- Maintain the reaction temperature between 10-80°C for 5-100 hours, then cool the mixture to 0-10°C to precipitate the crude product.
- Adjust the pH to 3.5-5.0 using dilute acid, add an anti-solvent like ethanol or acetone for crystallization, and purify via recrystallization in methanol/acetone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route offers compelling strategic advantages that directly impact the bottom line and operational resilience. The transition from chemical to enzymatic catalysis eliminates the reliance on volatile and expensive raw materials such as palladium on carbon, DCC, and EDC, which are subject to significant price fluctuations and supply constraints. By removing these costly reagents and the associated hazardous processing steps, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal. Additionally, the simplified workflow reduces the consumption of energy and solvents, further driving down the overall cost of goods sold (COGS) and enhancing the competitiveness of the final product in the global market.
- Cost Reduction in Manufacturing: The enzymatic process fundamentally lowers production costs by eliminating the need for expensive noble metal catalysts and complex protecting group chemistry. Traditional routes require significant investment in palladium catalysts and hazardous hydrogen gas infrastructure, along with the costs associated with their removal and recovery. In contrast, the papain-catalyzed method utilizes affordable, renewable enzymes and water as a solvent, drastically reducing reagent expenses. The simplified post-treatment process also lowers labor and utility costs, as there is no need for specialized high-pressure equipment or extensive solvent recovery systems, leading to a more economically efficient manufacturing model.
- Enhanced Supply Chain Reliability: Adopting this green synthesis method significantly de-risks the supply chain by reducing dependency on critical raw materials that are prone to shortages. Reagents like DCC and palladium catalysts often face supply bottlenecks due to their specialized nature and geopolitical factors affecting mining and chemical production. By shifting to widely available biological enzymes and commodity chemicals like L-valine hydrochloride and acyclovir, manufacturers can ensure a more stable and continuous supply of valacyclovir hydrochloride. This reliability is crucial for meeting the demands of large-scale pharmaceutical contracts and avoiding production delays caused by raw material scarcity.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing mild reaction conditions that are easily managed in large-scale reactors without the safety risks associated with high-pressure hydrogenation. The use of water as the primary solvent aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste discharge. This eco-friendly profile simplifies the permitting process for new production lines and reduces the liability associated with environmental compliance. The ease of waste treatment, primarily involving aqueous streams and benign salts, allows for rapid scale-up from pilot to commercial production volumes while maintaining a sustainable operational footprint.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial aspects of the enzymatic synthesis of valacyclovir hydrochloride, providing clarity for stakeholders evaluating this technology for integration into their manufacturing pipelines. These insights are derived directly from the technical disclosures and experimental data presented in patent CN112176011B, ensuring accuracy and relevance for decision-makers in the pharmaceutical sector.
Q: What are the primary advantages of the enzymatic route over traditional chemical synthesis for valacyclovir?
A: The enzymatic route described in patent CN112176011B eliminates the need for expensive noble metal catalysts like palladium on carbon and hazardous hydrogen gas. It utilizes water as a solvent instead of toxic organic solvents, significantly simplifying waste treatment and reducing environmental compliance costs while maintaining high product purity above 99.5%.
Q: How does the papain-catalyzed method impact impurity profiles compared to DCC/DMAP coupling?
A: Traditional methods using DCC or EDC often generate difficult-to-remove urea byproducts and require complex protection-deprotection steps (Cbz or Boc groups). The enzymatic method leverages the high specificity of papain, which minimizes side reactions and new impurity formation, resulting in a cleaner crude product that requires less intensive purification.
Q: Is this enzymatic process scalable for industrial production of antiviral intermediates?
A: Yes, the process is highly scalable as it operates under mild temperatures (10-80°C) and atmospheric pressure without requiring specialized high-pressure hydrogenation equipment. The use of robust, commercially available papain and simple aqueous workup procedures makes it ideal for large-scale commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valacyclovir Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of enzymatic catalysis in modern pharmaceutical manufacturing and are fully equipped to leverage technologies like patent CN112176011B for our clients. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our state-of-the-art facilities are designed to handle biocatalytic reactions with precision, supported by rigorous QC labs that guarantee stringent purity specifications for every batch of valacyclovir hydrochloride we produce. We are committed to delivering high-quality antiviral intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced enzymatic synthesis. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our expertise can enhance your project's success and accelerate your time to market.
