Advanced Iridium-Catalyzed Synthesis of Optically Pure 3-Aryl Sulfur-Substituted Propenes for Pharmaceutical Applications
Advanced Iridium-Catalyzed Synthesis of Optically Pure 3-Aryl Sulfur-Substituted Propenes for Pharmaceutical Applications
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient and stereoselective synthetic routes for complex molecular architectures. A pivotal advancement in this domain is detailed in patent CN102167674A, which discloses a robust method for synthesizing 3-aryl (alkyl) sulfur-substituted-1-propene compounds. These optically pure compounds are critical building blocks in the life sciences, finding extensive application in the synthesis of bioactive molecules, including cysteine derivatives, vitamin H analogs, and various protein cross-linking reagents. The significance of this technology lies in its ability to construct carbon-sulfur chiral centers with high precision, a task that has historically challenged organic chemists due to the strong coordinating nature of sulfur atoms which often deactivate transition metal catalysts. By leveraging a specialized iridium catalytic system, this invention provides a reliable pathway to access these valuable intermediates with exceptional enantiomeric excess, positioning it as a key technology for any reliable pharmaceutical intermediate supplier aiming to expand their portfolio of chiral synthons.
Historically, the construction of chiral carbon-sulfur bonds has relied heavily on Michael additions of sulfur nucleophiles to unsaturated ketones or aldehydes. While effective in certain contexts, these conventional methodologies frequently necessitate the use of stoichiometric amounts of chiral auxiliaries or organocatalysts, which inherently limits atom economy and generates substantial chemical waste. Furthermore, direct transition metal-catalyzed approaches have been hampered by the propensity of sulfur to poison the metal center, leading to low turnover numbers and poor catalytic activity. This limitation has restricted the development of scalable, catalytic processes for generating chiral sulfides, forcing many manufacturers to rely on resolution techniques that discard up to half of the produced material. The inability to efficiently utilize transition metals for this transformation has created a bottleneck in the supply chain for high-purity sulfur-containing intermediates, driving up costs and extending lead times for downstream drug development projects.
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes an iridium-catalyzed asymmetric allylic substitution reaction that effectively circumvents the issue of catalyst deactivation. By employing a specific class of chiral ligands in conjunction with an iridium precursor, the system maintains high catalytic activity even in the presence of soft sulfur nucleophiles. This method operates under remarkably mild conditions, typically between -20°C and 30°C, which preserves the integrity of sensitive functional groups often present in complex pharmaceutical substrates. The process demonstrates broad substrate scope, accommodating various aryl and alkyl thiolates as well as diverse allylic carbonate electrophiles, thereby offering a versatile platform for the synthesis of a wide array of chiral sulfur compounds. This technological leap represents a significant improvement in operational simplicity and environmental profile, making it an attractive option for cost reduction in fine chemical manufacturing where efficiency and selectivity are paramount.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Allylic Substitution
The core of this synthetic breakthrough lies in the unique interaction between the iridium metal center and the specialized chiral ligand system. The catalyst is generated in situ from a dimeric iridium precursor, [Ir(COD)Cl]2, and a chiral phosphine-amino ligand, which creates a sterically demanding environment around the metal. This specific coordination geometry is crucial for inducing high enantioselectivity during the nucleophilic attack. Unlike palladium systems which often form pi-allyl complexes susceptible to sulfur poisoning, the iridium complex forms a distinct metallacyclic intermediate that is more resistant to deactivation by the thiolate nucleophile. The ligand design, often featuring binaphthyl backbones with specific substituents as shown in the patent figures, dictates the facial selectivity of the allylic substitution, ensuring that the sulfur nucleophile attacks the allyl moiety from the desired trajectory to yield the (R) or (S) configured product with high fidelity.
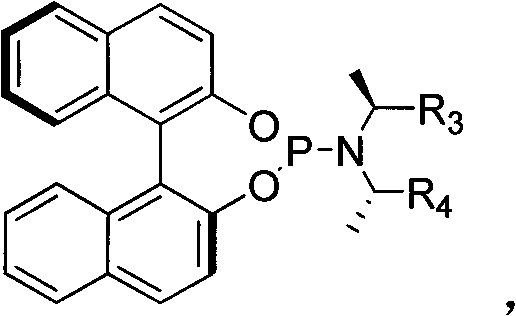
Furthermore, the inclusion of specific additives plays a pivotal role in modulating the reactivity and selectivity of the catalytic cycle. The patent highlights the efficacy of additives such as cesium fluoride, cesium carbonate, or silver bromide in enhancing the reaction outcome. These additives likely function by sequestering halide ions or activating the nucleophile, thereby facilitating the oxidative addition or reductive elimination steps within the catalytic cycle. The careful balance of these components allows for the suppression of side reactions, such as double bond isomerization or non-enantioselective background reactions, which are common pitfalls in allylic substitution chemistry. By optimizing the ratio of the iridium precursor, ligand, and additive, the process achieves regioselectivity ratios as high as 98:2 and enantiomeric excess values exceeding 99% in optimal cases. This level of control over the stereochemical outcome is essential for producing high-purity pharmaceutical intermediates that meet the stringent regulatory requirements for chiral drugs.
How to Synthesize 3-Phenylthio-1-Propene Derivatives Efficiently
The practical implementation of this chemistry involves a straightforward protocol that can be adapted for both laboratory scale discovery and larger scale production. The general procedure entails the preparation of the active iridium catalyst followed by the addition of the allylic carbonate substrate and the sodium thiolate nucleophile in a suitable organic solvent. The reaction is allowed to proceed under an inert atmosphere to prevent oxidation of the sensitive sulfur species or the catalyst. Following the completion of the reaction, which typically occurs within 2 to 48 hours depending on the substrate reactivity, the crude mixture is subjected to standard purification techniques such as column chromatography or recrystallization to isolate the target chiral sulfide. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare the catalyst by reacting [Ir(COD)Cl]2 with a chiral phosphine-amino ligand in an organic solvent.
- Mix the allyl carbonate substrate with sodium aryl thiolate and the prepared iridium catalyst in the presence of an additive like cesium fluoride.
- Stir the reaction mixture at temperatures between -20°C and 30°C for 2 to 48 hours, then isolate the product via chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iridium-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant simplification of the manufacturing process, which directly translates to improved operational efficiency and reduced overhead. By eliminating the need for stoichiometric chiral reagents and avoiding the wasteful resolution steps associated with older technologies, manufacturers can achieve a much higher effective yield of the desired enantiomer. This efficiency gain means that less raw material is required to produce the same amount of final product, leading to substantial cost savings in raw material procurement. Additionally, the mild reaction conditions reduce the energy burden on the production facility, as there is no need for extreme heating or cooling, further contributing to a lower carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The catalytic nature of this process implies that expensive chiral ligands and metal precursors are used in minute quantities relative to the substrate, drastically lowering the cost of goods sold compared to stoichiometric methods. The high selectivity minimizes the formation of diastereomers and regioisomers, which simplifies the purification process and reduces the consumption of silica gel and solvents during chromatography. Consequently, the overall waste disposal costs are significantly diminished, aligning with modern green chemistry principles and reducing the financial burden of environmental compliance.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for the use of commercially available starting materials, such as simple allylic carbonates and sodium thiolates, which are readily sourced from multiple suppliers. This diversity in raw material sourcing mitigates the risk of supply disruptions that can occur when relying on proprietary or niche reagents. Furthermore, the operational simplicity of the protocol reduces the likelihood of batch failures due to operator error or equipment limitations, ensuring a consistent and reliable flow of intermediates to downstream customers who depend on just-in-time delivery schedules.
- Scalability and Environmental Compliance: The method has been demonstrated to work effectively across a range of scales, from milligram discovery batches to multi-kilogram pilot runs, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates. The use of common organic solvents like dichloromethane or THF facilitates solvent recovery and recycling, which is a critical factor in large-scale industrial applications. By generating fewer byproducts and utilizing a catalytic system, the process inherently produces less hazardous waste, making it easier to obtain the necessary environmental permits and maintain compliance with increasingly strict global regulations on chemical manufacturing emissions.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled a set of frequently asked questions based on the technical specifications and experimental data provided in the patent literature. These answers address common concerns regarding catalyst stability, substrate compatibility, and purification strategies. Understanding these nuances is critical for R&D directors planning to integrate this chemistry into their existing process pipelines, as it allows for better risk assessment and resource allocation during the method transfer phase.
Q: Why is iridium catalysis preferred over traditional methods for chiral sulfur compounds?
A: Traditional methods often suffer from sulfur poisoning of the catalyst or require stoichiometric chiral reagents. The iridium-catalyzed asymmetric allylic substitution described in patent CN102167674A overcomes sulfur coordination issues, allowing for high enantioselectivity and catalytic efficiency under mild conditions.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction typically proceeds in organic solvents such as dichloromethane or THF at mild temperatures ranging from -20°C to 30°C. It utilizes an iridium complex generated in situ from [Ir(COD)Cl]2 and a chiral ligand, along with additives like cesium fluoride to enhance reactivity.
Q: How does this method impact production costs for pharmaceutical intermediates?
A: By employing a catalytic system with high regio- and enantioselectivity, this method reduces the need for expensive chiral resolving agents and minimizes waste generation. The mild operating conditions also lower energy consumption, contributing to overall cost reduction in manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl Sulfur-Substituted-1-Propene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN102167674A for accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into robust manufacturing processes. We are committed to delivering high-purity chiral sulfur intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation for chiral HPLC and NMR analysis. Our dedication to quality assurance guarantees that every batch of material we supply adheres to the highest industry standards, providing our partners with the confidence they need to advance their clinical programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of novel sulfur-containing scaffolds or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in iridium-catalyzed transformations can optimize your supply chain and reduce your overall development timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
