Scalable Metal-Free Synthesis of 3-Acyl Quinoxalinone Derivatives for Commercial API Production
Introduction to Advanced Quinoxalinone Functionalization
The pharmaceutical and agrochemical industries continuously demand efficient access to complex heterocyclic scaffolds, among which the quinoxalinone core stands out due to its profound biological significance. As detailed in Chinese Patent CN108033922B, a groundbreaking preparation method for 3-acyl quinoxalinone derivatives has been established, offering a transformative approach to constructing this vital pharmacophore. This technology addresses the critical need for sustainable chemistry by enabling the direct acylation of the quinoxalinone ring at the 3-position through a metal-free radical mechanism. Unlike traditional pathways that rely on harsh conditions or toxic heavy metals, this innovation utilizes inexpensive peroxide oxidants to drive the reaction under mild thermal conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing of high-purity pharmaceutical intermediates. The method's versatility allows for the use of diverse aldehydes and benzyl alcohols, expanding the chemical space available for drug discovery while simultaneously simplifying the supply chain logistics for commercial production.
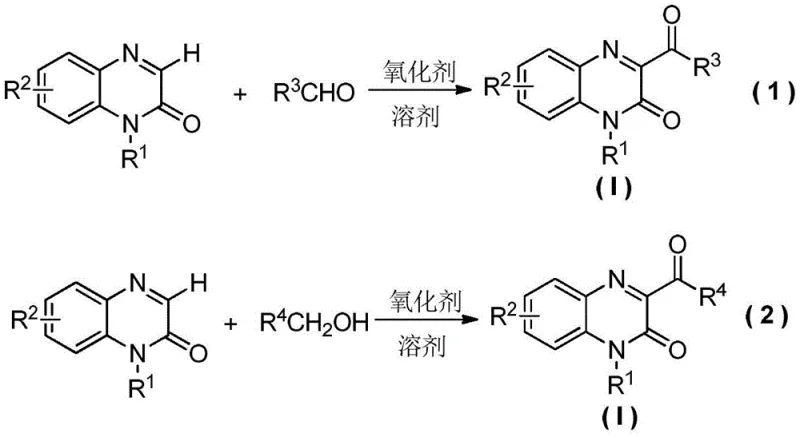
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-acyl quinoxalinone derivatives has been plagued by significant operational and economic inefficiencies that hinder large-scale adoption. As illustrated in the prior art analysis, conventional routes often involve multi-step sequences that drastically reduce overall atom economy and increase waste generation. For instance, one common pathway requires the initial condensation of o-phenylenediamine with bromo-ketoesters followed by a separate oxidative debromination step, which introduces unnecessary complexity and handling of hazardous halogenated intermediates. Another prevalent method relies on the use of transition metal catalysts, specifically silver salts like AgNO3, coupled with strong oxidants such as potassium persulfate. While effective on a small laboratory scale, these metal-catalyzed processes pose severe challenges for industrial application, including the high cost of noble metals and the rigorous, expensive purification steps required to remove trace metal residues to meet stringent pharmaceutical regulatory standards. Furthermore, some existing protocols necessitate high temperatures exceeding 110°C and prolonged reaction times, leading to energy inefficiency and potential safety hazards associated with thermal runaway in bulk reactors.
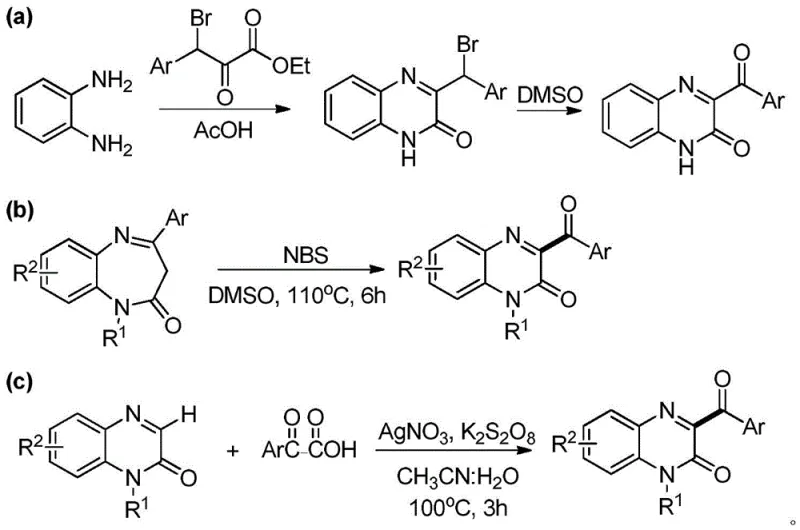
The Novel Approach
In stark contrast to these legacy technologies, the novel method disclosed in CN108033922B offers a streamlined, one-pot solution that fundamentally redefines the synthetic landscape for these derivatives. This approach leverages a metal-free radical C-H functionalization strategy, directly coupling substituted quinoxalin-2-ones with readily available aldehydes or benzyl alcohols. By eliminating the need for pre-functionalized starting materials like bromo-esters or expensive silver catalysts, the process achieves superior atom economy and significantly lowers the cost of goods sold (COGS). The reaction proceeds smoothly in common organic solvents such as 1,2-dichloroethane or acetonitrile, utilizing aqueous tert-butyl hydroperoxide (TBHP) as a benign and potent oxidant. This shift not only simplifies the operational workflow by reducing the number of unit operations but also enhances the environmental profile of the manufacturing process, aligning perfectly with modern green chemistry principles. The ability to tolerate a wide range of substituents on both the quinoxalinone ring and the acyl group ensures that this method is robust enough for the diverse needs of medicinal chemistry programs aiming to optimize structure-activity relationships rapidly.
Mechanistic Insights into Metal-Free Radical Acylation
Understanding the underlying chemical mechanism is crucial for R&D teams aiming to optimize this process for specific substrate classes. The reaction is initiated by the thermal decomposition of the peroxide oxidant, such as TBHP, which generates tert-butoxy radicals and hydroxyl radicals upon heating. These reactive species abstract a hydrogen atom from the aldehyde substrate, generating a nucleophilic acyl radical intermediate. This acyl radical then selectively attacks the electron-deficient C-3 position of the quinoxalin-2-one ring, forming a new carbon-carbon bond and a transient nitrogen-centered radical intermediate. The elegance of this mechanism lies in its subsequent oxidation step, where the intermediate undergoes single-electron transfer (SET) facilitated by the oxidant system, eventually leading to the restoration of aromaticity and the formation of the stable 3-acyl quinoxalinone product. This radical pathway avoids the formation of organometallic complexes, thereby sidestepping the issues of catalyst deactivation or ligand optimization that often plague transition metal catalysis. The entire cycle is driven by thermodynamic stability and radical kinetics, ensuring high conversion rates without the need for exotic reagents.
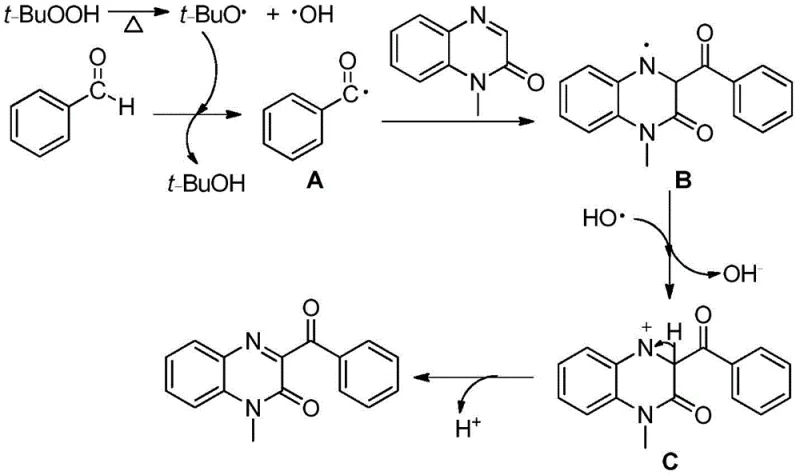
From an impurity control perspective, this mechanism offers distinct advantages over ionic or metal-catalyzed pathways. Since the reaction does not involve strongly basic or acidic conditions typically required for enolate chemistry or metal coordination, the risk of side reactions such as hydrolysis of the lactam ring or polymerization of the aldehyde is minimized. The primary byproducts are typically tert-butanol and water, which are easily removed during the aqueous workup phase, resulting in a crude product profile that is significantly cleaner than those obtained from silver-catalyzed methods. This inherent cleanliness reduces the burden on downstream purification processes, such as column chromatography or recrystallization, allowing for higher recovery yields of the final API intermediate. Furthermore, the radical nature of the reaction ensures excellent regioselectivity for the C-3 position, preventing the formation of difficult-to-separate regioisomers that could compromise the purity specifications required for clinical grade materials. This level of control is essential for maintaining batch-to-batch consistency in a commercial manufacturing environment.
How to Synthesize 3-Acyl Quinoxalinone Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal management to maximize yield and safety. The general protocol involves dissolving the quinoxalinone substrate and the aldehyde or alcohol coupling partner in a solvent like 1,2-dichloroethane, followed by the slow addition of the oxidant to control the rate of radical generation. Maintaining the reaction temperature within the optimal window of 60-90°C is critical; temperatures that are too low may result in incomplete initiator decomposition, while excessive heat could lead to non-selective radical degradation. The reaction progress should be monitored via TLC or HPLC, with typical completion times ranging from 5 to 9 hours depending on the electronic nature of the substrates. Upon completion, the mixture is subjected to a standard extractive workup using ethyl acetate and brine, followed by drying and concentration. The final purification is efficiently achieved using silica gel column chromatography, yielding the target 3-acyl quinoxalinone as a high-purity solid suitable for further biological evaluation or process development.
- Charge a reaction vessel with substituted quinoxalin-2-one derivatives and aldehyde or benzyl alcohol substrates in a suitable organic solvent such as 1,2-dichloroethane or acetonitrile.
- Add the oxidant, specifically 70% aqueous tert-butyl hydroperoxide (TBHP) or di-tert-butyl peroxide (DTBP), maintaining a molar ratio of substrate to oxidant between 1: 3 and 1:6 depending on the alcohol or aldehyde source.
- Heat the reaction mixture to a temperature range of 60-90°C for 5 to 9 hours under air atmosphere, followed by standard aqueous workup and column chromatography purification to isolate the high-purity 3-acyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible strategic benefits that extend far beyond simple chemical transformation. The elimination of transition metal catalysts, particularly expensive silver salts, removes a significant variable cost component from the bill of materials, directly contributing to substantial cost savings in pharmaceutical intermediate manufacturing. Moreover, the reliance on commodity chemicals like benzaldehydes and benzyl alcohols, which are produced on a massive global scale, ensures a stable and resilient supply chain that is less susceptible to the geopolitical fluctuations often associated with rare earth or precious metal sourcing. The operational simplicity of the process, which runs under air atmosphere and tolerates aqueous oxidant solutions, reduces the need for specialized inert gas infrastructure and rigorous moisture control, thereby lowering capital expenditure requirements for facility upgrades. These factors combined create a manufacturing profile that is not only economically superior but also logistically more robust, enabling reliable delivery schedules even in volatile market conditions.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of noble metal catalysts from the process flow. In traditional silver-catalyzed methods, the cost of the metal itself, combined with the expensive ligands and the specialized scavenging resins required to remove trace metal impurities to ppb levels, creates a heavy financial burden. By switching to an organic peroxide-mediated radical system, manufacturers can eliminate these line items entirely. Additionally, the high atom economy of the direct C-H functionalization means that less raw material is wasted as byproduct, further driving down the effective cost per kilogram of the active ingredient. The simplified workup procedure also reduces solvent consumption and labor hours, compounding the savings across the entire production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for API production, and this method strengthens resilience by utilizing widely available, off-the-shelf reagents. Unlike specialized organometallic reagents that may have long lead times or single-source suppliers, the aldehydes and peroxides used here are standard industrial chemicals with multiple qualified vendors globally. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the stability of the reagents allows for bulk purchasing and long-term storage without significant degradation, enabling strategic stockpiling to buffer against market volatility. The robustness of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or CDMO partners without requiring extensive re-validation of sensitive parameters.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental liabilities, but this technology is inherently designed for growth. The absence of heavy metals simplifies wastewater treatment and waste disposal, significantly reducing the environmental compliance costs associated with hazardous waste management. The reaction exotherm is manageable within standard jacketed reactors, and the use of aqueous TBHP solutions minimizes the risks associated with handling neat organic peroxides. This green chemistry profile aligns with increasingly strict global environmental regulations, future-proofing the manufacturing asset against tightening emission standards. The ability to run the reaction at moderate temperatures (60-90°C) also reduces energy consumption compared to high-temperature alternatives, contributing to a lower carbon footprint for the final product, a key metric for modern sustainable procurement strategies.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the scope and limitations of the patent CN108033922B. These insights are derived directly from the experimental data and mechanistic studies presented in the intellectual property documentation. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation. The following responses address critical aspects of catalyst usage, substrate compatibility, and operational parameters that define the practical utility of this synthesis method in a commercial context.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN108033922B is explicitly metal-free. It utilizes organic peroxides like TBHP to generate radicals, eliminating the need for costly silver salts or other transition metals, which significantly reduces raw material costs and simplifies downstream metal removal processes.
Q: What are the typical reaction conditions and yields for this process?
A: The reaction operates under mild conditions, typically between 60°C and 90°C, and completes within 5 to 9 hours. Experimental data indicates high synthesis efficiency, with isolated yields consistently reaching above 80% for various substrates including those with electron-donating or withdrawing groups.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly suitable for scale-up. It uses commercially available raw materials, avoids sensitive anhydrous conditions by tolerating aqueous oxidant solutions, and proceeds under air atmosphere, making it robust and safe for large-scale manufacturing of complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acyl Quinoxalinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-acyl quinoxalinone derivatives meets the highest quality standards required for pharmaceutical applications. We understand the critical importance of supply security and cost efficiency, and we are committed to leveraging advanced technologies like the metal-free radical acylation method to deliver superior value to our global clients.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments that will accelerate your development timeline. Let us collaborate to bring your next-generation therapeutics to market faster and more efficiently.
