Streamlining Nilotinib Production: A Technical Analysis of Direct Condensation Technology
The pharmaceutical landscape for tyrosine kinase inhibitors continues to evolve, driven by the urgent need for more efficient and sustainable manufacturing processes for critical oncology treatments. Patent CN103288804A introduces a significant technological breakthrough in the synthesis of Nilotinib, a potent second-generation BCR-ABL inhibitor used for treating chronic myeloid leukemia. Unlike traditional multi-step pathways that rely on complex cyclization or hazardous halogenation, this patent discloses a streamlined one-step condensation reaction. By directly coupling 4-methyl-3-[[4-(3-pyridyl)-2-pyrimidyl]amino]benzoic acid with a specific trifluoromethyl-substituted aniline derivative, the process achieves high purity and yield while drastically reducing the environmental footprint. This technical insight report analyzes the mechanistic advantages and commercial implications of this novel route for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Nilotinib has been plagued by synthetic inefficiencies and safety concerns inherent in earlier patented methodologies. As illustrated in the prior art, conventional routes often involve the construction of the pyrimidine ring through guanidine intermediates or rely on harsh halogenation strategies. For instance, some established methods utilize 3-iodo-4-methylbenzoyl chloride as a starting material, necessitating the use of toxic chlorinating agents such as phosphorus trichloride, phosphorus oxychloride, or thionyl chloride. These reagents not only pose severe occupational health risks but also generate substantial hazardous waste streams that require costly disposal protocols. Furthermore, alternative routes depicted in early world patents involve forming a guanidine fragment followed by cyclization with DMF-DMA, a process characterized by long reaction times and suboptimal yields that hinder economic viability.
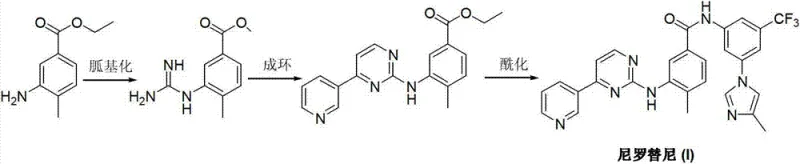
Another significant bottleneck in traditional synthesis is the reliance on transition metal catalysis for key coupling steps. Certain legacy pathways require the nucleophilic substitution of halogenated intermediates using expensive palladium catalysts to install the pyrimidine moiety. This dependency introduces critical supply chain vulnerabilities, including the volatility of precious metal prices and the rigorous regulatory requirements for removing trace metal impurities from the final Active Pharmaceutical Ingredient (API). The presence of residual palladium can compromise product quality and necessitate additional purification stages, thereby extending production lead times and inflating operational expenditures. These cumulative factors render conventional methods less attractive for modern, high-volume manufacturing environments focused on green chemistry principles.
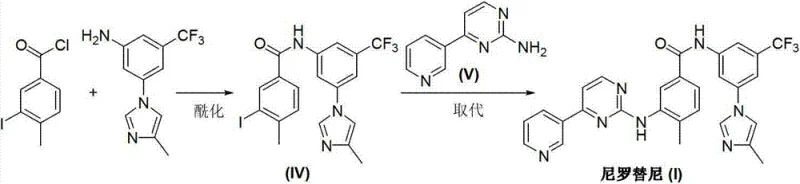
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN103288804A represents a paradigm shift towards simplicity and efficiency. The core innovation lies in the direct condensation of two advanced intermediates: 4-methyl-3-[[4-(3-pyridyl)-2-pyrimidyl]amino]benzoic acid (II) and 5-(4-methyl-1H-imidazole-1-yl)-3-trifluoromethyl phenylamine (III). This approach effectively bypasses the need for in-situ ring construction or hazardous halogen exchange reactions. By leveraging modern peptide coupling technology adapted for small molecule synthesis, the reaction proceeds under mild conditions with exceptional selectivity. The elimination of heavy metal catalysts and toxic chlorinating reagents not only simplifies the workflow but also aligns the manufacturing process with stringent international environmental regulations, offering a robust solution for sustainable API production.
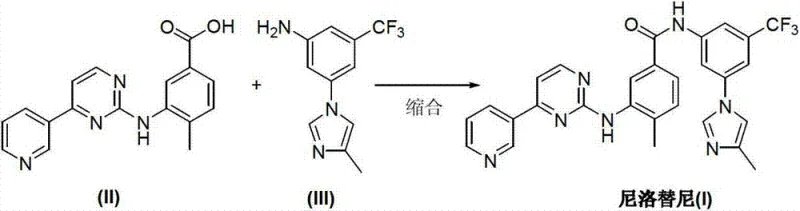
Mechanistic Insights into Organic Base-Promoted Condensation
The success of this novel synthetic route hinges on the precise selection of condensing agents and organic base promoters that facilitate the formation of the amide bond without compromising the integrity of sensitive functional groups. The patent specifies the use of phosphonium or uronium-based coupling reagents such as BOP (benzotriazole-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate), HBTU, or HATU. These agents activate the carboxylic acid group of intermediate (II) to form a highly reactive O-acylisourea or active ester intermediate, which is then susceptible to nucleophilic attack by the amine group of intermediate (III). The presence of strong non-nucleophilic bases like DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) or DBN plays a crucial role in scavenging the proton released during the reaction, thereby driving the equilibrium towards product formation and preventing the racemization or degradation of the chiral centers, although Nilotinib itself is achiral, this ensures overall structural fidelity.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed alternatives. The absence of transition metals eliminates the risk of metal-catalyzed side reactions, such as dehalogenation or homocoupling, which are common pitfalls in palladium-mediated processes. Furthermore, the reaction conditions—typically maintained between 50°C and 70°C in solvents like acetonitrile—are sufficiently mild to prevent the decomposition of the trifluoromethyl and imidazole moieties, which can be sensitive to extreme thermal stress or strong acidic/basic environments found in older methods. The resulting crude product exhibits a cleaner profile, significantly reducing the burden on downstream purification units and ensuring that the final API meets the rigorous purity specifications required for oncology therapeutics.
How to Synthesize Nilotinib Efficiently
The implementation of this condensation strategy requires careful attention to stoichiometry and reaction parameters to maximize yield and minimize byproduct formation. The patent recommends a molar ratio of the acid to the amine ranging from 1:1 to 1:2, with a preferred range of 1:1.3 to 1:6 to ensure complete consumption of the limiting reagent. Solvent selection is also critical, with acetonitrile identified as the optimal medium due to its ability to dissolve both reactants and the coupling agents while maintaining a stable reaction environment. The following guide outlines the standardized operational procedure derived from the patent embodiments, providing a clear roadmap for process chemists aiming to adopt this technology.
- Prepare the reaction mixture by combining 4-methyl-3-[[4-(3-pyridyl)-2-pyrimidyl]amino]benzoic acid (II) and a condensing agent such as BOP or HBTU in a solvent like acetonitrile.
- Add an organic base promoter such as DBU or DBN to activate the carboxylic acid and facilitate the nucleophilic attack.
- Introduce 5-(4-methyl-1H-imidazole-1-yl)-3-trifluoromethyl phenylamine (III) and maintain the temperature between 50-70°C to complete the amide bond formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this direct condensation technology translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary value proposition lies in the drastic simplification of the raw material portfolio. By utilizing commercially available advanced intermediates rather than commodity chemicals that require extensive in-house modification, manufacturers can reduce their exposure to upstream supply volatility. The elimination of specialized reagents like phosphorus oxychloride and palladium catalysts removes significant cost drivers associated with hazardous material handling, storage, and the complex waste treatment protocols mandated by environmental agencies. This streamlining of the input supply chain enhances overall operational agility and reduces the total cost of ownership for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic impact of removing expensive transition metal catalysts cannot be overstated. Palladium and other precious metals represent a significant capital tie-up and introduce price volatility risks into the cost structure. By shifting to an organic base-promoted system using reagents like BOP or HBTU, the process achieves substantial cost savings through the avoidance of metal recovery systems and the associated analytical testing for residual metals. Additionally, the higher atom economy of the direct condensation means less raw material is wasted as byproducts, leading to improved material utilization rates and lower variable costs per kilogram of finished API.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials significantly de-risks the supply chain. Traditional routes often depend on custom-synthesized building blocks or reagents with limited global suppliers, creating potential bottlenecks. In contrast, the intermediates required for this novel route are more commoditized, allowing for multi-sourcing strategies that ensure continuity of supply. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by equipment limitations or strict environmental controls, facilitating a more predictable and reliable delivery schedule for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: From a scale-up perspective, the mild thermal profile (50-70°C) and the absence of exothermic hazards associated with chlorinating agents make this process inherently safer for large-scale reactors. This safety margin allows for faster batch cycles and reduced downtime for cleaning and maintenance. Furthermore, the reduction in hazardous waste generation aligns with increasingly strict global environmental standards, minimizing the regulatory burden and potential fines associated with non-compliance. This 'green' credential is becoming a key differentiator in vendor selection processes for major pharmaceutical companies committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the technology for potential licensees and manufacturing partners.
Q: What are the primary advantages of the direct condensation method for Nilotinib compared to traditional routes?
A: The direct condensation method eliminates the need for toxic chlorinating agents like phosphorus oxychloride and expensive palladium catalysts required in halogenation routes. It offers higher atom economy, milder reaction conditions (50-70°C), and simplifies the purification process by avoiding heavy metal residues.
Q: Which condensing agents are most effective for this specific synthesis?
A: Patent CN103288804A identifies phosphonium-based coupling reagents such as benzotriazole-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP) and HBTU as highly effective. These agents, combined with strong organic bases like DBU or DBN, ensure high conversion rates and selectivity.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes readily available raw materials, avoids hazardous reagents that complicate waste treatment, and operates under mild thermal conditions, making it safer and more cost-effective for commercial scale-up compared to multi-step cyclization methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nilotinib Supplier
The technical advancements detailed in patent CN103288804A underscore the potential for a more efficient and sustainable future in oncology drug manufacturing. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Nilotinib intermediate or API we produce adheres to the highest global quality standards. We understand the critical nature of supply continuity in the pharmaceutical sector and are committed to being a dependable partner in your value chain.
We invite you to leverage our technical expertise to optimize your sourcing strategy. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel condensation route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both quality and profitability in your operations.
