Advanced Halogenation Technology for Scalable Production of Critical Prasugrel Intermediates
Advanced Halogenation Technology for Scalable Production of Critical Prasugrel Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiplatelet agents, particularly for the production of Prasugrel and its derivatives. A pivotal breakthrough in this domain is documented in patent CN101888988A, which discloses a novel and highly efficient process for the preparation of compounds of general formula (III). These 2-halo-1-cyclopropyl-2-substituted acetophenones serve as essential building blocks in the synthesis of thienopyridine derivatives. The innovation lies in a green oxidative halogenation strategy that replaces hazardous traditional reagents with a mixture of aqueous hydrogen halide and hydrogen peroxide. This technical advancement addresses long-standing challenges regarding purity, safety, and environmental compliance in the manufacturing of high-value pharmaceutical intermediates.
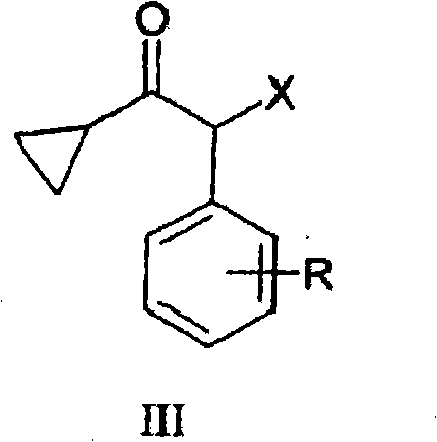
For procurement specialists and supply chain managers, the shift towards this methodology represents a significant opportunity for cost reduction in API manufacturing. By utilizing readily available commodity chemicals instead of specialized halogenating agents, the process inherently lowers raw material costs while simplifying the supply chain logistics. The ability to produce high-purity intermediates without resorting to complex purification techniques like column chromatography further enhances the economic viability of this route. As a reliable pharmaceutical intermediate supplier, understanding these underlying technological shifts is crucial for securing long-term supply continuity and maintaining competitive pricing structures in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-halo-1-cyclopropyl-2-substituted acetophenones has relied on methods that are fraught with significant technical and safety drawbacks. Traditional approaches often employ N-bromosuccinimide (NBS) in toxic solvents such as carbon tetrachloride or utilize elemental bromine, which poses severe corrosion risks and requires specialized reactor construction. Data from prior art indicates that reactions using NBS frequently result in crude product purities as low as 68%, accompanied by substantial amounts of unreacted starting material and difficult-to-remove isomers. Furthermore, the use of elemental halogens often leads to undesirable side reactions, specifically the opening of the sensitive cyclopropane ring, generating single and double bromo impurities that complicate downstream processing. These conventional methods not only burden the environment with hazardous waste but also necessitate expensive purification steps that are ill-suited for large-scale industrial application.
The Novel Approach
In stark contrast, the method disclosed in the patent utilizes an oxidative halogenation system comprising aqueous hydrogen peroxide and hydrogen halides within a water-miscible solvent matrix. This approach effectively generates the active halogenating species in situ, thereby avoiding the handling of dangerous elemental halogens or unstable reagents. Experimental results demonstrate that this novel route can achieve crude product purities approaching 90%, with specific examples showing contents of the desired formula (IV) compound reaching up to 94% when using alkali metal halides. The process significantly suppresses the formation of ring-opened byproducts, reducing dichloro or dibromo impurities to levels below 2%. This dramatic improvement in selectivity allows for the isolation of high-purity intermediates through simple distillation or even direct use in subsequent steps, eliminating the need for resource-intensive column chromatography and marking a substantial leap forward in process efficiency.
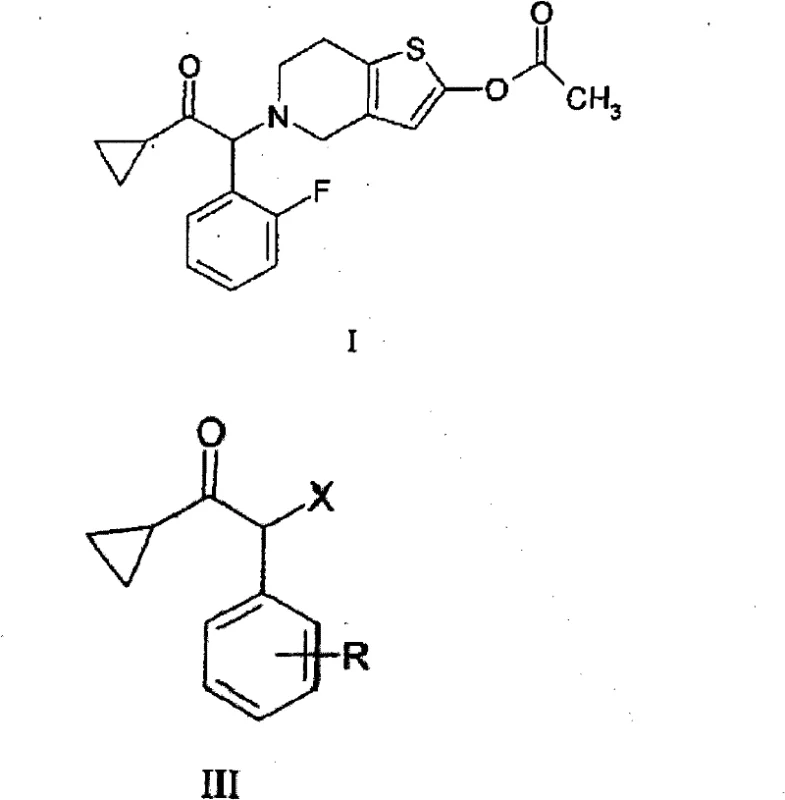
Mechanistic Insights into Oxidative Halogenation
The core of this technological advancement lies in the mechanistic elegance of the oxidative halogenation cycle. Rather than introducing pre-formed molecular halogen, the reaction relies on the oxidation of halide ions by hydrogen peroxide in an acidic medium. This in-situ generation ensures a controlled concentration of the active halogenating species, which is critical for preserving the integrity of the cyclopropane ring. The presence of a water-miscible solvent, such as dioxane, ethanol, or tetrahydrofuran, facilitates the solubility of the organic substrate while maintaining a homogeneous reaction environment that promotes efficient mass transfer. In embodiments where phase transfer catalysts like benzyltriethylammonium chloride are employed, the reaction kinetics are further enhanced, allowing for effective halogenation even in biphasic systems. This controlled reactivity is the key factor that minimizes the electrophilic attack on the cyclopropane ring, a common failure mode in traditional free-radical or harsh electrophilic halogenation protocols.
From an impurity control perspective, the mechanism offers distinct advantages over sulfuryl chloride or NBS mediated reactions. The absence of strong radical initiators like dibenzoyl peroxide reduces the likelihood of non-selective radical abstraction that leads to ring opening. Analytical data from the patent reveals that the ratio of mono-halo to di-halo impurities is heavily skewed towards the desired mono-substituted product. For instance, in chlorination variants using alkali metal chlorides and sulfuric acid, the formation of dichloro-derivatives is kept below 2.0%, ensuring that the crude product meets stringent quality specifications. This high level of chemoselectivity is paramount for R&D directors focused on minimizing the impurity profile of the final Active Pharmaceutical Ingredient (API), as it reduces the burden on downstream purification and ensures consistent batch-to-batch quality.
How to Synthesize 2-Halo-1-Cyclopropyl-2-Substituted Acetophenone Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction efficiency with operational safety. The process begins by combining the cyclopropylbenzyl ketone substrate with a water-miscible organic solvent and an aqueous source of the halide, such as hydrobromic acid or sodium bromide. Hydrogen peroxide is then introduced to drive the oxidation, with reaction temperatures typically maintained between 20°C and 100°C depending on the desired reaction rate and specific substrate reactivity. The detailed standardized synthetic steps for optimizing yield and purity are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale execution.
- Prepare a reaction mixture containing the cyclopropylbenzyl ketone substrate, a water-miscible organic solvent such as dioxane or ethanol, and an aqueous hydrogen halide solution.
- Add aqueous hydrogen peroxide solution to the mixture to initiate the in-situ generation of the halogenating species under controlled temperature conditions between 20°C and 100°C.
- Upon completion, isolate the crude product which typically exceeds 90% purity, optionally followed by simple vacuum distillation to achieve >97% purity without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxidative halogenation technology translates into tangible strategic benefits beyond mere chemical yield. The shift away from proprietary or specialized reagents like NBS towards commodity chemicals such as hydrogen peroxide and mineral acids fundamentally alters the cost structure of the intermediate. This substitution eliminates the volatility associated with the pricing of complex organic halogenating agents and ensures a more stable supply base. Additionally, the removal of toxic solvents like carbon tetrachloride simplifies waste management protocols and reduces the regulatory burden associated with hazardous material disposal, leading to significant operational cost savings.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive halogenating agents with low-cost oxidants and acids. By eliminating the need for column chromatography, a notoriously expensive and solvent-intensive purification step, manufacturers can drastically reduce both material and labor costs. The ability to use simple distillation or crystallization for purification further streamlines the production workflow, lowering energy consumption and increasing overall throughput capacity without requiring capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Relying on globally available commodity chemicals mitigates the risk of supply disruptions that often plague specialized reagent markets. The robustness of the reaction conditions allows for flexible sourcing of raw materials, ensuring continuous production capabilities even during market fluctuations. Furthermore, the simplified workup procedure reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand and shorter delivery cycles for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, utilizing standard reactor materials that do not require the exotic alloys needed for handling elemental halogens. The use of water-miscible solvents and the avoidance of persistent organic pollutants align with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. This scalability ensures that the transition from laboratory discovery to commercial tonnage production is seamless, supporting the growing global demand for antiplatelet therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational benefits and chemical advantages for potential partners and technical stakeholders.
Q: How does this new halogenation method improve upon traditional NBS bromination?
A: Traditional methods using N-bromosuccinimide (NBS) often result in significant impurities, including ring-opened byproducts, and require toxic solvents like carbon tetrachloride. The patented oxidative halogenation method achieves crude purities near 90% using safer, water-miscible solvents and eliminates the need for complex column chromatography purification.
Q: What represents the primary cost-saving mechanism in this synthetic route?
A: The primary cost reduction stems from replacing expensive halogenating agents like NBS or sulfuryl chloride with commodity chemicals such as hydrogen peroxide and hydrobromic acid. Furthermore, the elimination of toxic solvents and energy-intensive purification steps like column chromatography significantly lowers operational expenditures.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method is designed for industrial scale application. It utilizes standard reactor equipment, avoids hazardous elemental halogens that require special construction materials, and produces crude products pure enough for direct use or simple distillation, facilitating easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Halo-1-Cyclopropyl-2-Substituted Acetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative processes described in patents like CN101888988A are translated into reliable supply chains. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-halo-1-cyclopropyl-2-substituted acetophenone meets the exacting standards required for pharmaceutical synthesis.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall manufacturing costs.
