Revolutionizing Thiazoloquinoline Production: A Scalable One-Pot Strategy for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds that serve as critical backbones for bioactive molecules. A recent breakthrough detailed in patent CN108929337B introduces a highly efficient preparation method for 4,5-dihydrothiazolo[5,4-c]quinoline-2-amine derivatives, a structural motif known for its potent antibacterial, anti-inflammatory, and antitumor activities. This innovation addresses long-standing challenges in organic synthesis by replacing multi-step, labor-intensive protocols with a streamlined one-pot tandem reaction. By leveraging a cascade of Meyer-Schuster rearrangement, halogenation, and cyclization under acidic catalysis, this technology offers a robust solution for generating high-purity intermediates. For global procurement teams and R&D directors, understanding the mechanistic elegance and operational simplicity of this patent is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering complex scaffolds with enhanced economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4,5-dihydrothiazolo[5,4-c]quinoline core has been fraught with inefficiencies that hinder large-scale adoption. As illustrated in prior art literature, such as the route described in Tetrahedron (2006), the traditional synthesis relies on a cumbersome three-step sequence starting from 2-nitrophenylacetyl bromide. This legacy approach necessitates three distinct reaction environments, each requiring specific optimization and control parameters that complicate process engineering. Furthermore, the requirement to isolate the intermediate three times significantly inflates the cost of goods sold (COGS) due to material losses during purification, increased solvent consumption, and extended processing time. From an environmental perspective, the generation of substantial chemical waste through multiple work-up procedures makes this conventional route economically and ecologically disadvantageous for modern green chemistry standards.
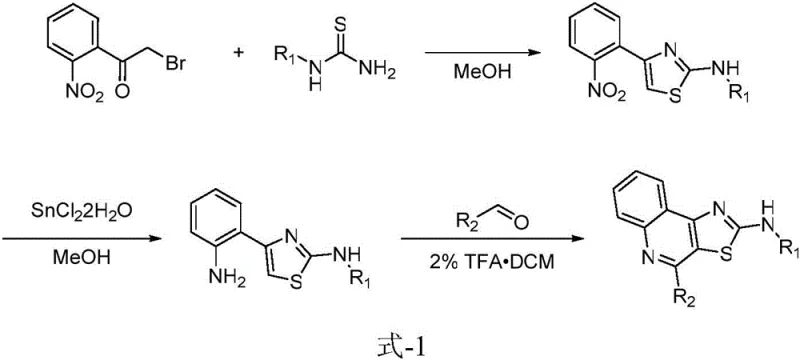
The Novel Approach
In stark contrast, the methodology disclosed in the present patent revolutionizes the synthetic landscape by consolidating the entire transformation into a single vessel. This novel approach utilizes N-protected o-propargyl alcohol-aniline derivatives as the starting material, which undergoes a seamless tandem reaction in the presence of N-halogenated succinimide and thiourea. The elimination of intermediate isolation steps not only accelerates the throughput but also drastically reduces the accumulation of impurities that often arise during transfer between reactors. By achieving yields as high as 90 percent under mild conditions, this one-pot strategy exemplifies process intensification. It allows manufacturers to bypass the logistical bottlenecks associated with multi-step synthesis, thereby enhancing the overall reliability of the supply chain for these valuable heterocyclic compounds.
![General reaction scheme for one-pot synthesis of 4,5-dihydrothiazolo[5,4-c]quinoline-2-amine](/insights/img/thiazoloquinoline-synthesis-one-pot-pharma-supplier-20260305201932-02.webp)
Mechanistic Insights into Acid-Catalyzed Tandem Cyclization
The success of this synthesis hinges on the precise orchestration of a tandem Meyer-Schuster rearrangement followed by halogenation and cyclization. Initially, the acid catalyst activates the propargylic alcohol moiety of the aniline derivative, facilitating a rearrangement that generates an alpha,beta-unsaturated ketone intermediate in situ. Subsequently, the N-halogenated succinimide acts as an electrophilic halogen source, introducing a halogen atom at the alpha-position of the newly formed ketone. This alpha-halo ketone is highly reactive towards nucleophilic attack by the sulfur atom of the thiourea derivative. The final cyclization step closes the thiazole ring, forming the fused thiazoloquinoline system. The choice of acid catalyst, ranging from trifluoromethanesulfonic acid to metal triflates like bismuth or scandium triflate, is critical for balancing the reaction rate and minimizing side reactions, ensuring high conversion rates without degrading the sensitive functional groups present on the aromatic rings.
Impurity control is inherently superior in this tandem system due to the "telescoped" nature of the reaction. In traditional stepwise synthesis, impurities generated in step one are carried forward and can react unpredictably in subsequent steps, leading to complex impurity profiles that are difficult to purge. Here, the reactive intermediates are consumed immediately as they are formed, preventing their accumulation. Moreover, the use of mild Lewis acids and controlled reflux conditions in solvents like dioxane or dichloroethane prevents the harsh degradation often seen with strong mineral acids. This results in a cleaner crude reaction mixture, which simplifies the downstream purification process, typically requiring only standard column chromatography or recrystallization to achieve the stringent purity specifications demanded by the pharmaceutical industry.
How to Synthesize 4,5-dihydrothiazolo[5,4-c]quinoline-2-amine Efficiently
The operational protocol for this synthesis is designed for maximum efficiency and reproducibility in a laboratory or pilot plant setting. The process begins by charging a sealed tube with the N-protected o-propargyl alcohol-aniline substrate, a suitable solvent such as dioxane or toluene, and a catalytic amount of acid. The mixture is heated under reflux until thin-layer chromatography (TLC) confirms the complete consumption of the starting aniline. At this juncture, the N-halogenated succinimide is introduced directly into the hot reaction mixture. Following further monitoring to ensure complete halogenation, an inorganic base like potassium carbonate and the thiourea component are added to trigger the final cyclization. The detailed standardized synthesis steps, including specific molar ratios and work-up procedures, are outlined below to guide technical teams in replicating this high-yielding transformation.
- Reflux N-protected o-propargyl alcohol-aniline with acid catalyst in a sealed tube until starting material disappears.
- Add N-halogenated succinimide to the reaction mixture and monitor conversion via TLC.
- Introduce inorganic base and thiourea derivative, then quench with water and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from multi-step to one-pot synthesis represents a paradigm shift in cost structure and risk management. The primary value driver here is the drastic simplification of the manufacturing workflow. By removing the need for intermediate isolation, the process eliminates the capital expenditure and operational overhead associated with additional filtration, drying, and storage equipment. This consolidation translates directly into cost reduction in pharmaceutical intermediate manufacturing, as fewer unit operations mean lower energy consumption and reduced labor hours per kilogram of product. Furthermore, the high atom economy and reduced solvent usage align with increasingly strict environmental regulations, mitigating the risk of compliance-related shutdowns or fines.
- Cost Reduction in Manufacturing: The economic benefits of this technology are derived from the fundamental elimination of processing steps rather than marginal improvements in yield. By avoiding the isolation of intermediates, manufacturers save significantly on solvent volumes, filtration media, and drying time. The ability to achieve yields up to 90 percent in a single pot means that raw material costs are optimized, as less starting material is wasted in purification losses. Additionally, the use of commodity chemicals like N-bromosuccinimide and thiourea ensures that input costs remain stable and predictable, shielding the supply chain from the volatility often associated with specialized reagents.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions and the availability of raw materials. The reagents required, such as N-protected anilines and succinimides, are widely available from global chemical suppliers, reducing the risk of single-source dependency. The tolerance of the reaction to various substituents on the aromatic ring allows for flexible sourcing of precursors. Moreover, the simplified process flow reduces the lead time for production batches, enabling faster response to market demand fluctuations. This agility is critical for maintaining inventory levels of high-purity pharmaceutical intermediates without the need for excessive safety stock.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of hazardous intermediates that require special handling. The reaction operates under standard reflux conditions in common organic solvents, which are easily managed in existing stainless steel reactors. From an environmental standpoint, the reduction in waste generation (E-factor) is substantial. Fewer work-up steps mean less aqueous waste and organic solvent discharge, simplifying wastewater treatment requirements. This green chemistry profile not only lowers disposal costs but also enhances the corporate sustainability metrics of the manufacturing entity, a key factor for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on catalyst selection, substrate compatibility, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: What are the primary advantages of this one-pot synthesis over traditional methods?
A: The novel method eliminates the need for three separate reaction environments and multiple intermediate isolations required by conventional routes, significantly reducing waste and operational complexity while achieving yields up to 90%.
Q: Which catalysts are compatible with this tandem cyclization process?
A: The process demonstrates versatility with various Lewis and Brønsted acids, including bismuth triflate, copper triflate, scandium triflate, and trifluoromethanesulfonic acid, allowing for optimization based on substrate sensitivity.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the use of readily available reagents like N-halosuccinimide and thiourea, combined with standard reflux conditions in common solvents like dioxane or toluene, supports robust scalability for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-dihydrothiazolo[5,4-c]quinoline-2-amine Supplier
As the demand for complex heterocyclic scaffolds continues to grow in the development of next-generation therapeutics, partnering with an experienced CDMO is vital for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 4,5-dihydrothiazolo[5,4-c]quinoline-2-amine derivatives we produce. We understand the critical nature of these intermediates in drug discovery and are committed to delivering materials that meet the highest quality standards.
We invite you to leverage our technical expertise to optimize your supply chain and reduce time-to-market for your drug candidates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us collaborate to bring your innovative pharmaceutical projects to fruition with efficiency and reliability.
