Scalable Manufacturing of Cyclosporin Derivatives via Copper-Catalyzed Substitution
Scalable Manufacturing of Cyclosporin Derivatives via Copper-Catalyzed Substitution
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of complex immunosuppressive agents, particularly cyclosporin derivatives which play a critical role in transplant medicine and autoimmune disease management. Recent intellectual property developments, specifically patent CN115298199A, have unveiled a transformative approach to synthesizing these high-value compounds with enhanced efficiency and purity profiles. This technical insight report analyzes the novel copper-catalyzed substitution method that replaces traditional drying agents with trialkylsilyl halides, offering a compelling value proposition for global supply chains. By addressing the inherent limitations of previous synthetic routes, this innovation ensures a more reliable pharmaceutical intermediates supplier capability for downstream API manufacturing. The following analysis details the mechanistic advantages and commercial implications of adopting this streamlined process for industrial-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sarcosine-modified cyclosporin compounds relied heavily on methods described in earlier patents such as US 6,583,265 and WO 2019/016572 A1, which necessitated the use of solid desiccants like molecular sieves to maintain anhydrous conditions. While effective in laboratory settings, the reliance on molecular sieves presents significant engineering bottlenecks when transitioning to commercial manufacturing scales. The filtration and removal of fine particulate drying agents from viscous reaction mixtures are time-consuming operations that increase batch cycle times and introduce potential risks of product loss through adsorption. Furthermore, the reproducibility of reactions utilizing solid desiccants can vary based on the activation state and particle size of the sieves, leading to inconsistent yields and impurity profiles that complicate regulatory validation. These operational inefficiencies directly impact the cost reduction in pharmaceutical intermediates manufacturing by inflating processing costs and reducing overall equipment effectiveness.
The Novel Approach
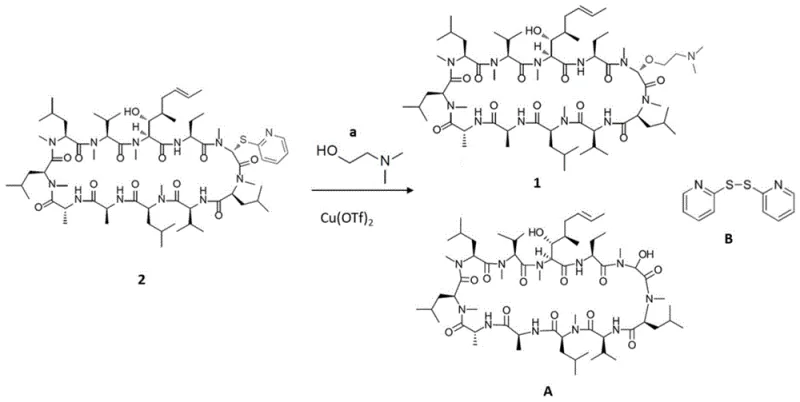
The methodology disclosed in CN115298199A introduces a paradigm shift by substituting solid molecular sieves with liquid trialkylsilyl halides, specifically trimethylchlorosilane (TMSCl), acting as a homogeneous water scavenger. This liquid additive integrates seamlessly into the reaction medium, eliminating the need for cumbersome filtration steps associated with solid desiccants and thereby streamlining the workup procedure. Experimental data within the patent indicates that the addition of TMSCl not only maintains the necessary anhydrous environment but actively enhances the conversion efficiency of the thiopyridyl intermediate to the desired cyclosporin derivative. By removing the physical barriers imposed by solid drying agents, manufacturers can achieve faster turnover rates and more consistent reaction kinetics, which are essential for maintaining a reliable pharmaceutical intermediates supplier status. This transition from heterogeneous to homogeneous water scavenging represents a critical optimization for process chemistry teams aiming to maximize throughput.
Mechanistic Insights into Copper-Catalyzed Sarcosine Substitution
The core of this innovative synthesis lies in the precise orchestration of a copper-catalyzed nucleophilic substitution at the sarcosine residue of the cyclosporin macrocycle. The process utilizes copper(II) triflate as a potent Lewis acid catalyst to activate the thiopyridyl leaving group, facilitating the attack by the aminoalcohol nucleophile under mild conditions. The inclusion of TMSCl plays a dual role: it scavenges trace water that could otherwise lead to hydrolysis of the sensitive intermediate, and it likely coordinates with the copper center to modulate its electrophilicity, thereby suppressing the formation of unwanted hydroxy-cyclosporin by-products. This mechanistic refinement is crucial for achieving high-purity pharmaceutical intermediates as it minimizes the generation of Side Product A, a hydrolytic adduct that is difficult to separate and reduces overall mass balance. The ability to control the impurity profile at the molecular level demonstrates a sophisticated understanding of organometallic catalysis in complex peptide systems.
Furthermore, the reaction conditions specified, such as the use of anhydrous THF and controlled temperature ranges between 0°C and room temperature, ensure the stability of the delicate cyclosporin backbone during the transformation. The stoichiometry is carefully balanced, with approximately 3.6 equivalents of copper salt and 2.4 equivalents of TMSCl relative to the substrate, ensuring complete consumption of the starting material without excessive reagent waste. This precision in reagent usage contributes to a cleaner reaction profile, as evidenced by HPLC analysis showing negligible amounts of starting material remaining after 16 hours. For R&D directors focused on commercial scale-up of complex pharmaceutical intermediates, this level of control over side reactions translates to simplified purification protocols and higher confidence in batch-to-batch consistency, ultimately securing the integrity of the final drug substance.
How to Synthesize Cyclosporin Derivatives Efficiently
Implementing this synthesis route requires strict adherence to anhydrous protocols and precise reagent dosing to replicate the high yields reported in the patent literature. The process begins with the preparation of the thiopyridyl intermediate, followed by the critical substitution step where the copper catalyst and silyl halide are introduced. Operators must ensure that all solvents and reagents are dried appropriately, as the presence of moisture can compromise the efficacy of the TMSCl scavenger and lead to increased hydrolysis. The reaction mixture is typically stirred for 16 hours to ensure full conversion, after which a standard aqueous workup involving pH adjustment and organic extraction isolates the crude product. Detailed standardized synthesis steps see the guide below.
- Activate Cyclosporin A by reacting with a disulfide compound to form a thiopyridyl intermediate.
- React the intermediate with an aminoalcohol in the presence of copper triflate and trimethylchlorosilane (TMSCl).
- Quench the reaction, extract with organic solvents, and purify via column chromatography to isolate the derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the elimination of molecular sieves offers tangible logistical and financial benefits that extend beyond simple reagent costs. The removal of solid filtration steps significantly reduces processing time, allowing manufacturing facilities to increase their batch frequency and overall production capacity without additional capital investment in hardware. This efficiency gain directly supports reducing lead time for high-purity pharmaceutical intermediates, enabling suppliers to respond more agilely to market demand fluctuations. Additionally, the liquid nature of TMSCl simplifies inventory management and handling compared to bulk solid desiccants that require regeneration or specialized disposal, thereby lowering the total cost of ownership for the manufacturing process. These operational improvements create a more resilient supply chain capable of sustaining continuous production schedules.
- Cost Reduction in Manufacturing: The substitution of molecular sieves with TMSCl eliminates the costs associated with the purchase, activation, and disposal of solid desiccants, while also reducing labor costs linked to filtration operations. By minimizing the formation of hydrolytic by-products, the process reduces the burden on downstream purification units, leading to substantial cost savings in chromatography resins and solvent consumption. The improved conversion efficiency means less starting material is wasted, optimizing the raw material utilization rate and enhancing the overall economic viability of the synthesis. These factors combine to deliver a significantly reduced cost of goods sold for the final derivative.
- Enhanced Supply Chain Reliability: Streamlining the workup procedure by removing solid filtration steps reduces the risk of batch failures due to clogging or incomplete drying, ensuring a more predictable production timeline. The use of common liquid reagents like TMSCl and copper triflate ensures that supply risks are minimized, as these materials are widely available from multiple global vendors compared to specialized grades of molecular sieves. This redundancy in the supply base protects against disruptions and guarantees a steady flow of materials to meet contractual obligations. Consequently, partners can rely on a more stable and consistent supply of critical intermediates for their own manufacturing pipelines.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction mixture facilitates easier scale-up from pilot plant to commercial tonnage, as heat and mass transfer are more uniform without solid suspensions. Reducing the volume of solid waste generated from spent molecular sieves aligns with green chemistry principles and simplifies environmental compliance reporting regarding hazardous waste disposal. The process operates at near-room temperature, reducing energy consumption for heating or cooling compared to more extreme conditions required by alternative methods. This sustainable approach enhances the corporate social responsibility profile of the manufacturing operation while maintaining high productivity standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this copper-catalyzed synthesis route. Understanding these nuances is vital for process engineers and quality assurance teams who are evaluating the feasibility of technology transfer. The answers are derived directly from the experimental data and embodiments provided in the source patent documentation, ensuring accuracy and relevance to real-world production scenarios. Reviewing these points helps mitigate risks associated with adopting new chemical methodologies.
Q: Why is TMSCl preferred over molecular sieves in this synthesis?
A: TMSCl acts as a water scavenger that is easier to handle on a large scale compared to solid molecular sieves, reducing filtration time and improving reaction conversion rates significantly.
Q: What is the role of Copper Triflate in the reaction?
A: Copper triflate serves as a Lewis acid catalyst that facilitates the nucleophilic substitution at the sarcosine residue, ensuring high selectivity and minimizing hydrolytic by-products.
Q: Can this method be applied to other Cyclosporin analogs?
A: Yes, the protocol is versatile and applicable to Cyclosporin C, D, and G derivatives by adjusting the starting cyclosporin substrate while maintaining the same catalytic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclosporin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly evaluated the copper-catalyzed route described in CN115298199A and confirmed its viability for large-scale production within our existing infrastructure. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and compliant with international regulatory standards. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of cyclosporin derivative meets the highest quality expectations for API synthesis.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for immunosuppressive intermediates. By leveraging this efficient synthesis technology, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Partnering with us ensures access to cutting-edge chemistry and a commitment to delivery excellence that drives your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
