Scalable Green Synthesis of 2-(3'-Quinolyl) Allyl Alcohol for Cardiovascular Drug Intermediates
The pharmaceutical industry continuously seeks robust and environmentally benign pathways for synthesizing complex heterocyclic intermediates, particularly those serving as precursors for cardiovascular therapeutics. Patent CN101792414A introduces a significant technological advancement in the preparation of 2-(3'-quinolyl) allyl alcohol, a critical building block in medicinal chemistry. This innovation leverages a novel catalytic system comprising a self-designed ionic liquid, specifically 1,3-bis(N-phenylcarbamoylethyl)imidazolium hexafluorophosphate, supported on solid magnesium oxide. By integrating this specialized ionic liquid with palladium acetate, the method achieves a highly efficient Heck coupling reaction that circumvents the limitations of traditional homogeneous catalysis. The strategic design of this process not only enhances reaction selectivity but also aligns with modern green chemistry principles by minimizing hazardous waste generation. For R&D directors and procurement specialists, this represents a viable pathway to secure high-purity intermediates while mitigating the environmental liabilities associated with conventional organophosphorus ligands.
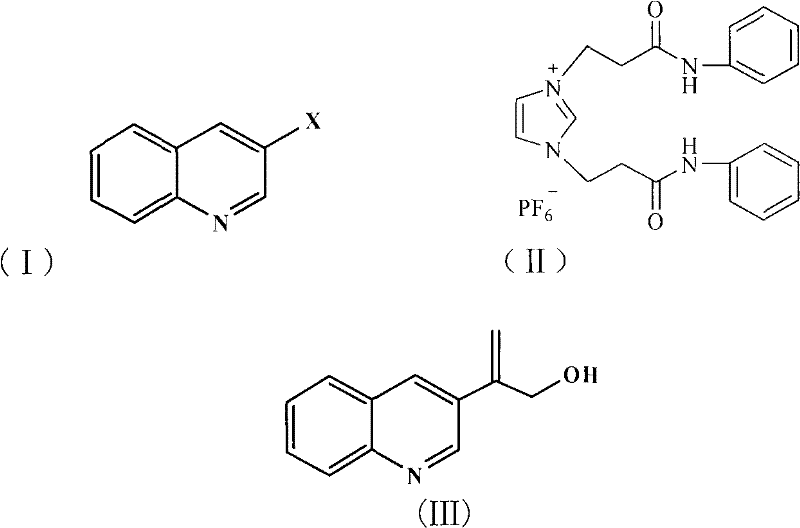
Traditional methodologies for constructing carbon-carbon bonds in quinoline derivatives often rely heavily on palladium-catalyzed cross-coupling reactions that necessitate the use of toxic and expensive organic phosphine ligands. These conventional systems frequently suffer from catalyst deactivation due to the aggregation of palladium species into inactive palladium black, leading to inconsistent batch quality and increased metal contamination in the final product. Furthermore, the separation of homogeneous catalysts from the reaction mixture typically requires complex purification steps, such as extensive chromatography or scavenging treatments, which drastically inflate manufacturing costs and extend lead times. The reliance on volatile organic amines as bases in these legacy processes also introduces significant safety hazards and waste disposal challenges, creating bottlenecks for supply chain managers aiming for sustainable operations. Consequently, there is an urgent industrial demand for alternative catalytic systems that offer superior stability and ease of separation without compromising reaction efficiency.
The novel approach detailed in the patent data addresses these critical pain points by employing a task-specific ionic liquid that functions dually as a reaction medium and a stabilizing ligand for the palladium catalyst. This unique ionic liquid architecture prevents the formation of palladium black, thereby maintaining high catalytic activity throughout the reaction cycle and enabling potential reuse of the catalytic system. The substitution of traditional organic bases with solid magnesium oxide further simplifies the workup procedure, as the solid base can be easily removed by filtration or extraction, eliminating the need for aqueous washes that generate large volumes of saline wastewater. This streamlined process flow significantly reduces the operational complexity and enhances the overall atom economy of the synthesis. By avoiding the use of organic phosphorus compounds entirely, the method offers a cleaner profile that is increasingly demanded by regulatory bodies and downstream pharmaceutical customers focused on impurity control.

Mechanistic Insights into Ionic Liquid-Stabilized Heck Coupling
The mechanistic superiority of this process lies in the specific interaction between the imidazolium-based ionic liquid and the palladium active species. The nitrogen atoms within the imidazolium ring, along with the carbamoyl side chains, provide a coordination environment that stabilizes the palladium center against reduction to metallic palladium. This stabilization effect ensures that the catalytic cycle proceeds efficiently through the oxidative addition, migratory insertion, and beta-hydride elimination steps characteristic of the Heck reaction. The ionic liquid also enhances the solubility of the inorganic base and the organic substrates, creating a pseudo-homogeneous reaction environment that facilitates mass transfer and improves reaction kinetics. For technical teams, understanding this stabilization mechanism is crucial for optimizing reaction parameters such as temperature and catalyst loading to maximize throughput while minimizing palladium leaching.
Impurity control is another critical aspect where this catalytic system excels, primarily due to the suppression of side reactions commonly associated with free palladium species. In traditional systems, palladium black can catalyze non-selective hydrogenation or homocoupling of the starting materials, leading to difficult-to-remove byproducts that compromise the purity of the pharmaceutical intermediate. The constrained environment provided by the ionic liquid matrix restricts the mobility of the palladium complexes, favoring the desired cross-coupling pathway over competing degradation routes. Additionally, the use of magnesium oxide as a mild solid base minimizes the risk of base-mediated hydrolysis or polymerization of the sensitive allyl alcohol moiety. This results in a cleaner crude reaction profile, reducing the burden on downstream purification units and ensuring that the final product meets stringent specifications for heavy metal residues and organic impurities.
How to Synthesize 2-(3'-Quinolyl) Allyl Alcohol Efficiently
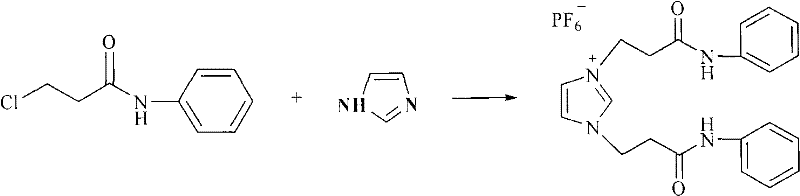
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable intermediate with high reproducibility and yield. The process begins with the precise combination of 3-haloquinoline, the functionalized ionic liquid, allyl alcohol, magnesium oxide, and palladium acetate in a polar aprotic solvent such as DMF. The reaction mixture is then subjected to controlled heating, typically within the range of 100°C to 130°C, for a duration of 10 to 20 hours to ensure complete conversion of the starting material. Following the reaction, the workup involves a straightforward extraction with toluene and drying over anhydrous magnesium sulfate, followed by purification via column chromatography. The detailed standardized synthesis steps for implementing this green technology in a pilot or production setting are provided below.
- Combine 3-haloquinoline, functionalized imidazolium ionic liquid, allyl alcohol, magnesium oxide, palladium acetate, and DMF in a reactor.
- Heat the mixture to 100°C-130°C and stir for 10-20 hours to facilitate the coupling reaction.
- Cool the reaction, extract with toluene, dry over magnesium sulfate, and purify via column chromatography using cyclohexane and acetone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ionic liquid-mediated Heck reaction offers substantial benefits for cost reduction in pharmaceutical intermediate manufacturing. The elimination of expensive and air-sensitive phosphine ligands directly lowers the raw material costs, while the ability to potentially recycle the ionic liquid-catalyst system reduces the consumption of precious palladium metal. This translates into a more predictable cost structure for procurement managers, shielding them from the volatility associated with noble metal markets. Furthermore, the simplified post-treatment process, which avoids complex aqueous workups and extensive neutralization steps, significantly reduces utility consumption and waste disposal fees. These operational efficiencies contribute to a lower total cost of ownership for the manufacturing process, making it an attractive option for large-scale production campaigns.
- Cost Reduction in Manufacturing: The removal of organic phosphine ligands eliminates a major cost driver and safety hazard, as these compounds are often expensive and require special handling protocols. By utilizing a stable ionic liquid system, the process reduces the frequency of catalyst replenishment and minimizes the loss of palladium, which is a significant factor in the overall economics of precious metal catalysis. The use of inexpensive solid magnesium oxide instead of soluble organic amines further drives down reagent costs and simplifies inventory management. Collectively, these factors create a leaner manufacturing process that maximizes resource utilization and minimizes variable production expenses.
- Enhanced Supply Chain Reliability: The robustness of this reaction system, characterized by its tolerance to varying substrate qualities and its resistance to catalyst deactivation, ensures consistent batch-to-batch performance. This reliability is critical for supply chain heads who must guarantee uninterrupted delivery of key intermediates to downstream API manufacturers. The simplified purification workflow reduces the risk of production delays caused by equipment fouling or extended processing times, thereby shortening the overall manufacturing cycle time. Additionally, the reduced dependency on specialized reagents with long lead times enhances the agility of the supply chain, allowing for faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The green nature of this technology, marked by low vapor pressure of the ionic liquid and minimal waste generation, aligns perfectly with increasingly stringent environmental regulations. Scaling this process from laboratory to commercial production does not require massive investments in specialized waste treatment infrastructure, as the effluent load is significantly lower than that of traditional methods. The absence of volatile organic amines improves workplace safety and reduces the need for complex ventilation systems, facilitating easier regulatory approval for new production lines. This environmental compatibility future-proofs the manufacturing asset against evolving sustainability mandates and enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the technology. Understanding these aspects is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the advantages of using ionic liquids in this Heck reaction?
A: The ionic liquid acts as both a solvent and a ligand stabilizer, preventing palladium black formation and allowing for catalyst reuse, which significantly reduces waste and cost compared to traditional phosphine ligands.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes simple solid bases like magnesium oxide and avoids volatile organic amines, making post-treatment easier and safer for large-scale commercial manufacturing.
Q: What is the typical yield for this synthesis method?
A: According to the patent data, optimized conditions using 3-iodoquinoline can achieve yields up to 82%, demonstrating high efficiency for pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3'-Quinolyl) Allyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and sustainable synthetic routes for high-value pharmaceutical intermediates like 2-(3'-quinolyl) allyl alcohol. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and reliable. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to implement green chemistry solutions, such as the ionic liquid-catalyzed Heck reaction, demonstrates our dedication to providing cost-effective and environmentally responsible manufacturing services to our global partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your sourcing strategy and secure a competitive advantage in the marketplace.
