Scalable Synthesis of 4-Aminonaphthalene-2,6-Dicarboxylic Acid via Zinc Reduction
The chemical industry is constantly seeking more efficient pathways for synthesizing critical aromatic intermediates, particularly those used in high-value applications like Metal-Organic Frameworks (MOFs) and pharmaceutical agents. A significant breakthrough in this domain is detailed in patent CN113548975A, which outlines a robust preparation method for 4-aminonaphthalene-2,6-dicarboxylic acid. This compound serves as a pivotal building block, yet its traditional synthesis has been plagued by low yields and hazardous conditions. The disclosed innovation introduces a streamlined two-step sequence that begins with a highly controlled nitration of 2,6-naphthalenedicarboxylic acid, followed by a novel reduction strategy that eschews expensive noble metals. By optimizing stoichiometric ratios and reaction parameters, this methodology not only enhances the overall yield but also drastically improves the safety profile of the manufacturing process. For R&D teams and procurement specialists alike, understanding this shift from catalytic hydrogenation to chemical reduction represents a major opportunity for cost optimization and supply chain resilience in the production of complex fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
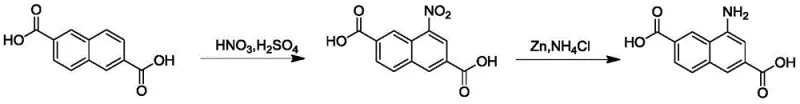
Historically, the synthesis of 4-aminonaphthalene-2,6-dicarboxylic acid has relied heavily on palladium-carbon (Pd/C) catalyzed hydrogenation for the reduction of the nitro group. While effective in laboratory settings, this approach presents substantial hurdles for industrial scale-up. The conventional literature methods often employ an excess of nitric acid, typically around 1.4 equivalents, during the initial nitration step. This surplus leads to the formation of undesirable dinitro byproducts, complicating the purification process and reducing the overall mass balance of the desired mono-nitro intermediate. Furthermore, the reliance on Pd/C necessitates the use of high-pressure hydrogen gas, which requires specialized, costly reactor infrastructure and rigorous safety protocols to mitigate explosion risks. The combination of expensive catalysts, high-pressure equipment, and complex impurity profiles makes the traditional route economically inefficient and operationally rigid for large-scale commercial manufacturing.
The Novel Approach
In stark contrast, the method described in patent CN113548975A introduces a paradigm shift by utilizing zinc powder and ammonium chloride for the reduction step. This chemical reduction system operates under atmospheric pressure and moderate temperatures, completely eliminating the need for high-pressure hydrogenation units. The process begins with a refined nitration step where the nitric acid equivalent is tightly controlled between 1.15 and 1.20, significantly minimizing side reactions. Following isolation, the nitro intermediate is reduced using zinc in a methanol and saturated ammonium chloride solution. This switch not only reduces raw material costs by replacing precious metals with abundant base metals but also simplifies the engineering requirements for the production facility. The result is a safer, more cost-effective pathway that maintains high product purity and yield, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks looking to optimize their portfolios.
Mechanistic Insights into Zinc-Mediated Reduction and Controlled Nitration
The success of this synthetic route lies in the precise mechanistic control exerted during both the electrophilic aromatic substitution and the subsequent electron transfer reduction. In the nitration phase, the use of concentrated sulfuric acid acts as both a solvent and a dehydrating agent, generating the active nitronium ion species. By maintaining the reaction temperature below 10°C during the dropwise addition of dilute nitric acid, the kinetic energy of the system is managed to favor mono-substitution at the 4-position of the naphthalene ring. This thermal control is critical because higher temperatures or excess nitrating agents would increase the probability of a second nitration event, leading to dinitro impurities that are difficult to separate. The subsequent room temperature stirring for 8 to 12 hours ensures complete conversion of the starting material without degrading the sensitive carboxylic acid functionalities, resulting in a white intermediate with purity exceeding 98%.
The reduction mechanism involves the transfer of electrons from the zinc metal to the nitro group, facilitated by the acidic environment provided by the ammonium chloride. Unlike catalytic hydrogenation which relies on surface adsorption, this heterogeneous chemical reduction proceeds through a series of electron and proton transfers that convert the nitro group sequentially into nitroso, hydroxylamine, and finally the amine. The use of methanol as a co-solvent ensures adequate solubility of the organic intermediate while allowing the inorganic zinc salts to remain in solution or be easily filtered. A unique feature of this process is the post-reaction purification step involving dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) at elevated temperatures (120-150°C). This hot filtration step effectively dissolves the target amine while leaving behind insoluble zinc oxides and other inorganic residues, ensuring that the final precipitated product is free from metal contamination and meets stringent purity specifications required for high-purity MOF linker applications.
How to Synthesize 4-Aminonaphthalene-2,6-Dicarboxylic Acid Efficiently
Implementing this synthesis requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent data. The process is divided into two distinct operational phases: the formation of the nitro-intermediate and its subsequent reduction to the amine. Operators must ensure that the nitration is conducted under strict ice-bath conditions to prevent runaway exotherms and byproduct formation. Once the intermediate is isolated and dried, the reduction step can be performed in standard glass-lined or stainless steel reactors without the need for pressure ratings. The following guide outlines the critical operational parameters derived from the patent examples to assist process engineers in scaling this technology.
- Perform controlled nitration of 2,6-naphthalenedicarboxylic acid using dilute nitric acid in concentrated sulfuric acid at low temperatures.
- Isolate the 4-nitro intermediate via precipitation and recrystallization to ensure high purity before reduction.
- Execute reduction using zinc powder and ammonium chloride in methanol, followed by hot solvent purification to remove impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this zinc-mediated synthesis offers profound strategic benefits beyond simple chemistry. The primary advantage is the drastic reduction in raw material costs associated with replacing palladium catalysts with zinc powder. Palladium is a volatile commodity with fluctuating prices, whereas zinc is abundant and inexpensive, leading to significant cost reduction in fine chemical manufacturing. Additionally, the elimination of high-pressure hydrogen gas removes a major safety bottleneck, allowing production to occur in facilities that may not be equipped for hazardous high-pressure operations. This flexibility enhances supply chain reliability by expanding the pool of potential contract manufacturing organizations (CMOs) capable of producing the material, thereby reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of production stoppages due to equipment maintenance or safety audits.
- Cost Reduction in Manufacturing: The substitution of noble metal catalysts with base metals fundamentally alters the cost structure of the synthesis. By removing the need for palladium on carbon, manufacturers avoid not only the high upfront cost of the catalyst but also the expenses related to its recovery, recycling, or disposal. Furthermore, the simplified equipment requirements mean lower capital depreciation costs per kilogram of product. The process also demonstrates higher overall yields compared to prior art, meaning less starting material is wasted, which directly translates to improved margin potential for buyers sourcing this key building block for electronic materials or drug discovery programs.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized reagents or complex logistics. This method utilizes commodity chemicals like sulfuric acid, nitric acid, methanol, and zinc powder, all of which are readily available in global markets with stable supply lines. The robustness of the reaction conditions, which tolerate slight variations better than sensitive catalytic hydrogenations, ensures consistent batch-to-batch quality. This reliability is crucial for long-term contracts where delivery consistency is paramount, allowing partners to secure a steady flow of materials for the commercial scale-up of complex polymer additives or advanced functional materials without fear of sudden supply disruptions.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a cleaner profile. The avoidance of high-pressure hydrogen reduces the risk profile of the plant, potentially lowering insurance premiums and regulatory scrutiny. While zinc waste must be managed, it is generally easier to handle and treat than spent noble metal catalysts laden with organic residues. The high purity achieved through the hot solvent wash reduces the need for extensive chromatographic purification, which in turn minimizes solvent waste generation. These factors collectively support a more sustainable manufacturing footprint, aligning with the increasing demand for green chemistry practices in the production of specialty chemicals and agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs.
Q: Why is zinc powder preferred over palladium carbon for this reduction?
A: Zinc powder combined with ammonium chloride eliminates the need for expensive noble metal catalysts and high-pressure hydrogenation equipment, significantly lowering capital expenditure and operational risks while maintaining comparable yields.
Q: How does the new nitration protocol improve product purity?
A: By strictly controlling the nitric acid equivalent to 1.15-1.20 and maintaining reaction temperatures below 10°C during addition, the formation of dinitro byproducts is effectively suppressed, simplifying downstream purification.
Q: What are the scalability advantages of this synthetic route?
A: The process utilizes standard atmospheric pressure reactors and common reagents like zinc and methanol, avoiding the safety complexities associated with high-pressure hydrogen gas, thus facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminonaphthalene-2,6-Dicarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis routes play in the competitiveness of the global chemical market. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN113548975A and is fully prepared to translate this laboratory-scale innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with the necessary corrosion-resistant reactors and filtration systems to handle the acidic and thermal conditions of this specific process, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch to meet international standards.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cost-effective technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Whether you require this intermediate for MOF research, dye synthesis, or pharmaceutical development, our commitment to quality and efficiency ensures that we are the ideal partner for your long-term growth and innovation goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
