Advanced Synthesis of Aripiprazole Intermediates Using Novel Salting-Out Purification Technology
Advanced Synthesis of Aripiprazole Intermediates Using Novel Salting-Out Purification Technology
The pharmaceutical industry continuously seeks robust synthetic routes for critical antipsychotic agents, and recent advancements documented in patent CN115368317A highlight a significant breakthrough in the production of aripiprazole intermediates. This intellectual property introduces a refined methodology for synthesizing 1-(2,3-dichlorophenyl)piperazine hydrochloride, a pivotal building block in the manufacture of this atypical antipsychotic medication. The core innovation lies in the strategic identification and elimination of a specific dimer impurity, designated as compound (I), which has historically plagued the quality profile of this intermediate. By redefining the purification landscape through a combination of aqueous dissolution and inorganic salt salting-out, this technology offers a pathway to achieve purity levels exceeding 99.6% while simultaneously streamlining operational complexity. For R&D directors and process chemists, understanding the structural nuances of the final drug is essential, as shown in the molecular architecture below.
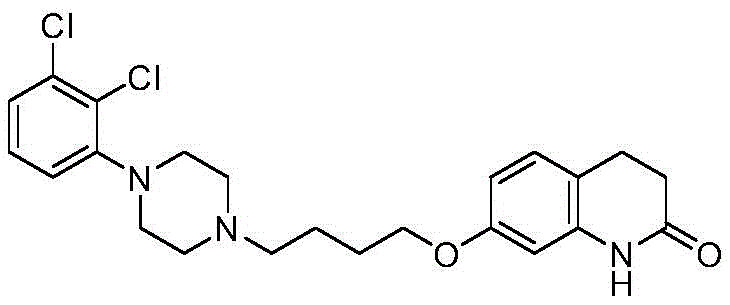
This structural clarity is not merely academic; it serves as the foundation for developing reliable analytical methods that ensure batch-to-batch consistency. The ability to produce high-purity pharmaceutical intermediates is a non-negotiable requirement for regulatory compliance and patient safety, making this patented approach a valuable asset for any organization aiming to secure a stable supply chain for mental health therapeutics. The transition from traditional purification techniques to this novel salting-out protocol represents a paradigm shift in how we approach impurity management in heterocyclic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of 1-(2,3-dichlorophenyl)piperazine hydrochloride has relied heavily on iterative recrystallization processes using organic solvents such as absolute ethanol. As detailed in the comparative examples of the patent data, these conventional routes often necessitate multiple cycles of dissolution and precipitation to achieve acceptable purity standards, yet they frequently fail to adequately suppress the formation of the persistent dimer impurity. In standard operations, even after rigorous recrystallization, impurity levels can remain as high as 3.39%, which poses significant risks for downstream coupling reactions and final drug substance quality. Furthermore, the reliance on large volumes of organic solvents creates substantial logistical burdens regarding solvent recovery, waste disposal, and operational safety, particularly when scaling from laboratory benchtops to multi-ton commercial production facilities. The yield penalties associated with these aggressive purification steps are also notable, with conventional methods often struggling to exceed yields of 58%, thereby inflating the cost of goods sold and reducing overall process efficiency.
The Novel Approach
In stark contrast, the innovative process described in CN115368317A replaces the cumbersome organic recrystallization regimen with a highly efficient water-based purification strategy coupled with sodium chloride salting-out. This method capitalizes on the distinct physicochemical properties of the target molecule versus the dimer contaminant, allowing for a dramatic simplification of the workflow. The reaction scheme illustrates the fundamental transformation where 2,3-dichloroaniline reacts with bis-(2-chloroethyl)amine hydrochloride to form the piperazine ring system.
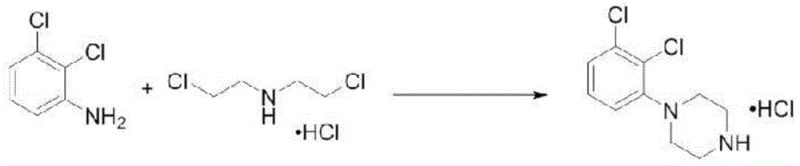
Following the initial cyclization at elevated temperatures, the crude product is subjected to a selective aqueous wash that effectively solubilizes the desired hydrochloride salt while leaving the hydrophobic dimer impurity behind or allowing it to be filtered out prior to the final crystallization step. By introducing a saturated sodium chloride solution at controlled low temperatures (0-10°C), the solubility of the target intermediate is suppressed, inducing rapid and high-yield crystallization. This approach not only boosts yields to approximately 70% but also drives impurity levels down to negligible amounts around 0.20%, demonstrating a clear superiority over legacy techniques. For procurement managers, this translates to a more predictable and cost-effective manufacturing process that minimizes raw material waste and maximizes output per batch.
Mechanistic Insights into Impurity Control and Salting-Out Crystallization
The success of this improved synthesis route hinges on a deep mechanistic understanding of the side reactions that generate the dimer impurity, structurally defined as Formula (I) in the patent documentation. This dimer arises from the intermolecular reaction of the chloroethyl amine species, creating a larger, more lipophilic molecule that co-precipitates with the desired product under standard organic crystallization conditions. The visual representation of this impurity highlights its extended carbon chain and dual aromatic systems, which contribute to its poor solubility in polar aqueous media compared to the mono-substituted piperazine target.
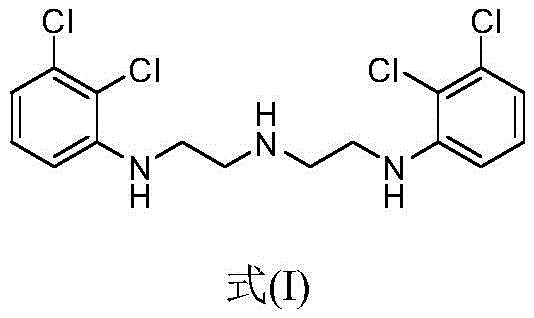
By elucidating this structure, process chemists can rationally design a purification protocol that exploits solubility differentials. Experimental data confirms that while 1-(2,3-dichlorophenyl)piperazine hydrochloride exhibits a solubility of 227mg/ml in water at 25°C, the dimer impurity is virtually insoluble, with a solubility of less than 0.01mg/ml. This massive disparity allows the aqueous dissolution step to act as a highly selective filter, effectively partitioning the product into the liquid phase while the impurity remains in the solid residue or is removed during the initial filtration of the crude cake. Subsequent addition of sodium chloride increases the ionic strength of the solution, reducing the dielectric constant of the solvent environment and forcing the organic salt out of the solution in a highly pure crystalline form. This mechanism ensures that the final product is substantially free of the dimer, providing a robust safeguard for the quality of the final aripiprazole API.
How to Synthesize 1-(2,3-Dichlorophenyl)piperazine Hydrochloride Efficiently
Implementing this advanced synthesis protocol requires precise control over reaction parameters and purification conditions to fully realize the benefits of impurity reduction and yield enhancement. The process begins with the careful charging of reactants in specific weight ratios, followed by a prolonged heating phase to ensure complete cyclization before initiating the critical cooling and precipitation sequence. Operators must adhere strictly to the temperature gradients specified, particularly the two-stage cooling process which optimizes the physical form of the crude precipitate for subsequent washing. The transition to the aqueous workup phase marks the divergence from traditional methods, requiring attention to the volume of water used for dissolution to ensure complete solubilization of the product without excessive dilution. Finally, the salting-out step demands accurate temperature maintenance between 0°C and 10°C to maximize crystal recovery and purity. For a detailed breakdown of the standardized operating procedures and specific equipment requirements, please refer to the technical guide below.
- React 2,3-dichloroaniline with bis-(2-chloroethyl)amine hydrochloride in NMP at 135°C for 29 hours, then cool and precipitate crude product with ethanol.
- Dissolve the crude filter cake in water at ambient temperature or up to 50°C to separate soluble product from insoluble dimer impurities.
- Add saturated sodium chloride solution to the filtrate at 0-10°C to induce salting-out crystallization, yielding high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic viability and continuity of pharmaceutical supply chains, the adoption of this salting-out technology presents compelling advantages that extend beyond mere chemical elegance. The shift away from multiple organic recrystallizations fundamentally alters the cost structure of manufacturing this key intermediate, offering tangible benefits in terms of operational expenditure and resource allocation. By eliminating the need for repeated solvent exchanges and the associated energy costs of evaporating large volumes of ethanol, manufacturers can achieve significant reductions in utility consumption and waste treatment fees. This process intensification allows for faster batch cycle times, as the filtration and salting-out steps are inherently quicker and easier to manage than complex recrystallization protocols, thereby increasing the overall throughput of the production facility without requiring additional capital investment in reactor capacity.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in organic solvent usage, specifically absolute ethanol, which is replaced largely by water and inexpensive inorganic salts like sodium chloride. Eliminating the need for multiple recrystallization steps means that less solvent needs to be purchased, stored, recovered, or disposed of, leading to substantial savings in raw material costs. Furthermore, the improvement in yield from approximately 58% in conventional methods to 70% in this novel process directly lowers the cost per kilogram of the active intermediate, providing a competitive edge in pricing negotiations with downstream API manufacturers. The simplified operational workflow also reduces labor hours and minimizes the risk of batch failures due to operator error during complex purification sequences.
- Enhanced Supply Chain Reliability: Dependence on complex organic solvent supply chains can introduce vulnerabilities, especially during periods of market volatility or logistical disruptions. By transitioning to a water-based purification system that utilizes commodity chemicals like sodium chloride, the manufacturing process becomes more resilient and less susceptible to raw material shortages. The robustness of the impurity control mechanism ensures consistent product quality, reducing the likelihood of batch rejections or delays caused by failing to meet stringent purity specifications. This reliability is crucial for maintaining uninterrupted supply to pharmaceutical clients who require just-in-time delivery of critical intermediates to keep their own API production schedules on track.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, reducing the inventory of flammable organic solvents on-site significantly lowers the fire hazard rating of the facility and simplifies regulatory compliance. The decrease in volatile organic compound (VOC) emissions aligns with increasingly strict global environmental regulations, positioning the manufacturer as a sustainable partner for green chemistry initiatives. The simplicity of the salting-out process makes it highly scalable, allowing for seamless technology transfer from pilot plants to multi-ton commercial reactors without the engineering challenges often associated with managing large volumes of hot organic solvents. This scalability ensures that the supply can easily be ramped up to meet surging market demand for aripiprazole without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, drawing directly from the experimental data and beneficial effects outlined in the intellectual property documentation. Understanding these details is vital for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement officers assessing the quality assurances provided by suppliers utilizing this technology. The answers provided reflect the specific advantages of the salting-out technique over traditional recrystallization, focusing on impurity profiles, solvent reduction, and process robustness.
Q: How does the new salting-out method improve impurity control compared to traditional recrystallization?
A: The novel method leverages the significant solubility difference between the target intermediate and the dimer impurity in water. While the product is highly soluble (227mg/ml), the dimer impurity is virtually insoluble (<0.01mg/ml), allowing for effective removal via filtration and subsequent salting-out without multiple organic solvent recrystallizations.
Q: What are the specific reaction conditions required to minimize dimer formation?
A: The process utilizes a precise weight ratio of 2,3-dichloroaniline to NMP to bis-(2-chloroethyl)amine hydrochloride of 90:450:144. The reaction is maintained at 135°C for 29 hours, followed by a controlled two-stage cooling process (first to 75-80°C, then to 0-5°C with ethanol) to optimize crude precipitation before purification.
Q: Does this process offer environmental benefits for large-scale manufacturing?
A: Yes, by replacing multiple rounds of organic solvent recrystallization with a water-based dissolution and inorganic salt salting-out step, the process drastically reduces the consumption of volatile organic compounds like absolute ethanol, simplifying waste treatment and lowering the overall environmental footprint.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2,3-Dichlorophenyl)piperazine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex pharmaceutical intermediates requires not only chemical expertise but also a commitment to scalable and compliant manufacturing practices. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We understand the critical importance of maintaining stringent purity specifications for intermediates like 1-(2,3-dichlorophenyl)piperazine hydrochloride, and our rigorous QC labs are equipped to detect and quantify trace impurities down to the ppm level, guaranteeing that every batch meets the highest international standards. By leveraging advanced purification technologies such as the salting-out method described in CN115368317A, we deliver products that facilitate smoother downstream processing for our global partners.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project requirements. Whether you are looking to reduce lead time for high-purity pharmaceutical intermediates or seeking a long-term strategic partner for your supply chain, we are prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical prowess can drive value and efficiency in your aripiprazole production strategy.
