Scalable Synthesis of High-Purity Metal Difluorophosphate for Next-Generation Lithium-Ion Batteries
The rapid expansion of the global electric vehicle market and the increasing demand for high-energy-density energy storage systems have placed unprecedented pressure on the supply chain of critical battery components. Among these, electrolyte additives play a pivotal role in enhancing the thermal stability, cycle life, and low-temperature performance of lithium-ion batteries. Patent CN113004322A introduces a groundbreaking preparation method for metal difluorophosphate salts, a class of compounds increasingly recognized for their ability to form stable solid-electrolyte interphases (SEI) on electrode surfaces. This technology addresses the long-standing challenges of traditional synthesis routes, offering a pathway to high-purity products through a mild, solution-phase fluorination process. By shifting away from hazardous high-temperature solid-state reactions, this innovation provides a robust foundation for the commercial scale-up of complex electronic chemicals, ensuring a reliable supply of next-generation battery materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of metal difluorophosphates, such as lithium difluorophosphate (LiPO2F2), has been plagued by severe process inefficiencies and safety hazards. Traditional methods often rely on the reaction of phosphorus pentoxide with lithium fluoride at extreme temperatures exceeding 350°C. These solid-phase reactions are not only energy-intensive but also suffer from poor heat transfer and mixing, leading to incomplete conversions and the formation of stubborn by-products like lithium phosphate. Furthermore, alternative routes involving difluorophosphoric anhydride or hexafluorophosphate precursors frequently require the handling of highly corrosive hydrogen fluoride gas or generate toxic silicon tetrafluoride as a waste stream. These factors create significant barriers to entry for manufacturers, complicating environmental compliance and driving up the operational costs associated with waste treatment and specialized equipment maintenance.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated two-step solution-phase strategy that operates under significantly milder conditions. The process initiates with the nucleophilic fluorination of dihalophosphate esters using safe, manageable fluorinating agents like potassium fluoride or ammonium fluoride in the presence of phase transfer catalysts. This eliminates the need for gaseous HF and allows the reaction to proceed at temperatures between 50°C and 60°C. The subsequent metathesis reaction with metal salts occurs in dry organic solvents, facilitating easy separation of the product from inorganic salts. This approach not only enhances the safety profile of the manufacturing facility but also dramatically improves the purity of the final electrolyte additive by avoiding the thermal decomposition pathways inherent in high-temperature processes.
Mechanistic Insights into Catalytic Fluorination and Salt Metathesis
The core of this technological breakthrough lies in the efficient halogen-fluorine exchange mechanism facilitated by quaternary ammonium or phosphonium salts. In the first stage, the dihalophosphate precursor, characterized by a central phosphorus atom bonded to halogen leaving groups, undergoes nucleophilic attack by fluoride ions. The phase transfer catalyst plays a critical role by solubilizing the inorganic fluoride source in the organic medium, thereby increasing the effective concentration of reactive fluoride species near the organic substrate. This catalytic cycle ensures that the substitution of chlorine or bromine atoms with fluorine proceeds to completion without requiring excessive thermal energy. The structural integrity of the phosphate backbone is maintained throughout this transformation, preserving the oxidation state of the phosphorus center which is crucial for the electrochemical stability of the final additive.
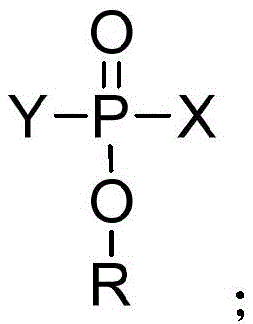
Following the formation of the difluorophosphate intermediate, the process transitions to a salt metathesis reaction where the organic or acidic proton is exchanged for a metal cation. This step is meticulously controlled by maintaining anhydrous conditions to prevent hydrolysis of the sensitive P-F bonds. The choice of solvent, ranging from acetonitrile to glycol ethers, is optimized to dissolve both the organic intermediate and the inorganic metal salt while allowing the final metal difluorophosphate product to precipitate or crystallize out upon cooling. This solubility differential is exploited during the purification stage, where recrystallization at sub-zero temperatures effectively excludes impurities such as unreacted metal halides or partially fluorinated species. The result is a highly crystalline product with a well-defined lattice structure, essential for consistent dissolution rates in battery electrolyte formulations.
How to Synthesize Metal Difluorophosphate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of moisture control and temperature regulation. The procedure begins with the activation of the fluorinating agent in a dry non-aqueous solvent, followed by the controlled addition of the dihalophosphate precursor. Operators must maintain strict inert atmosphere conditions, typically using nitrogen protection, to safeguard the reactive intermediates from atmospheric moisture which could lead to hydrolysis and acid formation. After the fluorination is complete, the reaction mixture is filtered to remove catalyst residues before concentrating the filtrate to isolate the oily difluorophosphate intermediate. The detailed standardized synthesis steps, including specific molar ratios and stirring rates required for optimal yield, are provided in the technical guide below.
- Fluorinate dihalophosphate precursors using a fluorinating agent and phase transfer catalyst in a non-aqueous solvent under mild heating.
- React the resulting difluorophosphate intermediate with a specific metal salt (e.g., Lithium, Sodium, Magnesium) in a dry organic solvent.
- Purify the crude metal difluorophosphate product through recrystallization in fresh organic solvents at low temperatures to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical superiority. The shift from hazardous gas-phase reagents to solid or liquid fluorinating agents significantly de-risks the supply chain, reducing reliance on specialized containment infrastructure and lowering insurance and compliance costs. The mild reaction conditions allow for the use of standard glass-lined or stainless-steel reactors rather than exotic alloys resistant to high-temperature corrosion, representing a substantial reduction in capital expenditure for new production lines. Furthermore, the high atom economy of the reaction means that raw material utilization is maximized, directly translating to lower variable costs per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of high-temperature calcination steps and the avoidance of expensive anhydride precursors lead to a drastic simplification of the energy profile. By operating at near-ambient pressures and moderate temperatures, the process reduces the load on HVAC and cooling systems, resulting in significant operational savings. Additionally, the ability to recycle the organic solvents used in the recrystallization steps further enhances the economic viability of the process, minimizing waste disposal fees and raw material procurement volumes.
- Enhanced Supply Chain Reliability: The versatility of the method regarding metal salts allows manufacturers to pivot quickly between producing lithium, sodium, or other metal difluorophosphates based on market demand without retooling the entire reactor train. This flexibility is crucial in a volatile market where battery chemistries are rapidly evolving. Moreover, the use of commercially available and stable starting materials ensures that production schedules are not disrupted by the scarcity of niche reagents, guaranteeing consistent lead times for downstream battery cell manufacturers.
- Scalability and Environmental Compliance: The solution-phase nature of the reaction facilitates straightforward scale-up from kilogram to tonne quantities, as heat and mass transfer issues are far easier to manage in liquids than in solids. From an environmental perspective, the process generates minimal hazardous waste; the absence of silicon tetrafluoride gas and the reduction of acidic off-gases simplify the scrubbing requirements. This aligns perfectly with the increasingly stringent environmental regulations governing chemical manufacturing in Europe and North America, ensuring long-term operational continuity without regulatory friction.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this material into their supply chains, we have compiled answers to common inquiries regarding the specification and handling of these electrolyte additives. The following responses are derived directly from the experimental data and process parameters detailed in the patent literature, providing a transparent view of the material's capabilities and limitations. Understanding these technical nuances is essential for quality assurance teams when defining incoming inspection criteria and storage protocols.
Q: What are the key advantages of this fluorination method over traditional solid-state synthesis?
A: Unlike traditional solid-phase methods requiring temperatures as high as 350°C which generate significant by-products like lithium phosphate, this solution-phase method operates at mild temperatures (50-60°C). This drastically reduces energy consumption, minimizes hazardous by-product formation such as silicon tetrafluoride, and simplifies the purification process, leading to higher overall atom economy and product purity.
Q: Which metal cations can be utilized in this synthesis protocol?
A: The process is highly versatile and supports a wide range of metal species including Lithium (Li), Sodium (Na), Potassium (K), Magnesium (Mg), Aluminum (Al), and transition metals like Zinc (Zn) or Copper (Cu). This flexibility allows manufacturers to tailor electrolyte additives for specific battery chemistries beyond standard lithium-ion systems, such as sodium-ion or magnesium-ion batteries.
Q: How does this method address impurity control in electrolyte additives?
A: The method utilizes a specific recrystallization step at low temperatures (-25°C to 0°C) using dried organic solvents like tetrahydrofuran or acetonitrile. This effectively removes unreacted metal salts and organic by-products. Furthermore, the use of phase transfer catalysts ensures complete conversion of the dihalophosphate precursor, reducing the presence of partially fluorinated impurities that can degrade battery performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metal Difluorophosphate Supplier
As the global demand for high-performance energy storage solutions continues to surge, the need for a dependable partner capable of delivering ultra-high-purity electrolyte additives has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN113004322A to produce metal difluorophosphates that meet the rigorous standards of top-tier battery manufacturers. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our stringent purity specifications and rigorous QC labs guarantee that every batch delivered is free from moisture and metallic impurities that could compromise battery safety.
We invite you to collaborate with us to optimize your electrolyte formulation and secure a stable supply of this critical component. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our efficient synthesis route can lower your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you power the next generation of energy storage technology.
