Advanced Oxiracetam Manufacturing: A Novel Synthetic Route for High-Purity Pharmaceutical Intermediates
Advanced Oxiracetam Manufacturing: A Novel Synthetic Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust, scalable, and safe methodologies for producing cognitive enhancers, and the technical disclosure within patent CN101914052B represents a significant leap forward in the manufacturing of oxiracetam. This specific intellectual property outlines a refined synthetic pathway that begins with the inexpensive and accessible raw material, 3-chloro-2-hydroxy propionitrile, effectively bypassing the hazardous and convoluted steps associated with legacy production methods. By shifting the synthetic foundation to a nitrile-based precursor, the process not only mitigates severe safety risks but also streamlines the purification workflow, resulting in a final active pharmaceutical ingredient with exceptional clarity and structural integrity. For procurement specialists and R&D directors alike, this patent offers a blueprint for reducing the total cost of ownership while simultaneously elevating the quality standards of the supply chain. The strategic implementation of this technology allows manufacturers to navigate the complex regulatory landscape of nootropic agents with greater confidence and efficiency.
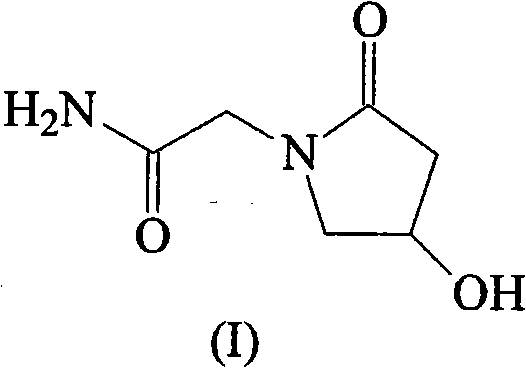
As illustrated in the structural representation, the target molecule possesses a specific stereochemical arrangement that is critical for its biological activity, and the new method ensures this architecture is preserved with minimal impurity formation. The transition from traditional batch processes to this optimized continuous-flow compatible chemistry marks a pivotal moment for suppliers aiming to secure long-term contracts with major multinational pharmaceutical corporations. Furthermore, the elimination of heavy metal catalysts and explosive reagents aligns perfectly with modern green chemistry initiatives, reducing the environmental footprint of the manufacturing facility. This holistic improvement in process design underscores the importance of adopting next-generation synthetic strategies to maintain competitiveness in the global market for pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxiracetam has been plagued by methodologies that are inherently dangerous, economically inefficient, and technically cumbersome, creating substantial bottlenecks for reliable oxiracetam supplier operations. Early approaches, such as those utilizing sodium azide for ring closure, introduced extreme safety hazards due to the explosive nature of azide salts, necessitating expensive containment infrastructure and rigorous safety protocols that drove up operational costs significantly. Other legacy routes relied on complex multi-step sequences involving ketene dimers or chiral reagents that were not only prohibitively expensive but also resulted in low overall yields due to cumulative losses at each stage. These traditional pathways often generated difficult-to-remove byproducts, leading to a final product purity that struggled to meet the stringent specifications required for human consumption without extensive and wasteful recrystallization efforts. Additionally, the reliance on scarce or highly specialized starting materials created supply chain vulnerabilities, where any disruption in the availability of a single precursor could halt entire production lines. The cumulative effect of these drawbacks was a manufacturing process that was fragile, costly, and environmentally taxing, failing to meet the demands of modern high-volume pharmaceutical production.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in the patent leverages a straightforward and chemically elegant pathway that transforms 3-chloro-2-hydroxy propionitrile into the desired pyrrolidone scaffold with remarkable efficiency. This method replaces hazardous azide chemistry with a controlled acid-catalyzed hydrolysis and subsequent base-mediated cyclization, drastically simplifying the reaction engineering requirements and enhancing workplace safety. By utilizing common industrial solvents like isopropyl ether and ethanol, the process avoids the need for exotic or toxic reaction media, thereby facilitating easier solvent recovery and recycling which contributes to substantial cost savings. The step-wise progression from nitrile to amide, then to the cyclic lactam, and finally to the acetamide derivative ensures that impurities are minimized at every juncture, allowing for a much cleaner final isolation. This streamlined logic not only accelerates the time-to-market for the finished API but also provides a robust framework for cost reduction in pharmaceutical intermediates manufacturing by maximizing atom economy and minimizing waste disposal fees. Ultimately, this approach represents a paradigm shift towards sustainable and economically viable production of high-value nootropic compounds.
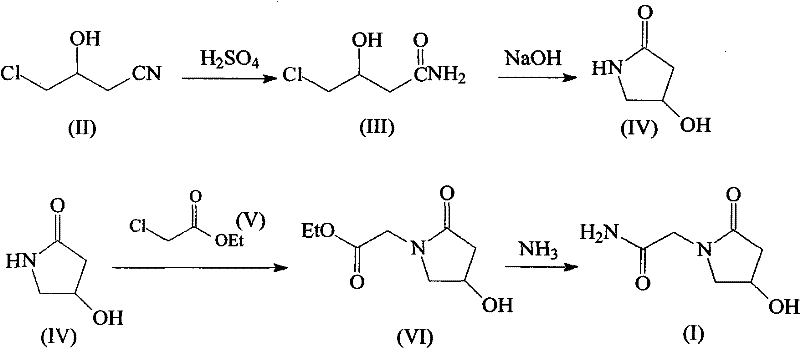
Mechanistic Insights into Nitrile Hydrolysis and Cyclization
The core of this innovative synthesis lies in the precise manipulation of functional groups through a sequence of hydrolysis, cyclization, alkylation, and ammonolysis reactions that are meticulously optimized for high conversion rates. The initial step involves the reaction of 3-chloro-2-hydroxy propionitrile with sulfuric acid under heated conditions, which facilitates the hydration of the nitrile group to form the corresponding primary amide, 4-chloro-3-hydroxy-butyramide, without compromising the integrity of the chlorine leaving group. Following this, the introduction of sodium hydroxide triggers an intramolecular nucleophilic substitution where the amide nitrogen attacks the carbon bearing the chlorine atom, closing the five-membered pyrrolidone ring to form 4-hydroxy-2-pyrrolidone. This cyclization is critical as it establishes the core heterocyclic structure of the molecule, and the use of mild basic conditions ensures that the hydroxyl group remains intact for subsequent functionalization. The mechanistic elegance of this sequence avoids the formation of polymeric byproducts often seen in harsher cyclization conditions, thereby preserving the yield and purity of the intermediate.
Subsequent functionalization is achieved through the alkylation of the pyrrolidone nitrogen with ethyl chloroacetate in the presence of sodium methylate, which acts as a strong base to deprotonate the amide nitrogen and generate a reactive nucleophile. This step installs the acetamide side chain essential for the biological activity of oxiracetam, and the choice of isopropyl ether as a solvent helps to drive the reaction to completion while suppressing side reactions. The final transformation involves the ammonolysis of the ethyl ester group using liquefied ammonia in an autoclave, a high-pressure environment that ensures the complete conversion of the ester to the primary amide without hydrolyzing the lactam ring. Throughout this entire cascade, the control of reaction parameters such as temperature and pressure is paramount to preventing the degradation of the sensitive hydroxyl group, ensuring that the final product retains the necessary physicochemical properties for therapeutic efficacy. This deep understanding of the reaction mechanism allows for precise troubleshooting and optimization during scale-up, guaranteeing consistent quality across different production batches.
How to Synthesize Oxiracetam Efficiently
The practical execution of this synthesis requires careful attention to reaction conditions and workup procedures to maximize the yield and purity of the final crystalline product. The process begins with the preparation of the chloro-hydroxy amide intermediate, followed by its cyclization to the pyrrolidone core, which serves as the pivotal building block for the entire molecule. Detailed standardized operating procedures for each of the four critical stages, including specific molar ratios, temperature profiles, and isolation techniques, are essential for replicating the high success rates reported in the patent literature. Operators must ensure strict adherence to safety protocols, particularly during the ammonolysis step involving pressurized ammonia, to maintain a safe working environment while achieving optimal conversion.
- React 3-chloro-2-hydroxy propionitrile with sulfuric acid under heating to synthesize 4-chloro-3-hydroxy-butyramide.
- Perform a condensation reaction on the butyramide intermediate using sodium hydroxide to form 4-hydroxy-2-pyrrolidone.
- Alkylate the pyrrolidone with ethyl chloroacetate in the presence of sodium methylate to produce the ethyl acetate intermediate.
- Conduct ammonolysis with liquefied ammonia in an autoclave to finalize the synthesis of oxiracetam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers a compelling value proposition centered around risk mitigation, cost efficiency, and supply continuity. By transitioning away from hazardous reagents like sodium azide, companies can significantly reduce their insurance premiums and regulatory compliance burdens, leading to a more stable and predictable operational expenditure profile. The use of commodity chemicals as starting materials ensures that the supply chain is resilient against market volatility, as these precursors are widely available from multiple global vendors, reducing the risk of single-source dependency. Furthermore, the simplified purification process reduces the consumption of solvents and energy, directly translating to lower variable costs per kilogram of produced API. This efficiency gain allows manufacturers to offer more competitive pricing to their clients while maintaining healthy profit margins, a crucial factor in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the reduction in the number of purification steps drastically lowers the direct material and processing costs associated with production. By avoiding the need for specialized equipment to handle explosive materials, capital expenditure requirements are also minimized, allowing for faster ROI on manufacturing assets. The high yield of each individual step cumulatively results in a superior overall process yield, meaning less raw material is wasted to generate the same amount of finished product. These factors combine to create a lean manufacturing model that is highly resistant to inflationary pressures on raw material costs.
- Enhanced Supply Chain Reliability: Sourcing 3-chloro-2-hydroxy propionitrile is far more straightforward than procuring specialized azide salts or ketene dimers, ensuring that production schedules can be maintained without interruption. The robustness of the chemical process means that batch failures are rare, providing customers with a dependable source of high-quality intermediates that they can rely on for their own formulation timelines. This reliability fosters stronger long-term partnerships between suppliers and pharmaceutical companies, as trust is built on consistent delivery performance and product quality. Additionally, the scalability of the process means that supply can be rapidly ramped up to meet surges in demand without the need for complex process re-engineering.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, simplifying the disposal process and reducing the environmental impact of the manufacturing facility. The use of recyclable solvents like ethanol and isopropyl ether aligns with green chemistry principles, helping companies meet their sustainability goals and corporate social responsibility targets. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors, making the technology easily transferable to existing manufacturing sites without major infrastructure upgrades. This ease of scale-up ensures that the technology remains viable from pilot plant quantities all the way to multi-ton commercial production runs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel oxiracetam synthesis method, providing clarity for stakeholders evaluating its potential. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is critical for assessing the feasibility of integrating this route into an existing production portfolio or for qualifying a new supplier for critical raw materials.
Q: What are the primary advantages of the nitrile-based route over traditional azide methods?
A: The nitrile-based route described in CN101914052B eliminates the use of hazardous sodium azide, significantly improving operational safety and reducing the need for specialized waste treatment facilities compared to older azide reduction methods.
Q: How does this method address the issue of low product purity found in prior art?
A: By utilizing a streamlined four-step sequence with specific recrystallization protocols using isopropanol and ethanol, this method achieves a final product purity of 99.9%, overcoming the difficult purification challenges associated with previous complex routes.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials like 3-chloro-2-hydroxy propionitrile and standard reaction conditions (50-60°C), making it highly scalable and cost-effective for industrial manufacturing without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN101914052B and have integrated these advanced principles into our own state-of-the-art manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with the latest analytical instrumentation to guarantee that every batch of oxiracetam meets or exceeds international pharmacopeial standards. Our commitment to quality is matched only by our dedication to safety and environmental stewardship, making us a preferred partner for companies seeking sustainable and compliant supply chain solutions.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can deliver a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By collaborating with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of our advanced synthetic approach. Let us help you secure a stable, high-quality supply of oxiracetam that empowers your drug development programs and strengthens your market position. Contact us today to initiate a dialogue about your upcoming projects and discover the NINGBO INNO PHARMCHEM advantage.
