Advanced Grignard Alkylation Strategy for Commercial Scale-up of 4-Octyl Phenethyl Alcohol
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for complex intermediates, and the preparation of 4-octyl phenethyl alcohol (CAS No.: 162358-05-6) represents a critical challenge in this domain. Patent CN111978154A introduces a transformative preparation method that leverages Grignard chemistry to overcome the historical limitations of yield and cost associated with this valuable compound. This technical disclosure outlines a sophisticated two-step process involving the synthesis of an octyl magnesium halide reagent followed by a strategic alkylation reaction, optionally utilizing protecting group chemistry to maximize purity. For R&D directors and procurement specialists, this patent signifies a shift away from expensive, multi-step precursor routes toward a more direct, atom-economical synthesis that aligns with modern green chemistry principles. The ability to produce high-purity 4-octyl phenethyl alcohol efficiently is paramount for downstream applications in fragrance, pharmaceutical, and specialty chemical sectors, making this intellectual property a cornerstone for reliable supply chain strategies.
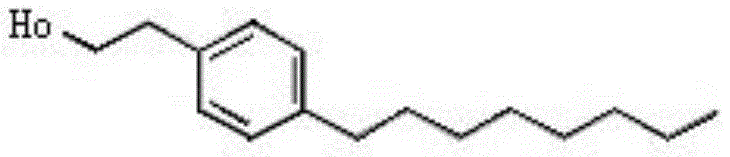
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN111978154A, the industrial synthesis of 4-octyl phenethyl alcohol was plagued by significant economic and technical inefficiencies that hindered large-scale adoption. Traditional methods often relied on the one-step synthesis from 4-octylphenylethanol acetate, which, while boasting a theoretical yield of 95%, suffered from exorbitantly high raw material costs and a convoluted synthesis of the acetate precursor itself. Alternative routes involving the reaction of n-octyl magnesium bromide with 4-(2-acetoxy ethyl) phenol trifluoromethanesulfonate presented another set of challenges, including relatively low synthesis yields and the necessity for high reaction temperatures that complicated industrial control. Furthermore, these legacy processes generated substantial quantities of wastewater and exhaust gas, creating severe environmental compliance burdens and increasing the total cost of ownership for manufacturers. The reliance on specialized, expensive leaving groups like triflates further exacerbated the cost structure, making the final product less competitive in price-sensitive markets.
The Novel Approach
The novel approach disclosed in the patent fundamentally reengineers the synthetic pathway by optimizing the Grignard alkylation step to achieve superior efficiency and environmental performance. By synthesizing the Grignard reagent R1-Mg-X1 (where R1 is octyl and X1 is a halogen) in situ using anhydrous ether or tetrahydrofuran (THF), the process ensures high reactivity of the nucleophile. This reagent is then coupled with a phenethyl alcohol derivative (R2-CH2-X2) under controlled thermal conditions, typically between 110°C and 120°C, to effect the carbon-carbon bond formation. Crucially, the method allows for the incorporation of protecting groups, such as trityl, acetyl, or silyl ethers, which safeguard the hydroxyl functionality during the aggressive alkylation phase. This strategic modification not only minimizes by-product formation but also facilitates easier purification, resulting in a cleaner crude product profile. The transition to this methodology represents a significant leap forward in cost reduction in fine chemical manufacturing, as it replaces costly precursors with commodity alkyl halides.
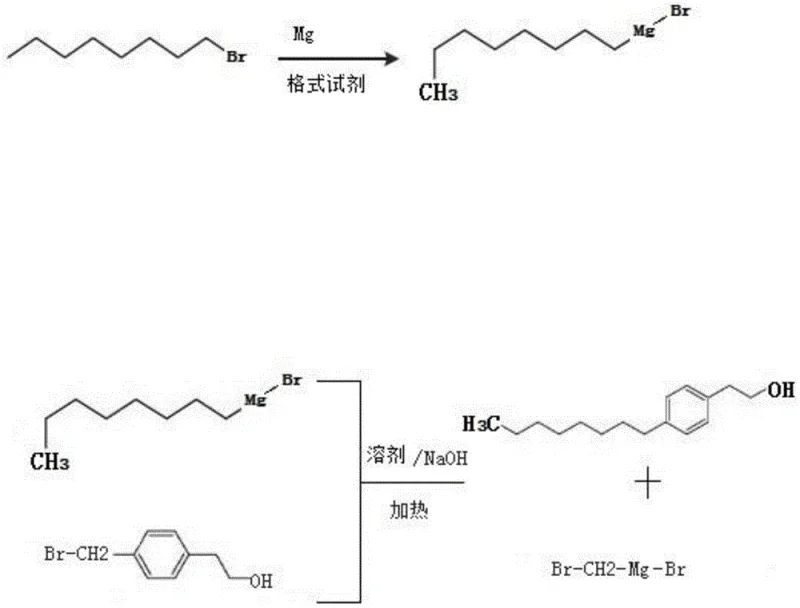
Mechanistic Insights into Grignard Alkylation and Protection Strategies
The core mechanistic advantage of this process lies in the precise control of the nucleophilic substitution reaction between the organomagnesium species and the electrophilic benzyl halide. In the first stage, the formation of the Grignard reagent is catalyzed by iodine, which activates the magnesium surface and initiates the oxidative insertion of magnesium into the carbon-halogen bond of bromooctane or chlorooctane. The use of THF as a solvent is particularly critical, as its cyclic ether structure forms stable complexes with the magnesium center, enhancing the solubility and stability of the resulting organometallic species. This stabilization is essential for maintaining the reactivity of the reagent during the subsequent heating phase. When the Grignard reagent encounters the electrophile, typically a bromomethyl-substituted aromatic ring, a concerted SN2-type displacement occurs, forging the new C-C bond that links the octyl chain to the phenethyl backbone. The presence of a base, such as sodium hydroxide, in the reaction mixture helps to neutralize acidic by-products and drives the equilibrium toward the desired product.
Furthermore, the patent elucidates a sophisticated mechanism for impurity control through the use of protecting groups, which is vital for achieving the stringent purity specifications required by pharmaceutical clients. In scenarios where the free hydroxyl group of the phenethyl alcohol might interfere with the Grignard reagent—potentially leading to protonation and destruction of the organometallic species—the hydroxyl is masked as a trityl ether. This bulky protecting group effectively shields the oxygen atom, preventing side reactions while remaining stable under the alkylation conditions. Following the coupling reaction, the protecting group is cleanly removed via catalytic hydrogenation using 10% Pd/C in a methanol/ethyl acetate mixture. This deprotection step is highly selective and mild, ensuring that the sensitive aromatic ring and the newly formed alkyl chain remain intact. This dual-phase strategy of protection and deprotection ensures that the final impurity profile is drastically simplified, reducing the burden on downstream purification units.
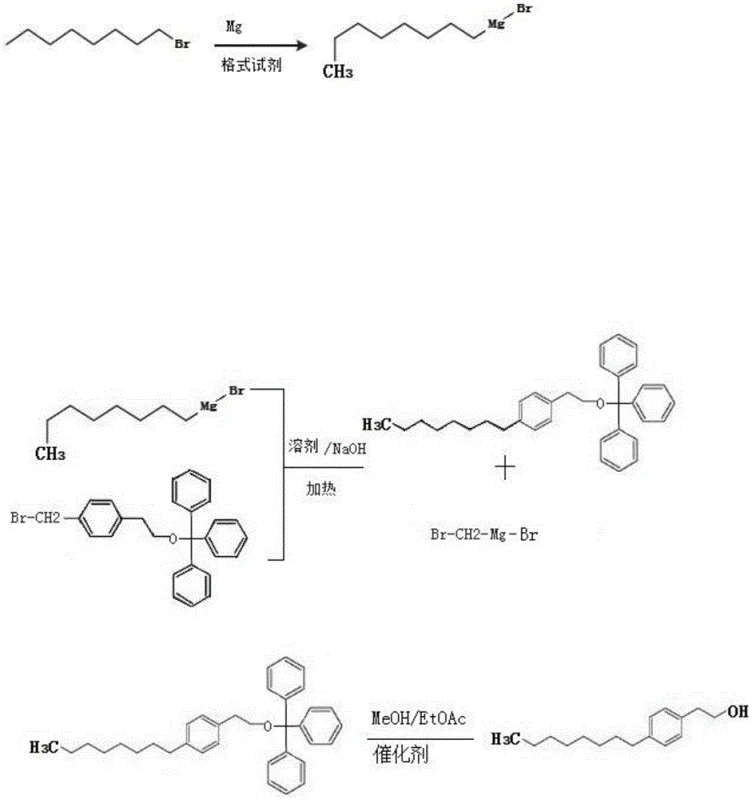
How to Synthesize 4-Octyl Phenethyl Alcohol Efficiently
The synthesis of 4-octyl phenethyl alcohol via this patented Grignard route requires careful attention to anhydrous conditions and temperature control to maximize the yield, which has been demonstrated to reach up to 80% in optimized pilot experiments. The process begins with the rigorous drying of all glassware and solvents, as moisture is the primary enemy of Grignard reagents. The formation of n-octyl magnesium bromide is initiated by adding iodine to magnesium chips in THF, followed by the slow addition of bromooctane to manage the exotherm. Once the gray, nearly transparent Grignard solution is obtained, it is reacted with the protected or unprotected phenethyl precursor in the presence of a base. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to assist technical teams in replicating this high-efficiency protocol.
- Synthesize n-octyl magnesium bromide by reacting bromooctane with magnesium chips in anhydrous THF or ether under inert gas protection, utilizing iodine as a catalyst.
- Protect the hydroxyl group of the phenethyl alcohol precursor using trityl chloride (TrCl) in the presence of DMAP and pyridine to form the trityl ether.
- Perform the alkylation reaction between the protected precursor and the Grignard reagent at 110-120°C, followed by catalytic hydrogenation to remove the protecting group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN111978154A offers profound strategic benefits that extend beyond simple yield improvements. The shift from specialized, high-cost precursors like triflates to commodity chemicals such as bromooctane and magnesium metal fundamentally alters the cost structure of the molecule. This transition eliminates the dependency on single-source suppliers for exotic starting materials, thereby diversifying the supply base and mitigating the risk of raw material shortages. Additionally, the simplified reaction workflow reduces the number of unit operations required, which directly translates to lower energy consumption and reduced labor costs per kilogram of finished product. The ability to run the reaction at moderate temperatures (110-120°C) rather than extreme conditions further enhances process safety and reduces the wear and tear on reactor vessels, contributing to long-term capital expenditure savings.
- Cost Reduction in Manufacturing: The elimination of expensive leaving groups and the use of abundant alkyl halides significantly lowers the direct material costs associated with production. By avoiding the complex synthesis of acetate or triflate precursors, manufacturers can realize substantial cost savings that improve margin potential in competitive markets. The streamlined process also reduces solvent usage and waste disposal fees, as the reaction generates fewer hazardous by-products compared to traditional methods. This economic efficiency makes the Grignard route highly attractive for large-volume commercial production where even marginal cost reductions have a significant impact on the bottom line.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents like bromooctane and magnesium ensures a stable and continuous supply of inputs, reducing the lead time for high-purity 4-octyl phenethyl alcohol. The robustness of the Grignard reaction against minor variations in feedstock quality adds a layer of resilience to the manufacturing process, ensuring consistent output even when raw material specifications fluctuate slightly. This reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery schedules of global pharmaceutical and fragrance clients who require uninterrupted supply chains.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated from laboratory bench scales to pilot plant operations with consistent results. The reduced generation of wastewater and exhaust gas aligns with increasingly stringent environmental regulations, minimizing the need for expensive effluent treatment infrastructure. This green production capability not only lowers compliance risks but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important in vendor selection criteria for multinational corporations committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Grignard-based synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent, providing clarity on yield expectations, reagent choices, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of the Grignard method described in CN111978154A?
A: The method offers significantly higher yields (up to 80% in optimized examples) compared to prior art, operates at lower reaction temperatures (110-120°C), and utilizes readily available alkyl halides, reducing raw material costs and environmental impact.
Q: Why is a protecting group used in the synthesis of 4-octyl phenethyl alcohol?
A: Protecting groups such as trityl are employed to shield the hydroxyl functionality during the harsh Grignard alkylation conditions. This prevents side reactions and ensures the integrity of the alcohol group, which is subsequently restored via mild catalytic hydrogenation.
Q: Can alkyl chlorides be used instead of bromides in this process?
A: Yes, the patent demonstrates that n-octyl magnesium chloride can be synthesized and used; however, alkyl bromides are generally preferred due to their higher reactivity and faster reaction kinetics, whereas chlorides may require more vigorous conditions or result in lower yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Octyl Phenethyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global fine chemical market. Our team of expert chemists has extensively analyzed the Grignard alkylation pathway described in CN111978154A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors capable of handling moisture-sensitive Grignard reactions safely and efficiently, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the absence of residual halides and heavy metals, guaranteeing a product quality that exceeds industry standards for pharmaceutical and specialty applications.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this Grignard-based process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more sustainable and profitable supply model.
