Scalable Synthesis of Codling Moth Pheromone Intermediate via Novel Borohydride Reduction Technology
The development of efficient synthetic routes for insect sex pheromones is critical for modern integrated pest management strategies, particularly for controlling devastating pests like the codling moth (Cydia pomonella). Patent CN103360248A introduces a groundbreaking methodology for the synthesis of the key intermediate (2E, 4E)-2,4-hexadienol acetate, utilizing sorbic acid as a readily available starting material. This innovation addresses long-standing challenges in the field by replacing hazardous, moisture-sensitive reagents with a robust borohydride anion reducing agent system in an inorganic aqueous alkaline environment. By shifting away from traditional lithium aluminium hydride or red aluminum reductions, this technology offers a pathway that is not only chemically elegant but also operationally superior for industrial applications. The process ensures high stereoselectivity and purity, which are paramount for the biological efficacy of the final pheromone product, while simultaneously reducing the environmental footprint associated with heavy metal waste and corrosive byproducts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (2E, 4E)-2,4-hexadienol acetate has relied on pathways that pose significant safety and operational hurdles for large-scale manufacturing. As illustrated in the prior art, conventional Route One typically involves the esterification of sorbic acid followed by a rigorous reduction step using lithium aluminium hydride (LiAlH4) complexed with aluminum chloride.
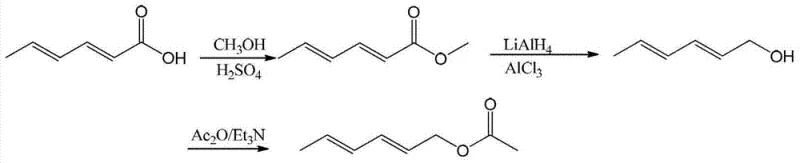
This approach demands strictly anhydrous conditions due to the pyrophoric nature of LiAlH4, requiring specialized equipment and inert atmosphere protocols that drastically increase capital expenditure and operational complexity. Furthermore, the post-reaction quenching of such powerful hydrides generates substantial amounts of aluminum salt waste, complicating downstream processing and environmental compliance. Similarly, Route Two employs thionyl chloride for esterification and Vitride (sodium bis(2-methoxyethoxy)aluminum hydride) for reduction, which introduces toxic glycol ether byproducts and volatile sulfur compounds that are difficult to manage in a green chemistry framework.
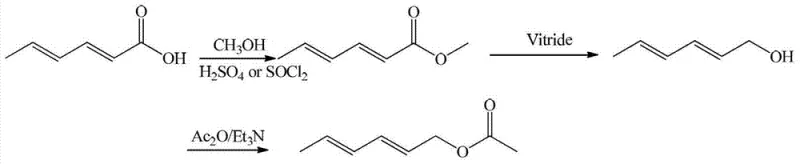
The Novel Approach
In stark contrast, the methodology disclosed in CN103360248A circumvents these pitfalls by employing a mixed anhydride strategy coupled with a mild aqueous reduction system. Instead of converting sorbic acid directly to an ester using corrosive acids or chlorinating agents, the process activates the carboxylic acid via reaction with ethyl chloroformate to form an active mixed anhydride in situ. This intermediate is then subjected to reduction using a borohydride anion reducing agent, such as sodium borohydride or potassium borohydride, dissolved in a dilute inorganic base aqueous solution. This fundamental shift allows the reduction to proceed in a homogeneous system that tolerates trace moisture, thereby eliminating the need for expensive drying agents and rigid anhydrous protocols. The result is a streamlined workflow that maintains high stereochemical integrity while significantly lowering the barrier to entry for safe, commercial-scale production.
Mechanistic Insights into Mixed Anhydride Activation and Aqueous Borohydride Reduction
The core innovation of this synthesis lies in the precise control of the reduction potential through the formation of a mixed anhydride intermediate. When sorbic acid reacts with ethyl chloroformate in the presence of triethylamine, it generates a highly reactive species that is more susceptible to nucleophilic attack by hydride ions than the parent acid, yet less prone to over-reduction or side reactions compared to simple esters. The subsequent addition of the borohydride/inorganic base aqueous solution creates a unique biphasic or emulsified environment where the hydride transfer occurs selectively at the carbonyl carbon of the anhydride. The presence of the inorganic base, such as sodium hydroxide or potassium hydroxide, stabilizes the borohydride species and modulates its reactivity, preventing the reduction of the conjugated diene system which is sensitive to harsh reducing conditions. This delicate balance ensures that the (2E, 4E) geometry of the double bonds is preserved throughout the transformation, yielding (2E, 4E)-hexadiene-1-ol with exceptional purity.
Furthermore, the mechanism facilitates superior impurity control by avoiding the formation of complex aluminum coordination complexes that often trap product or generate difficult-to-remove byproducts in traditional methods. In the LiAlH4 route, the formation of alkoxyaluminum intermediates requires careful acidic hydrolysis, which can lead to polymerization of the sensitive diene alcohol if not meticulously controlled. Conversely, the borohydride reduction produces simple borate salts that are easily separated during the aqueous workup. The use of toluene extraction effectively partitions the organic product from the inorganic salts, and the final distillation steps are simplified due to the cleaner crude profile. This mechanistic advantage translates directly into higher isolated yields and reduced loss of valuable material during purification, addressing a critical pain point in the manufacturing of high-value pheromone intermediates where every percentage point of yield impacts profitability.
How to Synthesize (2E, 4E)-2,4-Hexadienol Acetate Efficiently
The practical implementation of this synthesis involves a logical three-step sequence that can be adapted for both laboratory and pilot plant scales. The process begins with the activation of sorbic acid under nitrogen protection, followed by the critical reduction step where temperature control between -20°C and 0°C is maintained to ensure selectivity. The final esterification with acetic anhydride is straightforward and high-yielding. For detailed operational parameters, stoichiometry, and specific workup procedures validated by experimental data, please refer to the standardized protocol below.
- Formation of active mixed anhydride from sorbic acid and ethyl chloroformate in THF with triethylamine at 0-25°C.
- Selective reduction of the mixed anhydride using a borohydride anion reducing agent in an inorganic base aqueous solution at -20 to 0°C.
- Final esterification of the resulting (2E, 4E)-hexadiene-1-ol with acetic anhydride to yield the target acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this patented technology offers transformative benefits that extend beyond mere chemical efficiency. The shift from exotic, hazardous reagents to commodity chemicals fundamentally alters the cost structure and risk profile of the manufacturing process. By utilizing sorbic acid, a mass-produced food preservative, as the starting material, the supply chain becomes inherently more stable and less susceptible to the volatility associated with specialty fine chemical feedstocks. Moreover, the elimination of pyrophoric reagents like lithium aluminium hydride removes the need for specialized storage facilities and hazardous material handling certifications, thereby reducing insurance costs and regulatory burdens for the manufacturing site.
- Cost Reduction in Manufacturing: The economic implications of replacing Vitride or LiAlH4 with sodium or potassium borohydride are profound, as the latter are produced on a massive global scale and are significantly less expensive per mole of hydride delivered. Additionally, the avoidance of thionyl chloride and concentrated sulfuric acid eliminates the costs associated with corrosion-resistant equipment (such as glass-lined reactors) and the neutralization of acidic off-gases. The simplified workup procedure, which does not require complex filtration of gelatinous aluminum salts, reduces labor hours and solvent consumption during the isolation phase. These cumulative factors contribute to a substantially lower cost of goods sold (COGS), allowing for more competitive pricing in the agrochemical intermediate market without sacrificing margin.
- Enhanced Supply Chain Reliability: Relying on reagents that are sensitive to moisture and air, such as those used in conventional routes, introduces significant fragility into the supply chain, where minor deviations in storage or transport can render batches unusable. The new aqueous borohydride system is robust and forgiving, ensuring consistent batch-to-batch quality even under less-than-ideal logistical conditions. This reliability minimizes the risk of production delays caused by reagent degradation or failed quality control checks on incoming raw materials. Furthermore, the use of common solvents like toluene and THF, rather than specialized glycol ethers, ensures that solvent supply remains uninterrupted, securing the continuity of production schedules essential for meeting seasonal demand in the agricultural sector.
- Scalability and Environmental Compliance: Scaling up reactions involving energetic hydrides often requires disproportionate increases in safety infrastructure, whereas the aqueous borohydride reduction scales linearly with minimal additional risk. The waste stream generated by this process consists primarily of benign inorganic salts and triethylamine hydrochloride, which are far easier to treat in standard wastewater facilities compared to the heavy metal-laden sludge from aluminum-based reductions. This alignment with green chemistry principles not only simplifies environmental permitting but also future-proofs the manufacturing process against increasingly stringent global regulations on industrial effluent. The ability to operate with lower equipment requirements and simpler waste management makes this route exceptionally well-suited for rapid capacity expansion to meet growing market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners and licensees.
Q: Why is the borohydride/aqueous base system superior to Lithium Aluminium Hydride for this synthesis?
A: The borohydride system operates under mild, non-anhydrous conditions, eliminating the safety hazards and strict moisture control required by pyrophoric LiAlH4, while simplifying waste treatment.
Q: What is the overall yield advantage of this new method compared to conventional routes?
A: The novel route achieves a total recovery significantly higher than conventional literature methods (which range from 50% to 67.7%), with specific embodiments demonstrating yields exceeding 60% with high purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses easily available raw materials like sorbic acid, avoids corrosive reagents like thionyl chloride, and generates manageable waste streams, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2E, 4E)-2,4-Hexadienol Acetate Supplier
The technological advancements detailed in CN103360248A represent a significant leap forward in the production of codling moth pheromone intermediates, combining high purity with operational safety. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel borohydride reduction system are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of (2E, 4E)-2,4-hexadienol acetate meets the exacting standards required for effective pest control formulations.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and sustainability in the competitive agrochemical landscape.
