Revolutionizing Pheromone Intermediate Production via Mild Acetylide Chemistry
The global demand for effective and environmentally friendly pest control solutions has intensified the focus on semiochemicals, particularly sex pheromones used in integrated pest management (IPM) programs. A critical bottleneck in this supply chain has always been the efficient production of key aliphatic alcohol intermediates. Patent CN110294664B, published in March 2023, addresses this challenge directly by disclosing a novel, robust synthetic method for 2-tetradecyn-1-ol (CAS: 51309-22-9). This compound serves as the foundational building block for the sex pheromones of the American White Moth (Hyphantria cunea), a devastating invasive species affecting forestry and agriculture worldwide. The patent outlines a transformative shift from hazardous, cryogenic organolithium chemistry to a mild, one-pot acetylide alkylation strategy. By utilizing acetylene gas as a cheap starting material and avoiding extreme low-temperature conditions, this technology promises to redefine the cost structure and safety profile of agrochemical intermediate manufacturing. The strategic importance of this innovation lies not just in the chemistry itself, but in its potential to secure the supply chain for high-volume pheromone dispensers used in monitoring and mating disruption.
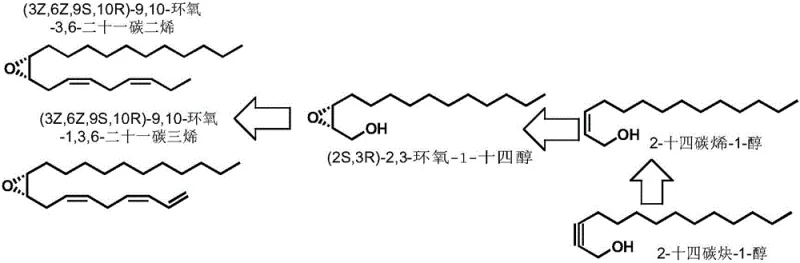
The significance of this patent extends beyond mere academic interest; it represents a viable industrial solution for a reliable agrochemical intermediate supplier seeking to optimize their portfolio. The traditional reliance on complex multi-step syntheses often results in low overall yields and prohibitive costs, limiting the widespread adoption of pheromone-based pest control. By streamlining the synthesis of 2-tetradecyn-1-ol, this method directly impacts the economic feasibility of producing the final pheromone components, such as (3Z,6Z,9S,10R)-9,10-epoxy-3,6-heneicosadiene. For R&D directors and procurement managers alike, understanding the mechanistic advantages and operational simplicity of this new route is essential for evaluating long-term sourcing strategies and potential cost reduction in agrochemical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 2-tetradecyn-1-ol relied heavily on methodologies that were inherently difficult to scale and fraught with operational hazards. As detailed in the background art of the patent, the conventional route typically involves the reaction of propargyl alcohol with 1-bromoundecane using n-butyllithium (n-BuLi) as a strong base. This reaction must be conducted in a solvent system comprising hexamethylphosphoric triamide (HMPA) and tetrahydrofuran (THF). The most critical limitation of this approach is the stringent requirement for cryogenic conditions, specifically maintaining the reaction temperature at -78°C. Such extreme cold necessitates specialized equipment and significant energy expenditure, creating a substantial barrier to commercial scale-up. Furthermore, organolithium reagents like n-BuLi are pyrophoric and extremely sensitive to moisture and oxygen, demanding rigorous anhydrous and anaerobic conditions throughout the process. These factors combine to create a high-risk operational environment with complex safety protocols, making the conventional method unsuitable for the high-volume production required by the agricultural sector.
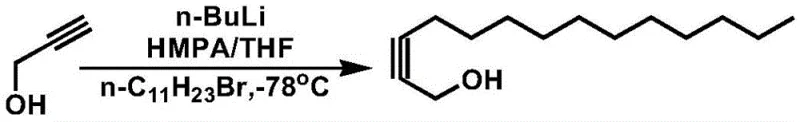
The Novel Approach
In stark contrast to the legacy methods, the invention described in CN110294664B introduces a paradigm shift by employing acetylene gas as the primary carbon source. This novel approach bypasses the need for expensive and hazardous organolithium bases entirely. Instead, it utilizes a variety of accessible bases such as sodium hydroxide, potassium hydroxide, or even Grignard reagents to generate the reactive acetylide species in situ. The reaction proceeds under remarkably mild conditions, with temperatures ranging from -20°C to 80°C, eliminating the need for cryogenic cooling. The process is designed as a "one-pot" synthesis, where the acetylene is first converted to an acetylide salt, alkylated with 1-bromoundecane to form a 1-tridecyne salt, and subsequently reacted with a formaldehyde source (paraformaldehyde, aqueous formaldehyde, or formaldehyde gas) to yield the final alcohol. This telescoping of steps not only simplifies the workflow but also minimizes material loss between isolation stages, thereby enhancing the overall atom economy and yield of the high-purity agrochemical intermediate.
Mechanistic Insights into One-Pot Acetylide Alkylation
The core of this technological breakthrough lies in the efficient generation and stabilization of the acetylide anion under non-cryogenic conditions. In the initial step, acetylene gas is bubbled into a suspension of a base (e.g., NaOH or KOH) in a polar aprotic or etheral solvent like THF or sulfolane. The base deprotonates the terminal alkyne to form the metal acetylide salt. Unlike the transient and highly reactive lithium acetylides formed at -78°C, these salts exhibit sufficient stability to allow for the subsequent alkylation step at ambient or slightly elevated temperatures. When 1-bromoundecane is introduced, a nucleophilic substitution (SN2) occurs, extending the carbon chain to form the 1-tridecyne intermediate. The brilliance of this mechanism is its tolerance; the reaction does not require the absolute exclusion of moisture that characterizes organolithium chemistry, allowing for more flexible process control. This robustness is a key factor in ensuring consistent quality and reducing batch-to-batch variability, which is critical for commercial scale-up of complex agrochemical intermediates.
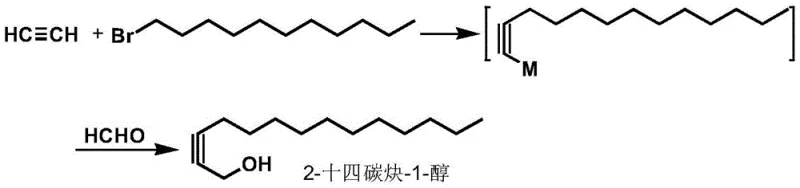
Following the alkylation, the reaction mixture is treated with a formaldehyde source to install the hydroxymethyl group, completing the synthesis of 2-tetradecyn-1-ol. The patent highlights the versatility of this step, noting that paraformaldehyde, formalin, or gaseous formaldehyde can all be effectively utilized. The reaction temperature for this final hydroxymethylation step is controlled between 0°C and 100°C, further emphasizing the thermal flexibility of the process. From an impurity control perspective, the use of simpler reagents and the avoidance of aggressive lithiation reduces the formation of side products such as polymerization byproducts or over-alkylated species. The resulting crude product can be purified via standard techniques like column chromatography or distillation to achieve purities exceeding 99%, as demonstrated in the patent examples. This high level of purity is essential because downstream derivatization into the final epoxide pheromones requires precise stereochemistry and functional group integrity, which can be compromised by trace impurities from the starting alcohol.
How to Synthesize 2-Tetradecyn-1-ol Efficiently
Implementing this synthesis in a pilot or production plant requires careful attention to gas handling and exotherm management, although the risks are far lower than those associated with cryogenic lithiation. The process begins with the preparation of the base-solvent system, followed by the controlled introduction of acetylene gas to ensure saturation without over-pressurization. Once the acetylide salt is formed, the alkyl halide is added dropwise to manage the heat of reaction. Finally, the formaldehyde source is introduced to complete the carbon skeleton assembly. The simplicity of the workup—often involving aqueous quenching and organic extraction—makes this route highly attractive for process chemists looking to streamline operations. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature profiles validated in the patent, please refer to the standardized guide below.
- Generate acetylide salt by reacting acetylene gas with a base (e.g., NaOH, KOH, or Grignard reagent) in a suitable solvent like THF or MTBE at temperatures between -20°C and 80°C.
- Alkylate the acetylide salt by adding 1-bromoundecane to the reaction mixture, stirring for 2-5 hours to form the 1-tridecyne salt intermediate.
- Complete the one-pot synthesis by introducing a formaldehyde source (paraformaldehyde, aqueous solution, or gas) at 0-100°C to yield the final 2-tetradecyn-1-ol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the conventional n-BuLi route to this acetylene-based methodology offers profound strategic benefits. The most immediate impact is seen in the raw material cost structure. Acetylene is a commodity chemical produced on a massive scale globally, making it significantly cheaper and more readily available than propargyl alcohol or specialized organolithium reagents. Furthermore, the elimination of HMPA, a solvent with known toxicity and regulatory scrutiny issues, simplifies environmental compliance and waste disposal protocols. The ability to run the reaction at near-ambient temperatures drastically reduces the energy load on the facility, removing the need for expensive cryogenic cooling systems and the associated maintenance costs. These factors collectively contribute to a leaner, more cost-effective manufacturing process that enhances the competitiveness of the final pheromone products in the global market.
- Cost Reduction in Manufacturing: The replacement of expensive, hazardous reagents like n-butyllithium and HMPA with commodity bases (NaOH, KOH) and common solvents (THF, MTBE) fundamentally alters the bill of materials. By removing the dependency on cryogenic infrastructure, the capital expenditure (CAPEX) for new production lines is significantly lowered, while operational expenditure (OPEX) is reduced through lower energy consumption and simplified safety measures. This economic efficiency allows for more competitive pricing of the final agrochemical intermediate, enabling broader adoption of pheromone-based pest control technologies.
- Enhanced Supply Chain Reliability: Sourcing n-butyllithium and maintaining a cold chain for its transport can be logistically challenging and prone to disruptions. In contrast, acetylene and 1-bromoundecane are stable, widely available chemicals with robust supply networks. The simplified process also reduces the lead time for production batches, as there is no need for extensive cooldown periods or complex inert atmosphere setups. This reliability ensures a steady flow of materials to downstream formulators, mitigating the risk of stockouts during peak pest seasons when demand for monitoring traps is highest.
- Scalability and Environmental Compliance: The "one-pot" nature of the synthesis minimizes the number of unit operations, reducing the physical footprint required for manufacturing. Fewer transfer steps mean less opportunity for material loss and contamination, leading to higher overall yields. Additionally, the avoidance of toxic solvents like HMPA aligns with increasingly stringent global environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste. This green chemistry advantage not only future-proofs the production facility against regulatory changes but also enhances the brand reputation of the manufacturer as a sustainable partner in the agricultural value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in CN110294664B, providing a clear picture of what partners can expect when adopting this technology. Understanding these nuances is vital for making informed decisions about process integration and supplier qualification.
Q: What are the primary advantages of the new acetylene-based synthesis over the traditional propargyl alcohol route?
A: The new method eliminates the need for cryogenic conditions (-78°C) and strict anhydrous environments required by n-butyllithium chemistry. It utilizes inexpensive acetylene gas and operates at much milder temperatures (-20°C to 80°C), significantly reducing energy consumption and safety risks associated with organolithium reagents.
Q: Can this process be scaled for industrial production of pheromone intermediates?
A: Yes, the process is specifically designed for scalability. The one-pot nature reduces unit operations, and the use of common solvents like THF or MTBE alongside robust bases like NaOH or KOH makes it highly suitable for large-scale manufacturing compared to the hazardous scale-up limitations of the prior art.
Q: What purity levels can be achieved with this synthetic method?
A: Experimental data from the patent indicates that the method consistently yields 2-tetradecyn-1-ol with high purity, ranging from 97.5% to 99.5% after standard purification steps like column chromatography, meeting the stringent requirements for downstream pheromone synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Tetradecyn-1-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful deployment of advanced pest management solutions relies on the availability of high-quality, cost-effective intermediates. Our team of expert process chemists has thoroughly analyzed the methodology disclosed in CN110294664B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless and efficient. Our state-of-the-art facilities are designed to handle gaseous reagents and exothermic reactions safely, while our rigorous QC labs guarantee that every batch of 2-tetradecyn-1-ol meets stringent purity specifications required for sensitive pheromone synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal for securing your supply of this critical agrochemical intermediate. Let us help you drive down costs and secure your production pipeline with our proven expertise in fine chemical manufacturing.
