Advanced Synthetic Route for 3,5-Disubstituted Pyrazoles: Enhancing Commercial Scalability and Purity
Advanced Synthetic Route for 3,5-Disubstituted Pyrazoles: Enhancing Commercial Scalability and Purity
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for higher purity and greater efficiency in the production of active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is detailed in patent CN101407496B, which discloses a robust and practical synthetic method for 3,5-disubstituted pyrazoles. This class of compounds serves as a critical scaffold in medicinal chemistry, finding extensive applications in the development of anti-inflammatory agents, kinase inhibitors, and agrochemical actives. The disclosed technology addresses long-standing inefficiencies in prior art, specifically targeting the issues of excessive reagent consumption, prolonged reaction durations, and suboptimal yields that have historically plagued the commercial manufacturing of these valuable intermediates. By optimizing solvent systems and stoichiometric ratios, this innovation offers a pathway to significantly enhance the economic viability of pyrazole production.
For R&D directors and process chemists, the implications of this patent extend beyond mere academic interest; it represents a tangible opportunity to streamline supply chains and reduce the cost of goods sold (COGS) for complex drug candidates. The method leverages a sequential condensation and cyclization strategy that avoids the use of hazardous or expensive transition metal catalysts, thereby simplifying the impurity profile and reducing the burden on downstream purification processes. As a reliable pharmaceutical intermediate supplier, understanding and adopting such optimized routes is essential for maintaining competitiveness in a market that increasingly values sustainability and operational excellence. The following analysis dissects the technical nuances of this synthesis, providing a comprehensive view of its mechanistic advantages and commercial potential for global procurement teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-disubstituted pyrazoles has been fraught with significant technical and economic challenges that hinder large-scale adoption. Traditional methodologies, such as the condensation of alpha,beta-unsaturated ketones with hydrazines or the reaction of 1,3-diketones with hydrazine derivatives, often suffer from poor regioselectivity and difficult purification protocols. More critically, emerging methods involving the direct reaction of aldehydes, hydrazines, and alkynes have demonstrated severe limitations in efficiency. Literature precedents indicate that achieving acceptable conversion rates often necessitates the use of massive excesses of alkyne reagents, sometimes up to 5 equivalents, which drastically inflates raw material costs given the high price of functionalized alkynes. Furthermore, these conventional processes frequently require extended reaction times, often exceeding 48 hours, which ties up reactor capacity and increases energy consumption. The combination of low yields, typically ranging from 20% to 60%, and the generation of complex byproduct mixtures makes the isolation of high-purity product arduous and economically unsustainable for commercial operations.
The Novel Approach
In stark contrast to these inefficient legacy processes, the method described in patent CN101407496B introduces a paradigm shift through rigorous optimization of reaction parameters. The core innovation lies in the strategic selection of toluene as the reaction solvent and the precise control of stoichiometry, particularly regarding the alkyne component. By reducing the alkyne loading from the conventional 5 equivalents down to merely 1.5 equivalents, the process achieves a dramatic improvement in atom economy and cost efficiency without sacrificing conversion. Additionally, the reaction time for the critical cyclization step is slashed from 48 hours to just 12 hours, facilitated by the use of an aqueous sodium hydroxide system that effectively promotes the transformation under mild thermal conditions (45°C). This novel approach not only boosts isolated yields to an impressive range of 80-90% but also simplifies the workup procedure, allowing for the direct isolation of products with purity levels reaching up to 95% after standard processing. This represents a substantial leap forward in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Promoted Cyclization
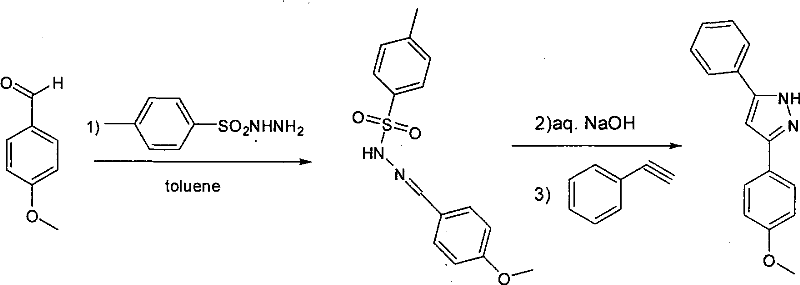
The chemical elegance of this synthesis lies in its multi-step cascade nature, which proceeds through a well-defined sequence of condensation and cycloaddition events. The process initiates with the formation of a sulfonyl hydrazone intermediate via the condensation of the starting aldehyde with a sulfonyl hydrazine, such as p-toluenesulfonyl hydrazide or methanesulfonyl hydrazide. This step is conducted in toluene at ambient temperature, ensuring high conversion without the need for aggressive heating. Subsequently, the addition of an aqueous base, specifically 5M NaOH, serves a dual purpose: it neutralizes the acidic byproducts and activates the hydrazone species, potentially generating a reactive diazo intermediate in situ. This activated species is then poised for nucleophilic attack or cycloaddition with the terminal alkyne. The presence of the base facilitates the deprotonation of the alkyne or the stabilization of the transition state required for ring closure, ultimately leading to the formation of the pyrazole core with the expulsion of the sulfonyl group. The choice of toluene is crucial here, as it provides an ideal biphasic environment that supports the interaction between the organic substrates and the aqueous base while allowing for easy separation post-reaction.
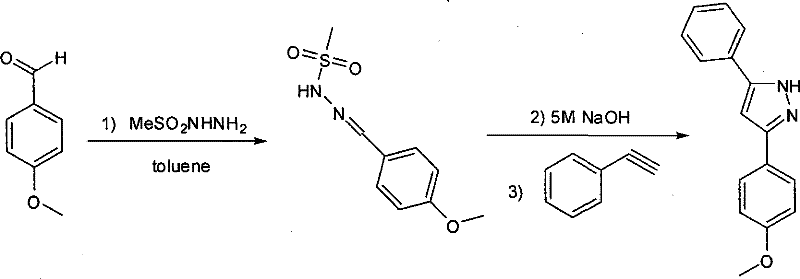
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of heavy metal contamination, a critical quality attribute for API intermediates destined for regulatory submission. Furthermore, the high selectivity of the base-promoted cyclization minimizes the formation of oligomeric byproducts or regioisomers that often complicate the purification of pyrazoles synthesized via other routes. The reaction conditions are sufficiently mild to preserve sensitive functional groups on both the aldehyde and alkyne substrates, as evidenced by the successful synthesis of derivatives containing bromo and methoxy substituents without degradation. This robustness ensures a clean impurity profile, reducing the need for extensive chromatographic purification and enabling the use of simpler crystallization or extraction techniques for final isolation, which is a key factor in scaling up complex pharmaceutical intermediates.
How to Synthesize 3,5-Disubstituted Pyrazole Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified in the patent to ensure reproducibility and maximum yield. The process is designed to be operationally simple, utilizing readily available reagents and standard equipment, which lowers the barrier to entry for contract manufacturing organizations. The following overview outlines the critical stages of the synthesis, emphasizing the specific conditions that drive the high efficiency of the transformation. For a detailed breakdown of the standardized operating procedures, please refer to the technical guide below.
- Dissolve the aldehyde compound in toluene and react with sulfonyl hydrazine at room temperature for 3 hours to form the intermediate hydrazone.
- Add aqueous NaOH solution (equimolar to hydrazine) to the mixture and stir at room temperature for 20 minutes to activate the intermediate.
- Introduce 1.5 equivalents of the alkyne compound, heat the reaction to 45°C, and maintain stirring for 12 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized synthetic route translates directly into enhanced operational resilience and significant cost avoidance. The primary driver of value is the drastic reduction in raw material consumption, particularly regarding the alkyne component, which is often the most expensive reagent in the sequence. By lowering the stoichiometric requirement from 5 equivalents to 1.5 equivalents, the process inherently reduces the variable cost per kilogram of the final product. Moreover, the shortened reaction cycle time of 12 hours compared to the industry standard of 48 hours allows for increased throughput within existing manufacturing facilities, effectively expanding capacity without capital expenditure. This efficiency gain is compounded by the simplified downstream processing; the ability to achieve high crude purity (up to 96% in some embodiments) minimizes solvent usage for recrystallization and reduces the load on waste treatment systems, aligning with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the optimized stoichiometry and reduced energy inputs. By eliminating the need for a large excess of alkyne, the direct material cost is significantly lowered, which is a critical factor when sourcing high-purity pharmaceutical intermediates at scale. Additionally, the use of inexpensive inorganic bases like sodium hydroxide and common solvents like toluene avoids the premium pricing associated with specialized catalysts or exotic reagents. The high yield of 80-90% further amplifies these savings by maximizing the output from each batch, thereby reducing the effective cost of goods sold and improving the overall margin structure for the final API.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on commodity chemicals that are widely available from multiple global vendors. Unlike processes dependent on scarce or proprietary catalysts, this method utilizes aldehydes, sulfonyl hydrazines, and alkynes that can be sourced with short lead times, mitigating the risk of production delays due to raw material shortages. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in reagent quality, ensuring consistent batch-to-batch performance. This reliability is essential for reducing lead time for high-purity intermediates, allowing pharmaceutical companies to maintain lean inventory levels without compromising on production schedules.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its straightforward workup procedure, which involves standard phase separation and solvent removal techniques that are easily transferable from gram to metric-ton scale. The absence of heavy metals simplifies the environmental, health, and safety (EHS) profile of the manufacturing process, reducing the costs associated with hazardous waste disposal and regulatory reporting. Furthermore, the high atom economy and reduced solvent intensity contribute to a lower carbon footprint, supporting corporate sustainability goals. This alignment with green chemistry principles makes the process attractive for long-term commercial scale-up of complex pharmaceutical intermediates in a regulated environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations. For further technical clarification or custom feasibility studies, our team is available to provide detailed support.
Q: How does this new method improve upon traditional pyrazole synthesis?
A: Traditional methods often require excessive alkyne usage (up to 5 equivalents) and prolonged reaction times (48 hours), resulting in yields between 20-60%. This patented method reduces alkyne consumption to 1.5 equivalents and shortens reaction time to 12 hours, achieving yields of 80-90%.
Q: What are the critical reaction conditions for optimal yield?
A: The process utilizes toluene as the preferred solvent. The key parameters include a 1:1 molar ratio of aldehyde to sulfonyl hydrazine, the use of 5M NaOH aqueous solution for activation, and maintaining the cyclization step at 45°C for 12 hours.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. It employs common solvents like toluene, avoids expensive transition metal catalysts, and simplifies purification through standard aqueous workups, making it highly economical for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Disubstituted Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN101407496B can be reliably replicated on an industrial scale. We are committed to delivering stringent purity specifications through our rigorous QC labs, which are equipped with advanced analytical instrumentation to monitor every critical quality attribute of the 3,5-disubstituted pyrazole intermediates we produce. Our dedication to process optimization allows us to offer solutions that balance cost-efficiency with the uncompromising quality standards required by the global pharmaceutical industry.
We invite procurement leaders and R&D directors to engage with us to explore how this advanced synthetic route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data for our pyrazole portfolio and to discuss route feasibility assessments for your upcoming projects. Let us collaborate to drive efficiency and innovation in your drug development pipeline, ensuring a secure and cost-effective supply of these critical building blocks.
