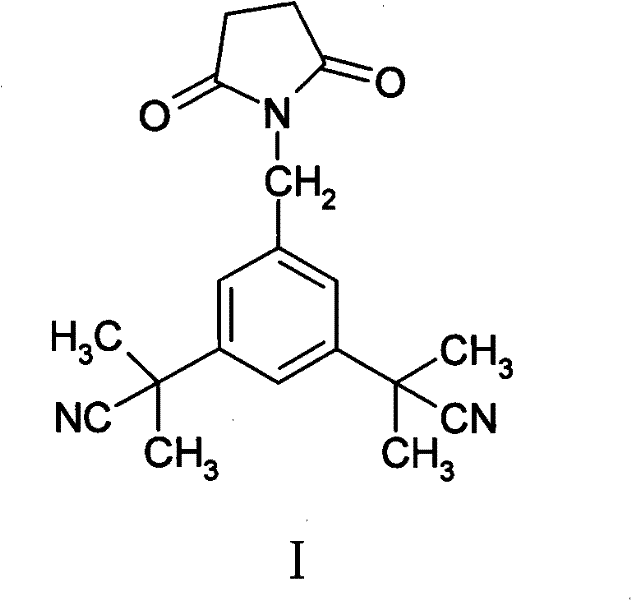
Advanced Synthesis and Isolation of Anastrozole Impurity I for Pharmaceutical QC
The pharmaceutical industry's relentless pursuit of safety and efficacy places immense pressure on the quality control of Active Pharmaceutical Ingredients (APIs), particularly for potent oncology drugs like Anastrozole. As detailed in Chinese Patent CN102050775B, the precise identification and quantification of process-related impurities are paramount for regulatory approval and patient safety. This patent discloses a sophisticated preparation method for alpha, alpha, alpha', alpha'-tetramethyl-5-(succinimide-1-methyl)-1,3-benzenediacetonitrile, a specific structural analog known as Impurity I, which arises during the synthesis of Anastrozole. By leveraging an integrated synthetic route that capitalizes on the inherent byproducts of the main reaction, this technology offers a robust pathway for generating high-purity reference standards essential for modern chromatographic analysis.
For R&D directors and quality assurance teams, the ability to access authentic impurity standards is not merely a regulatory checkbox but a fundamental requirement for method validation. The structural complexity of Anastrozole derivatives demands rigorous analytical protocols to distinguish the active drug from potential genotoxic or pharmacologically active byproducts. The methodology outlined in this patent provides a reliable source for this critical reference material, ensuring that analytical laboratories can accurately monitor the purity profile of Anastrozole batches throughout the manufacturing lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of specific pharmaceutical impurities for reference purposes has been a logistical and financial bottleneck for many fine chemical manufacturers. Conventional approaches often involve designing a completely independent synthetic route solely for the impurity, which requires unique starting materials, distinct reaction conditions, and separate purification trains. This siloed approach significantly inflates the cost of goods sold (COGS) for reference standards and extends lead times, as the impurity synthesis cannot leverage the economies of scale associated with the main API production. Furthermore, standalone synthesis routes may introduce different impurity profiles or stereochemical variances that do not accurately reflect the actual byproduct formed during the commercial manufacturing of the API, leading to discrepancies in analytical results.
The Novel Approach
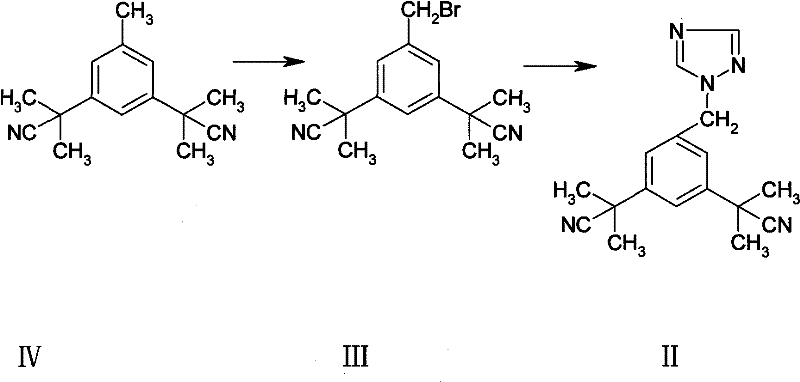
The innovative strategy presented in patent CN102050775B circumvents these inefficiencies by embedding the impurity generation directly within the primary Anastrozole synthesis workflow. Instead of treating the succinimide derivative as waste, the process recognizes it as a valuable co-product formed through the interaction of the benzylic bromide intermediate with succinimide, a natural byproduct of the N-bromosuccinimide (NBS) bromination step. By optimizing the workup and purification stages, manufacturers can simultaneously isolate the target API and the critical impurity standard from the same reaction batch. This dual-output capability drastically simplifies the supply chain for reference materials, ensuring that the impurity standard is chemically identical to the one found in production batches, thereby enhancing the accuracy of quality control assays.
Mechanistic Insights into Radical Bromination and Nucleophilic Substitution
The core chemical transformation driving this process involves a free-radical bromination followed by a competitive nucleophilic substitution. Initially, 3,5-bis(2-cyano-2-propyl)toluene (Compound IV) undergoes benzylic bromination using N-bromosuccinimide (NBS) in the presence of a radical initiator like benzoyl peroxide (Lucidol). This step generates the reactive benzylic bromide intermediate (Compound III) while releasing succinimide as a stoichiometric byproduct. In a standard Anastrozole synthesis, the succinimide is typically removed as waste; however, in this specific context, the proximity of the nucleophilic succinimide nitrogen to the electrophilic benzylic carbon of Compound III creates an opportunity for a secondary SN2 reaction. This side reaction leads to the N-alkylation of the succinimide ring, forming the target Impurity I (alpha, alpha, alpha', alpha'-tetramethyl-5-(succinimide-1-methyl)-1,3-benzenediacetonitrile).
Understanding this mechanistic pathway is crucial for controlling the levels of Impurity I in the final API. While the primary goal of the main synthesis is to react Compound III with 1,2,4-triazole to form Anastrozole (Compound II), the presence of succinimide necessitates careful monitoring. The patent details a purification protocol involving acid-base extraction and selective recrystallization to separate Impurity I from the desired Anastrozole product. By adjusting pH levels and utilizing specific solvent systems like ethyl acetate and n-hexane, the process effectively isolates the impurity in high purity (98-100%), transforming a potential quality liability into a valuable analytical asset.
How to Synthesize alpha, alpha, alpha', alpha'-tetramethyl-5-(succinimide-1-methyl)-1,3-benzenediacetonitrile Efficiently
The synthesis of this critical reference standard relies on a streamlined two-step sequence that mirrors the industrial production of Anastrozole, ensuring scalability and relevance. The process begins with the radical bromination of the precursor toluene derivative, followed by a substitution reaction where the impurity is generated alongside the main product. Detailed operational parameters, including specific molar ratios of NBS and initiators, as well as precise temperature controls during the reflux and stirring phases, are essential to maximize the yield of the intermediate bromide while managing the formation of the succinimide adduct. The subsequent isolation steps utilize differential solubility and acid-base properties to purify the target molecule from the complex reaction matrix.
- Perform radical bromination of 3,5-bis(2-cyano-2-propyl)toluene using N-bromosuccinimide (NBS) to generate the benzylic bromide intermediate.
- React the bromide intermediate with 1,2,4-triazole under phase transfer catalysis to form Anastrozole, allowing the succinimide byproduct to concurrently form Impurity I.
- Isolate the target impurity from the reaction mixture through acid-base extraction and recrystallization using ethyl acetate and n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this integrated synthesis method offers significant strategic benefits beyond mere technical feasibility. By aligning the production of the impurity standard with the main API manufacturing schedule, companies can eliminate the need for external sourcing of expensive reference materials, which often carry high markups and long delivery windows. This vertical integration ensures a consistent, on-demand supply of critical QC materials, reducing the risk of production delays caused by missing analytical standards. Furthermore, the ability to generate high-purity impurities internally enhances the company's regulatory standing, as it demonstrates a deep understanding and control over the entire impurity profile of the drug substance.
- Cost Reduction in Manufacturing: The primary economic advantage lies in the valorization of waste streams. Since the succinimide moiety required for Impurity I is generated in situ during the bromination step, there is no need to purchase additional succinimide reagents or dedicate separate reactor vessels for its synthesis. This elimination of redundant raw material costs and processing steps leads to substantial savings in operational expenditures. Additionally, the shared purification infrastructure means that the marginal cost of producing the impurity standard is negligible compared to standalone synthesis, allowing for significant cost reduction in pharmaceutical intermediate manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: Relying on third-party vendors for niche impurity standards introduces vulnerability into the supply chain, particularly when those vendors face their own raw material shortages or regulatory hurdles. By internalizing the production of Anastrozole Impurity I using this patented method, manufacturers secure a self-sufficient supply of reference materials. This autonomy ensures that quality control laboratories never face stockouts of critical standards, thereby maintaining uninterrupted release testing for Anastrozole batches. The robustness of the described chemical route, which utilizes common industrial reagents like NBS and triazoles, further guarantees that the supply of these standards remains stable and resilient against market fluctuations.
- Scalability and Environmental Compliance: The process described in the patent is inherently scalable, as it leverages unit operations already present in standard API plants, such as reflux reactors and crystallization tanks. This compatibility facilitates the seamless scale-up of impurity production from gram-scale laboratory batches to kilogram-scale commercial runs without requiring new capital investment. From an environmental perspective, the method adheres to green chemistry principles by maximizing atom economy; rather than discarding the succinimide byproduct, it is converted into a useful product. This reduction in chemical waste lowers the burden on effluent treatment facilities and supports the company's sustainability goals, making the overall manufacturing process more environmentally compliant and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific Anastrozole impurity. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making.
Q: What is the primary application of alpha,alpha,alpha',alpha'-tetramethyl-5-(succinimide-1-methyl)-1,3-benzenediacetonitrile?
A: This compound serves as a critical reference standard (Impurity I) for the quality control and HPLC analysis of Anastrozole API, ensuring regulatory compliance regarding impurity profiles.
Q: How does this patented method improve the availability of the impurity standard?
A: The method integrates the formation of the impurity within the standard Anastrozole synthesis workflow, utilizing the succinimide byproduct from the NBS bromination step, thereby avoiding the need for a separate, costly standalone synthesis.
Q: What represents the key purity specification for this reference material?
A: According to the patent data, the isolated compound achieves a purity level of 98% to 100% following recrystallization, making it highly suitable for quantitative analytical calibration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anastrozole Impurity I Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your analytical data is only as good as the reference standards you use. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demand for high-purity pharmaceutical intermediates and reference materials with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Anastrozole Impurity I we supply meets the exacting standards required for global regulatory submissions. Our commitment to quality ensures that your quality control processes remain robust, reliable, and fully compliant with international pharmacopeial requirements.
We invite you to collaborate with us to optimize your supply chain for Anastrozole related substances. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D and commercial production goals efficiently and economically.
