Revolutionizing NEP Inhibitor Intermediate Production via Advanced Catalytic Hydrogenation
The pharmaceutical industry's relentless pursuit of effective cardiovascular treatments has placed Neutral Endopeptidase (NEP) inhibitors at the forefront of therapeutic development. Patent CN103080072A introduces a groundbreaking methodology for synthesizing critical intermediates used in the production of these life-saving medications, specifically targeting gamma-amino-delta-biphenyl-alpha-methyl alkanoic acid backbones. This intellectual property represents a significant leap forward in process chemistry, addressing long-standing challenges related to stereoselectivity and yield that have historically plagued the manufacturing of compounds such as N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-(2R)-methylbutanoic acid ethyl ester. By leveraging advanced transition metal catalysis, this new process offers a robust alternative to conventional hydrogenation techniques, promising enhanced purity profiles and operational efficiency for global supply chains.
For R&D directors and process chemists, the implications of this technology are profound, as it provides a versatile toolkit for constructing complex chiral centers with high precision. The patent details multiple synthetic routes, including the isomerization of methylene-pyrrolidinones and the subsequent ring-opening to form unsaturated amino acid derivatives, which are then subjected to highly selective hydrogenation. This comprehensive approach not only expands the chemical space available for drug discovery but also solidifies the foundation for reliable large-scale manufacturing. As we delve deeper into the technical specifics, it becomes evident that this innovation is not merely an incremental improvement but a transformative strategy for producing high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of NEP inhibitor intermediates has been fraught with difficulties, particularly concerning the control of stereochemistry during the reduction of olefinic precursors. Traditional methods, such as those described in earlier patents like US5217996, often relied on non-selective hydrogenation over palladium on carbon, which frequently resulted in mixtures of diastereomers that were challenging and costly to separate. These conventional approaches typically suffered from low diastereomeric ratios, necessitating extensive downstream purification processes such as recrystallization or chromatography, which significantly drove up production costs and reduced overall throughput. Furthermore, the reliance on harsh reaction conditions or expensive stoichiometric reagents in older methodologies often led to safety concerns and environmental burdens, making them less attractive for modern green chemistry initiatives. The inability to consistently achieve high levels of stereochemical purity meant that manufacturers faced unpredictable yields and variable product quality, creating bottlenecks in the supply of active pharmaceutical ingredients.
The Novel Approach
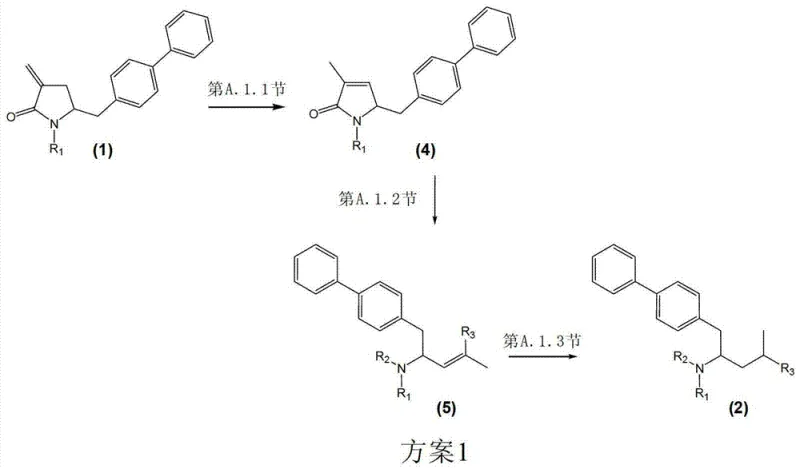
In stark contrast, the methodology outlined in CN103080072A presents a sophisticated solution by employing tailored transition metal catalysts combined with specific chiral ligands to dictate the stereochemical outcome of the hydrogenation step. This novel approach allows for the precise conversion of unsaturated intermediates, such as formula (5), into the desired saturated amino acid derivatives, formula (2), with exceptional diastereoselectivity. By utilizing catalysts based on rhodium, ruthenium, or iridium complexed with ligands like Josiphos, Walphos, or BINAP derivatives, the process can achieve diastereomeric ratios exceeding 99:1 in optimized embodiments, effectively eliminating the need for difficult separations. Additionally, the patent describes alternative pathways, such as the isomerization of formula (1) to formula (4) using palladium catalysts, providing manufacturers with flexibility in starting material selection. This versatility ensures that production can be adapted based on raw material availability and cost, thereby enhancing the overall resilience of the manufacturing process against market fluctuations.
Mechanistic Insights into Chiral Transition Metal Catalyzed Hydrogenation
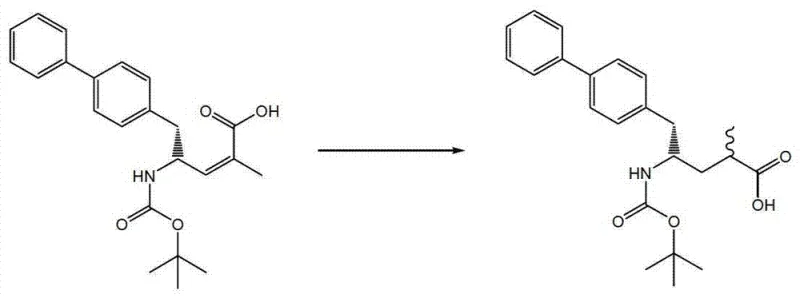
The core of this technological advancement lies in the intricate mechanism of asymmetric hydrogenation, where the geometry of the chiral ligand surrounding the transition metal center directs the addition of hydrogen to the olefinic double bond. When the unsaturated amino acid derivative, represented by formula (5a), is exposed to hydrogen gas in the presence of a chiral catalyst, the substrate coordinates to the metal center in a specific orientation dictated by steric and electronic interactions with the ligand. For instance, when using rhodium complexes with bulky phosphine ligands like SL-J003-1 or SL-W008-1, the catalyst creates a chiral pocket that favors the formation of the (2R, 4S) isomer over the (2S, 4S) isomer. This level of control is achieved through a delicate balance of ligand bite angle and electronic properties, which stabilize the transition state leading to the desired product while destabilizing the pathway to the unwanted diastereomer. The patent extensively catalogs various ligand structures, demonstrating that subtle modifications in the ligand backbone can dramatically shift the selectivity, offering chemists a fine-tuning mechanism to optimize the process for specific target molecules.
Beyond stereocontrol, the mechanism also addresses impurity management by minimizing side reactions such as over-reduction or isomerization of the double bond prior to hydrogenation. The use of homogeneous catalysts allows for uniform activity throughout the reaction mixture, ensuring consistent conversion rates and reducing the formation of byproducts that often arise from heterogeneous surface effects. Furthermore, the patent highlights the importance of solvent choice, noting that mixtures of ethanol and tetrahydrofuran or pure isopropyl acetate can influence the solubility of the substrate and the stability of the catalytic species. By carefully selecting reaction parameters such as temperature, pressure, and catalyst loading, manufacturers can suppress the formation of trace impurities that might otherwise compromise the safety profile of the final drug substance. This mechanistic understanding empowers process engineers to design robust control strategies that maintain product quality within tight specifications, even as the process is scaled up to commercial volumes.
How to Synthesize NEP Inhibitor Intermediate Efficiently
The synthesis of these high-value intermediates involves a streamlined sequence that begins with the preparation of the unsaturated lactam or acid precursor, followed by the critical stereoselective hydrogenation step. Detailed protocols within the patent describe the preparation of catalyst solutions, the handling of air-sensitive reagents, and the specific workup procedures required to isolate the product in high purity. Operators are guided through the precise addition of hydrogen gas under controlled pressure, typically ranging from 20 to 80 bar, to ensure complete conversion without degrading the sensitive functional groups present in the molecule. The following section outlines the standardized operational steps derived from the patent's exemplary embodiments, providing a clear roadmap for implementation in a GMP environment.
- Isomerization of methylene-pyrrolidinone precursors to methyl-pyrrolinone using palladium catalysts.
- Base-mediated ring opening of the lactam structure to form the unsaturated amino acid derivative.
- Stereoselective hydrogenation of the olefinic bond using chiral transition metal complexes to establish the desired stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers substantial strategic benefits that extend far beyond simple technical metrics. The primary advantage lies in the significant reduction of manufacturing costs driven by the elimination of complex purification steps. Because the catalytic system delivers such high diastereoselectivity, the need for expensive chromatographic separations or multiple recrystallizations is drastically minimized, leading to lower solvent consumption and reduced waste disposal costs. This efficiency translates directly into a more competitive cost structure for the final active pharmaceutical ingredient, allowing companies to maintain healthy margins even in price-sensitive markets. Moreover, the ability to use a variety of commercially available ligands and metal precursors reduces the risk of supply disruptions caused by reliance on a single proprietary reagent, ensuring a more stable and predictable sourcing landscape.
Enhanced supply chain reliability is another critical benefit, as the robustness of the catalytic process allows for consistent batch-to-batch performance. The patent demonstrates that the reaction tolerates a range of conditions and substrate concentrations, meaning that minor variations in raw material quality do not necessarily lead to batch failures. This resilience is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers. Additionally, the scalability of the process is inherently supported by the use of standard hydrogenation equipment found in most multipurpose chemical plants, removing the need for specialized capital investment. From an environmental compliance perspective, the atom economy of the hydrogenation reaction is excellent, and the potential for catalyst recycling further aligns with sustainability goals, making this technology an attractive option for companies aiming to reduce their carbon footprint while optimizing operational expenditures.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical implementation and commercial viability of this synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on how this technology can be integrated into existing manufacturing workflows. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this new process for their specific product portfolios.
Q: What represents the key technical breakthrough in this NEP inhibitor synthesis?
A: The patent describes a novel route utilizing specific chiral transition metal catalysts (Rh, Ru, Ir) with ligands like Josiphos or Walphos to achieve high diastereoselectivity during the hydrogenation of the olefinic intermediate, overcoming limitations of prior art methods.
Q: How does this process improve supply chain reliability for API manufacturers?
A: By offering multiple synthetic pathways (isomerization vs. selenation) and robust catalytic conditions, the method reduces dependency on single-source reagents and minimizes the risk of batch failures due to poor stereocontrol.
Q: Can this methodology be scaled for commercial production?
A: Yes, the process utilizes standard hydrogenation equipment and commercially available ligands, making it highly suitable for scale-up from kilogram to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NEP Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing cutting-edge synthetic technologies to stay competitive in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic methods described in CN103080072A can be successfully translated from the laboratory to the plant floor. We are committed to delivering high-purity NEP inhibitor intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle complex chiral syntheses with precision, guaranteeing the consistency and reliability that top-tier pharmaceutical partners demand.
We invite you to collaborate with us to explore how this advanced manufacturing process can optimize your supply chain and reduce your overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in catalytic hydrogenation can become a cornerstone of your successful drug development program.
