Advanced Catalytic Strategy for Oxaziclomefone Production and Commercial Scale-up
Advanced Catalytic Strategy for Oxaziclomefone Production and Commercial Scale-up
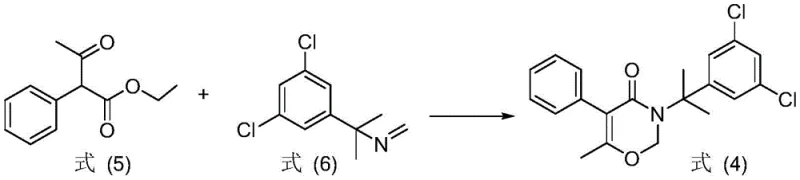
The global demand for high-efficiency paddy field herbicides continues to drive innovation in the synthesis of key active ingredients like oxaziclomefone. A recent technological breakthrough, documented in patent CN108623529B, introduces a significantly optimized preparation method that addresses long-standing inefficiencies in conventional manufacturing routes. This novel approach utilizes a sophisticated catalytic system comprising alcohol, alkali, and free radical polymerization inhibitors to facilitate a direct condensation reaction. By reacting specific keto-ester derivatives with 1-methyl-1-(3,5-dichlorophenyl)ethylamine in the presence of paraformaldehyde, the process achieves superior yield and purity profiles. For R&D directors and procurement specialists, this patent represents a critical opportunity to re-evaluate supply chain strategies for agrochemical intermediates. The method not only shortens the synthetic route but also enhances the stability of raw materials, offering a robust foundation for commercial production. This report provides a deep technical analysis of the mechanistic advantages and the resulting commercial implications for the global agrochemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of oxaziclomefone has been plagued by complex multi-step procedures that introduce significant operational burdens and cost inefficiencies. One prevalent conventional route involves the reaction of 1-ethyl acetophenone with N-methylene-1-methyl-1-(3,5-dichlorophenyl)ethylamine under reflux in xylene. However, this pathway necessitates the prior preparation of the amine intermediate via a reaction with aqueous formaldehyde, creating a disjointed two-step process that switches between aqueous and anhydrous conditions. This transition requires tedious water removal steps, leading to substantial loss of intermediate products and a reduction in the total overall yield. Furthermore, alternative routes relying on the hydrolysis of ethyl 1-acetylphenylacetate using sulfuric acid generate large volumes of acidic wastewater, posing severe environmental compliance challenges. The intermediates in these traditional methods, such as 1-acetylphenylacetic acid, exhibit poor structural stability, which complicates storage and handling while increasing the risk of impurity formation during synthesis.
The Novel Approach
In stark contrast to the fragmented traditional methods, the novel approach disclosed in the patent streamlines the entire synthesis into a more cohesive and efficient operation. By utilizing paraformaldehyde as a stable source of formaldehyde and introducing a tailored catalyst system, the reaction bypasses the need for unstable intermediates and harsh acidic conditions. The new method allows for the direct reaction of formula (1) or (2) compounds with the amine substrate in a single pot under anhydrous conditions. This consolidation eliminates the need for intermediate isolation and the associated solvent switches that typically degrade process efficiency. The use of paraformaldehyde with a polymerization degree n of 2 to 100 ensures a controlled release of formaldehyde, which minimizes side reactions. Consequently, this route not only simplifies the production process but also significantly improves the stability of the reaction system, making it far more suitable for large-scale industrial application where consistency and safety are paramount.
Mechanistic Insights into One-Pot Condensation Catalysis
The core innovation of this synthesis lies in the carefully balanced catalytic system that drives the condensation reaction while suppressing degradation pathways. The catalyst mixture, typically comprising an alcohol, a base, and a free radical polymerization inhibitor, plays a multifaceted role in the reaction mechanism. The alcohol component, such as ethanol or methanol, acts as a co-solvent and proton source that facilitates the formation of the reactive iminium species from the amine and paraformaldehyde. Simultaneously, the base, which can be an organic amine like triethylamine or an inorganic carbonate, deprotonates the active methylene group of the keto-ester, enabling nucleophilic attack. Crucially, the inclusion of a radical polymerization inhibitor, such as sodium sulfide or hydroquinone, prevents the polymerization of reactive intermediates at the elevated temperatures required for reflux. This inhibition is vital for maintaining high selectivity and preventing the formation of tarry by-products that would otherwise complicate downstream purification.
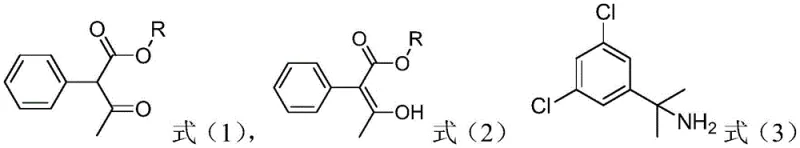
Impurity control is another critical aspect where the new mechanism offers distinct advantages over prior art. In traditional acid-catalyzed routes, the harsh conditions often lead to the hydrolysis of ester groups or the decomposition of sensitive functional groups, resulting in a complex impurity spectrum that is difficult to separate. The new anhydrous basic conditions are much milder on the molecular structure, preserving the integrity of the ester and ketone functionalities until the cyclization occurs. The stability of the starting materials, specifically the C1-C5 alkyl esters, ensures that the reaction proceeds with high atom economy. By avoiding the generation of unstable intermediates like 1-acetylphenylacetic acid, the process inherently reduces the burden on purification steps. This mechanistic robustness translates directly into a higher purity profile for the final oxaziclomefone product, which is a key metric for R&D directors evaluating the feasibility of a new supply source for regulatory filings.
How to Synthesize Oxaziclomefone Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the catalytic system. The process begins with the mixing of paraformaldehyde, the selected alcohol, base, and inhibitor in a suitable organic solvent such as xylene or toluene. This mixture is heated under reflux to activate the catalyst system before the addition of the amine and keto-ester substrates. The reaction temperature is maintained between 110-180°C for a duration of 4 to 24 hours, allowing sufficient time for the condensation and cyclization to reach completion. Detailed standard operating procedures regarding specific molar ratios and work-up protocols are essential for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent data to ensure optimal yield and quality.
- Mix paraformaldehyde, alcohol, base, and radical inhibitor in a solvent like xylene and heat under reflux.
- Add 1-methyl-1-(3,5-dichlorophenyl)ethylamine and the keto-ester substrate to the reaction mixture.
- Maintain reflux temperature between 110-180°C for 4 to 24 hours, then isolate product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers substantial strategic benefits beyond mere technical superiority. The simplification of the synthetic route directly correlates to a reduction in manufacturing complexity, which lowers the operational expenditure associated with labor, energy, and equipment utilization. By eliminating the need for multi-step intermediate preparations and the handling of corrosive acids, the facility requirements become less stringent, allowing for more flexible production scheduling. The use of commercially available and stable raw materials mitigates the risk of supply disruptions caused by the scarcity of specialized intermediates. This stability ensures a more predictable production timeline, which is crucial for meeting the just-in-time delivery demands of global agrochemical companies. Furthermore, the reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the long-term liability and disposal costs for the manufacturer.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and corrosive acids removes the need for expensive removal steps and specialized corrosion-resistant equipment. This process intensification leads to significant cost savings by reducing the number of unit operations and the consumption of auxiliary materials. The higher yield achieved means less raw material is wasted per kilogram of final product, directly improving the cost of goods sold. Additionally, the simplified work-up procedure reduces solvent consumption and energy usage during distillation and drying phases. These cumulative efficiencies result in a more competitive pricing structure without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, commodity-grade starting materials such as paraformaldehyde and simple alkyl esters ensures that the supply chain is resilient against market fluctuations. Unlike routes dependent on unstable intermediates that require immediate use or special storage conditions, this method allows for bulk purchasing and inventory management of raw materials. The robustness of the reaction conditions also means that production is less susceptible to minor variations in environmental factors, ensuring consistent output quality. This reliability is essential for maintaining continuous supply to downstream formulators and preventing stockouts in the agricultural season.
- Scalability and Environmental Compliance: The one-pot nature of the reaction facilitates easier scale-up from pilot plant to commercial production volumes without the need for complex re-engineering of the process flow. The absence of acidic wastewater significantly reduces the burden on effluent treatment plants, making the process more environmentally sustainable. This compliance with green chemistry principles enhances the corporate social responsibility profile of the supply chain. The ability to scale efficiently ensures that the manufacturer can respond rapidly to increases in market demand, providing a secure source of high-purity agrochemical intermediates for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxaziclomefone synthesis method. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into existing supply chains. The responses cover aspects of catalyst selection, reaction safety, and product quality assurance.
Q: Why is the new catalytic system superior to traditional sulfuric acid hydrolysis?
A: The new method eliminates the need for large amounts of sulfuric acid, thereby avoiding the generation of significant acidic wastewater and removing the instability issues associated with 1-acetylphenylacetic acid intermediates.
Q: How does the radical polymerization inhibitor impact product purity?
A: The inclusion of inhibitors like Na2S or hydroquinone prevents unwanted side reactions and polymerization during the high-temperature reflux, ensuring a cleaner impurity profile and higher isolated yield.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses commercially available raw materials with good stability and simplifies the operation from multi-step aqueous/anhydrous switches to a single anhydrous reflux, facilitating easier scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaziclomefone Supplier
The technical potential of this optimized synthesis route underscores the importance of partnering with a CDMO expert capable of executing complex chemical transformations with precision. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in actual manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of oxaziclomefone meets the high standards required by global regulatory bodies. We understand the critical nature of agrochemical supply chains and are committed to delivering consistent quality and reliability.
We invite procurement leaders to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how this method compares to your current sourcing strategy. We encourage you to contact us for specific COA data and route feasibility assessments to validate the commercial viability for your specific application. Our team is ready to provide the technical support and capacity needed to secure your supply of high-purity agrochemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
