Advanced Synthesis of Air-Stable Trialkyl Phosphine Borofluoride for Catalytic Applications
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the demand for safer, more stable, and easily handleable catalytic ligands. Patent CN103012477A introduces a robust preparation method for trialkyl phosphine borofluoride, addressing critical pain points associated with traditional phosphine synthesis. This innovation shifts the paradigm from handling pyrophoric and malodorous free phosphines to generating air-stable borofluoride salts that retain high reactivity upon deprotection. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal advancement in securing reliable supply chains for complex catalytic intermediates. The methodology leverages a Grignard-based approach coupled with phosphorus tribromide, offering a distinct advantage over legacy chlorination routes by minimizing toxicity risks and enhancing product shelf-life.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trialkyl phosphines has relied heavily on the reaction of phosphorus trichloride with Grignard reagents, a process fraught with significant operational and safety hazards. Phosphorus trichloride is a highly toxic and corrosive substance, subjecting manufacturers to stringent regulatory controls and complicating procurement logistics. Furthermore, the resulting free trialkyl phosphines are notoriously unstable in air, rapidly oxidizing to phosphine oxides which render them useless for catalytic applications. This sensitivity necessitates rigorous inert atmosphere handling throughout the entire lifecycle, from synthesis to storage, drastically increasing infrastructure costs. Additionally, the conventional process generates substantial amounts of hazardous waste and emits noxious odors, creating environmental compliance challenges that modern green chemistry initiatives strive to eliminate. The inability to store these intermediates long-term also disrupts supply chain continuity, forcing just-in-time production models that are vulnerable to bottlenecks.
The Novel Approach
The novel approach detailed in the patent data circumvents these issues by utilizing phosphorus tribromide (PBr3) in conjunction with alkyl Grignard reagents at controlled low temperatures. This substitution not only mitigates the extreme toxicity associated with PCl3 but also facilitates a cleaner reaction profile with fewer side products. Crucially, the process incorporates an immediate salification step using fluoroboric acid (HBF4), converting the sensitive trialkyl phosphine into a thermodynamically stable borofluoride salt. This transformation effectively locks the phosphine in a non-oxidizable state, allowing for ambient storage and transport without degradation. The ability to isolate the product as a stable solid via recrystallization further simplifies downstream processing, eliminating the need for complex distillation of oily, air-sensitive liquids. This strategic shift from unstable oils to stable salts fundamentally alters the economic and logistical feasibility of producing high-purity phosphine ligands for industrial catalysis.
Mechanistic Insights into Grignard-Mediated Phosphination and Salification
The core of this synthetic strategy lies in the nucleophilic attack of the alkyl Grignard reagent on the phosphorus center of phosphorus tribromide. Under strictly anhydrous conditions and low temperatures ranging from -10°C to 25°C, the Grignard species adds sequentially to the phosphorus atom, displacing bromide ions to form the trialkyl phosphine intermediate. The use of tetrahydrofuran (THF) as the solvent is critical here, as it effectively solvates the magnesium cation, enhancing the nucleophilicity of the carbanion and ensuring homogeneous reaction kinetics. Following the formation of the phosphine, the reaction mixture is treated with fluoroboric acid, which acts as a proton source and a source of the tetrafluoroborate anion. This step protonates the phosphine to form a phosphonium cation, which immediately pairs with the non-coordinating BF4- anion to precipitate the stable salt. This mechanistic pathway ensures that the reactive phosphine never exists in a free state for extended periods, thereby preventing oxidative degradation.
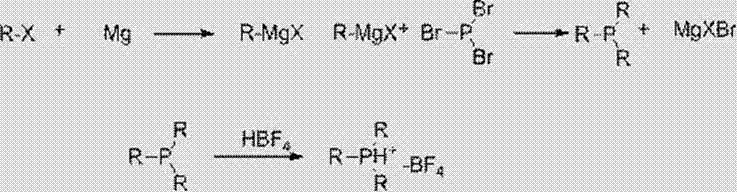
Impurity control is inherently built into this mechanism through the physical properties of the intermediates and the final salt. The initial reaction with PBr3 is highly selective, and any unreacted Grignard reagent is quenched during the aqueous workup with ammonium chloride. The subsequent extraction steps utilize solvents like methyl tert-butyl ether (MTBE) or dichloromethane to separate the organic phosphine species from inorganic magnesium salts. The final recrystallization from ethanol and MTBE serves as a powerful purification tool, leveraging the differential solubility of the borofluoride salt versus organic impurities. This multi-stage purification ensures that the final product meets stringent purity specifications required for pharmaceutical and electronic applications, typically achieving purities exceeding 97% as verified by NMR analysis. The robustness of this mechanism allows for scalability without compromising the integrity of the phosphorus-carbon bonds.
How to Synthesize Trialkyl Phosphine Borofluoride Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of the Grignard reagent, followed by its slow addition to the phosphorus tribromide solution under inert gas protection. Temperature management is paramount during this exothermic phase to prevent runaway reactions. Once the phosphine intermediate is formed, the addition of fluoroboric acid must be conducted with care to manage gas evolution and heat. The subsequent workup involves liquid-liquid extraction to remove inorganic byproducts, followed by solvent recovery and recrystallization to isolate the final solid product. For a detailed breakdown of the specific operational parameters, stoichiometry, and safety protocols required for laboratory and pilot-scale execution, please refer to the standardized synthesis guide below.
- Prepare the alkyl Grignard reagent by reacting alkyl halides with magnesium in THF under inert atmosphere.
- React the Grignard reagent with phosphorus tribromide (PBr3) at low temperatures (-10 to 25°C) to form the trialkyl phosphine intermediate.
- Treat the intermediate with fluoroboric acid (HBF4) to form the stable borofluoride salt, followed by extraction and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. By transitioning to a process that yields air-stable solids, companies can drastically simplify their warehousing and logistics requirements, eliminating the need for specialized inert-gas storage facilities. The ability to recycle organic solvents such as THF and MTBE directly within the process loop significantly reduces raw material consumption and waste disposal costs, aligning with sustainability goals. Furthermore, the use of phosphorus tribromide, while still requiring careful handling, presents a lower regulatory burden compared to phosphorus trichloride, streamlining procurement approvals. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding to market demands without the fragility associated with handling pyrophoric materials.
- Cost Reduction in Manufacturing: The elimination of expensive and complex inert-atmosphere storage infrastructure for the final product leads to substantial capital expenditure savings. Additionally, the direct recycling of extraction solvents minimizes the volume of fresh solvent required per batch, driving down variable production costs. The higher selectivity of the PBr3 route reduces the formation of hard-to-remove impurities, lowering the cost associated with extensive purification steps and improving overall process efficiency.
- Enhanced Supply Chain Reliability: Converting air-sensitive liquids into stable crystalline salts extends the shelf life of the product indefinitely under normal conditions. This stability allows manufacturers to build strategic inventory buffers, mitigating the risk of supply disruptions caused by production delays or transportation bottlenecks. It also broadens the range of qualified logistics providers, as the material no longer requires hazardous Class 4.2 (pyrophoric) shipping classifications, facilitating smoother global distribution.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to kilogram scales with consistent yields. The reduction in hazardous waste generation, particularly the avoidance of chlorinated byproducts associated with PCl3 routes, simplifies wastewater treatment and lowers environmental compliance costs. This green chemistry profile makes the technology attractive for production in regions with strict environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of trialkyl phosphine borofluorides. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for integrating this material into your catalytic processes or supply chain strategy effectively.
Q: Why is the borofluoride salt form preferred over free trialkyl phosphines?
A: Free trialkyl phosphines are highly air-sensitive, prone to oxidation, and often possess unpleasant odors. Converting them to borofluoride salts significantly enhances air stability, simplifies storage, and eliminates odor issues, making them safer for industrial handling.
Q: What are the advantages of using PBr3 over PCl3 in this synthesis?
A: Phosphorus tribromide (PBr3) offers a safer reaction profile compared to the highly toxic phosphorus trichloride (PCl3). Additionally, the PBr3 route described in patent CN103012477A allows for easier purification and higher purity of the final phosphine intermediate.
Q: Can the solvents used in this process be recycled?
A: Yes, the process is designed with green chemistry principles in mind. Organic solvents such as THF and MTBE used during extraction and crystallization can be directly recovered and recycled, reducing waste generation and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trialkyl Phosphine Borofluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality phosphine ligands play in the synthesis of advanced pharmaceuticals and functional materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trialkyl phosphine borofluoride meets the exacting standards required for sensitive catalytic applications. Our commitment to quality assurance means that you receive a product that performs consistently, batch after batch, safeguarding the integrity of your downstream synthesis.
We invite you to collaborate with us to optimize your sourcing strategy for these vital intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency and reduce your total cost of ownership.
