Advanced Synthesis of Eperisone Hydrochloride Impurity F for Global Quality Control
The pharmaceutical industry's relentless pursuit of safety and efficacy mandates rigorous quality control, particularly for central muscle relaxants like Eperisone Hydrochloride. Patent CN112390767A introduces a groundbreaking preparation method for Eperisone Hydrochloride Impurity F, a critical reference standard required for validating the purity of the active pharmaceutical ingredient (API). Unlike traditional methods that struggle with regioselectivity, this novel approach leverages a precise Grignard-based synthetic strategy to construct the specific ortho-substituted carbon skeleton with exceptional fidelity. By utilizing 2-bromoethylbenzene as the foundational building block, the process inherently bypasses the formation of unwanted para-isomers, a common bottleneck in aromatic substitution chemistry. This technical breakthrough not only streamlines the synthesis of this complex beta-aminoketone derivative but also establishes a robust framework for producing high-purity reference materials essential for regulatory compliance and batch release testing in global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eperisone and its related impurities has relied heavily on Friedel-Crafts acylation strategies, which present significant challenges for producing specific ortho-substituted impurities. In the conventional pathway, ethylbenzene is reacted with propionyl chloride in the presence of Lewis acids like aluminum chloride. While effective for generating the para-substituted API, this electrophilic aromatic substitution is governed by steric and electronic factors that favor the para-position, making the isolation of the ortho-isomer (Impurity F) extremely inefficient. The resulting reaction mixture typically contains a complex array of isomers that require laborious and costly chromatographic separations to isolate the trace ortho-impurity. Furthermore, the harsh acidic conditions often lead to side reactions and polymerization, reducing overall yield and complicating the purification profile. Consequently, relying on the main synthesis line to harvest Impurity F is practically unfeasible for generating the gram-to-kilogram quantities needed for comprehensive quality control and stability studies.
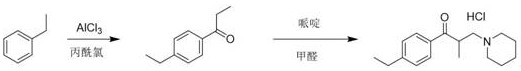
The Novel Approach
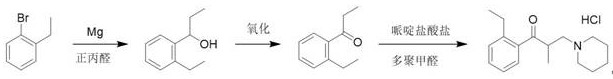
In stark contrast, the methodology disclosed in CN112390767A adopts a convergent strategy that builds the carbon skeleton from a pre-functionalized aromatic precursor. By initiating the synthesis with 2-bromoethylbenzene, the ortho-ethyl group is locked in place before the ketone functionality is introduced, effectively solving the regioselectivity problem at the source. The process proceeds through a controlled Grignard addition to n-propionaldehyde, followed by a mild oxidation step to generate the key intermediate, 2-ethyl propiophenone. This intermediate then undergoes a Mannich condensation with paraformaldehyde and piperidine hydrochloride to install the aminomethyl side chain. This route is not only chemically elegant but also operationally superior, as it avoids the generation of isomeric byproducts entirely. The final product is obtained through a straightforward crystallization process, yielding a reference standard of exceptional purity without the need for preparative HPLC or column chromatography.
Mechanistic Insights into Grignard Addition and Mannich Condensation
The core of this synthetic innovation lies in the precise execution of the Grignard reaction and the subsequent oxidative transformation. The formation of the Grignard reagent from 2-bromoethylbenzene requires careful thermal management, typically initiated with iodine at temperatures between -5°C and 5°C to prevent Wurtz-type coupling side reactions. Once formed, the organomagnesium species attacks the carbonyl carbon of n-propionaldehyde with high nucleophilicity, establishing the secondary alcohol center with the correct stereochemical potential. Following the quench, the oxidation step is critical; the patent highlights the versatility of using Swern oxidation conditions (DMSO activated by oxalyl chloride or trifluoroacetic anhydride) or solid-supported reagents like PCC. These oxidation protocols operate under mild conditions that preserve the sensitive aromatic ring and prevent over-oxidation or degradation of the alkyl side chains, ensuring the integrity of the 2-ethyl propiophenone intermediate prior to the final amination step.
Following the formation of the ketone, the Mannich reaction serves as the final assembly step to construct the beta-aminoketone pharmacophore characteristic of Eperisone derivatives. This reaction involves the in situ generation of an iminium ion from paraformaldehyde and piperidine hydrochloride, which then acts as a potent electrophile. The enolizable alpha-carbon of the 2-ethyl propiophenone attacks this iminium species, forging the carbon-nitrogen bond. The use of isopropanol as a solvent at elevated temperatures (90-100°C) facilitates this condensation while maintaining solubility of the reactants. Crucially, the reaction conditions are tuned to minimize poly-amination or degradation, allowing the crude product to be purified simply by adjusting pH and performing a recrystallization from an isopropanol-acetone mixture. This mechanistic control ensures that the final Impurity F structure matches the theoretical formula exactly, providing a reliable benchmark for analytical validation.
How to Synthesize Eperisone Hydrochloride Impurity F Efficiently
The synthesis of this critical reference standard is designed for operational simplicity, moving away from complex multi-step protections towards a direct linear sequence. The process begins with the activation of magnesium metal in anhydrous tetrahydrofuran, followed by the slow addition of the bromo-starting material to generate the Grignard reagent. Subsequent addition of n-propionaldehyde yields the alcohol intermediate, which is isolated via extraction and concentration. The oxidation step converts this alcohol to the corresponding ketone, which is then subjected to the Mannich conditions with piperidine salts. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility across different laboratory settings.
- Preparation of Grignard Reagent: React 2-bromoethylbenzene with magnesium metal in anhydrous ether or THF, initiated by iodine at low temperatures (-5 to 5°C).
- Oxidation to Ketone: Convert the intermediate alcohol, 1-(2-ethylphenyl)propanol, into 2-ethyl propiophenone using oxidants like Swern reagents or PCC.
- Mannich Condensation: React the ketone with paraformaldehyde and piperidine hydrochloride in isopropanol at 90-100°C, followed by crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the purification train. By eliminating the need for preparative chromatography—a technique that is notoriously difficult to scale and expensive to operate due to solvent consumption and resin costs—the manufacturing process becomes significantly more cost-effective. The reliance on standard unit operations such as liquid-liquid extraction, distillation, and crystallization means that the production of this impurity standard can be seamlessly integrated into existing multipurpose reactor suites without requiring specialized equipment. This compatibility reduces capital expenditure barriers and accelerates the time-to-market for supplying certified reference materials to pharmaceutical clients who are under pressure to validate their API batches against stringent regulatory guidelines.
- Cost Reduction in Manufacturing: The economic viability of producing high-purity impurities is often compromised by low yields and expensive purification methods. This novel route addresses both issues by ensuring high regioselectivity from the first step, thereby maximizing the yield of the desired ortho-isomer. The elimination of chromatographic purification steps results in substantial savings on silica gel, eluents, and processing time. Furthermore, the starting materials, such as 2-bromoethylbenzene and n-propionaldehyde, are commodity chemicals available from multiple global suppliers, mitigating the risk of raw material price volatility. The overall process efficiency translates directly into a lower cost of goods sold (COGS), allowing suppliers to offer competitive pricing for these high-value reference standards while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for pharmaceutical manufacturers who cannot afford interruptions in their quality control testing. The robustness of this synthetic route, characterized by its tolerance to standard industrial conditions and the use of stable intermediates, ensures consistent batch-to-batch quality. The ability to produce the impurity in high purity (>99%) through simple crystallization reduces the risk of batch rejection due to specification failures. Additionally, the modular nature of the synthesis allows for flexible production scheduling; the key ketone intermediate can be stockpiled and converted to the final impurity on demand, providing a buffer against sudden spikes in demand for Eperisone quality testing. This flexibility enhances the resilience of the supply chain against external disruptions.
- Scalability and Environmental Compliance: As regulatory bodies increasingly scrutinize the environmental footprint of pharmaceutical manufacturing, the green chemistry attributes of this process become a significant asset. The avoidance of heavy metal catalysts (often used in alternative coupling reactions) and the minimization of solvent waste through efficient crystallization protocols align with modern sustainability goals. The process is inherently scalable from gram-scale laboratory synthesis to multi-kilogram commercial production without encountering the heat transfer or mixing limitations often associated with exothermic Friedel-Crafts reactions. This scalability ensures that suppliers can meet the growing global demand for impurity standards as more generic versions of Eperisone enter the market, all while adhering to strict environmental, health, and safety (EHS) regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Eperisone Hydrochloride Impurity F. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for R&D and quality assurance professionals seeking to implement this standard in their workflows.
Q: Why is the Grignard route preferred over Friedel-Crafts for this impurity?
A: The Grignard route starts with 2-bromoethylbenzene, which already possesses the required ortho-substitution pattern. This eliminates the difficult separation of ortho/para isomers inherent in Friedel-Crafts acylation of ethylbenzene, ensuring higher regio-purity.
Q: What is the achieved purity of the final impurity standard?
A: The patented process utilizes a specific crystallization protocol using isopropanol and acetone, achieving a final purity of greater than 99%, making it suitable as a certified reference substance.
Q: Is this process scalable for industrial reference material production?
A: Yes, the process avoids complex chromatographic separations. It relies on standard unit operations like extraction, distillation, and crystallization, which are easily transferable from laboratory to pilot and commercial scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eperisone Hydrochloride Impurity F Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your pharmaceutical products depends on the quality of your reference standards. Our team of expert chemists has extensively analyzed the synthetic pathways described in CN112390767A and possesses the technical capability to replicate and optimize this route for commercial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need milligram quantities for method development or kilogram batches for routine QC, we can deliver. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications using advanced analytical techniques such as HPLC, NMR, and MS, guaranteeing that every vial of Impurity F meets the highest international standards.
We invite pharmaceutical manufacturers and CROs to collaborate with us to secure a stable supply of this critical impurity standard. By leveraging our optimized manufacturing processes, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, helping you reduce overall quality control costs. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments for related analogues, and establish a partnership that ensures the continued safety and efficacy of your Eperisone formulations.
