Optimizing Beta-Nicotinamide Mononucleotide Synthesis: A Technical Breakthrough for Industrial Scale-Up
The global demand for beta-nicotinamide mononucleotide (NMN), a pivotal precursor in the biosynthesis of nicotinamide adenine dinucleotide (NAD+), has surged due to its profound implications in anti-aging research and metabolic health. However, the industrial realization of this potential has been historically constrained by the limitations of existing synthesis technologies. Patent CN111253448B addresses these critical bottlenecks by disclosing a sophisticated preparation and purification method that fundamentally alters the economic and technical feasibility of NMN production. Unlike traditional biological synthesis which suffers from high enzyme costs and limited scalability, or standard chemical routes plagued by complex purification and low yields, this invention introduces a novel intermediate purification strategy. By focusing on the refinement of the furanose substrate prior to the critical phosphorylation step, the technology achieves a dramatic enhancement in both total yield and product purity. This report analyzes the technical merits of this approach, offering strategic insights for R&D directors seeking robust synthetic routes and procurement managers aiming for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of beta-nicotinamide mononucleotide has been hindered by the inherent complexity of the phosphorylation reaction. In standard protocols, the furanose substrate often contains a myriad of impurities, including unreacted starting materials, residual solvents like methanol and ammonia, and side products from previous condensation steps. When this crude mixture is subjected directly to phosphorylation agents such as phosphorus oxychloride, these impurities participate in competitive reactions. This leads to a chaotic reaction environment where the selectivity for the desired beta-anomer is compromised, resulting in a proliferation of byproducts. Consequently, the downstream purification becomes arduous, often requiring extensive resin column chromatography which drastically reduces the overall recovery rate. Data from comparative studies indicates that without intermediate purification, the total yield can stagnate around 61%, with the final product containing significant levels of impurities that complicate regulatory approval for pharmaceutical applications.
The Novel Approach
The breakthrough detailed in the patent lies in the decoupling of the synthesis and purification phases through a specialized extraction protocol. Instead of rushing to phosphorylate the crude furanose intermediate, the process first subjects the material to a rigorous biphasic extraction. Initially, a water-oil extraction removes non-polar contaminants, retaining the target intermediate in the aqueous phase. Subsequently, and most critically, the aqueous phase is extracted using a phosphorylation auxiliary agent, such as triethyl phosphate. This step selectively transfers the furanose intermediate into the phosphate phase, effectively isolating it from water-soluble impurities. 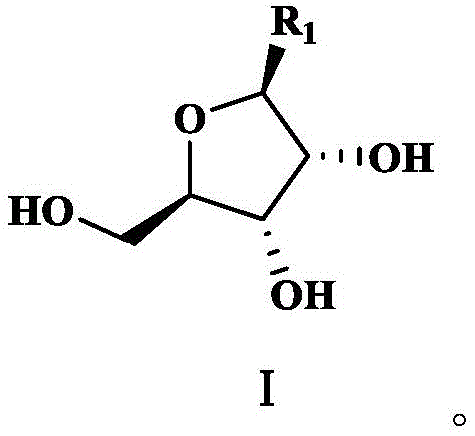 This purified intermediate, now dissolved in a medium compatible with the next reaction step, undergoes phosphorylation with significantly higher efficiency. As demonstrated in the patent's experimental data, this method elevates the content of beta-nicotinamide mononucleotide in the crude product to over 89% and boosts the total yield to approximately 73-75%.
This purified intermediate, now dissolved in a medium compatible with the next reaction step, undergoes phosphorylation with significantly higher efficiency. As demonstrated in the patent's experimental data, this method elevates the content of beta-nicotinamide mononucleotide in the crude product to over 89% and boosts the total yield to approximately 73-75%. 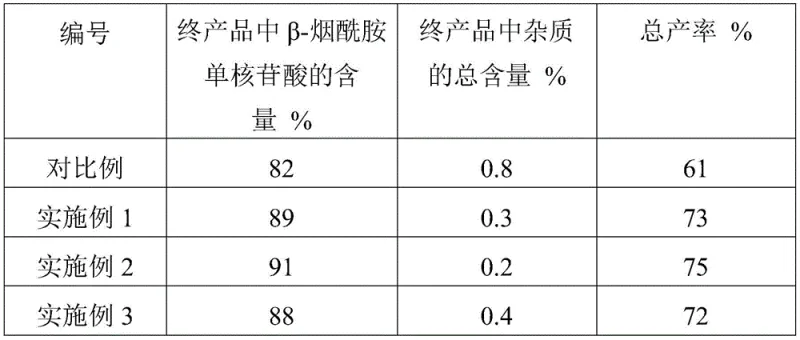 This represents a paradigm shift from reactive purification to proactive impurity management.
This represents a paradigm shift from reactive purification to proactive impurity management.
Mechanistic Insights into Biphasic Extraction and Phosphorylation
The core mechanism driving the success of this synthesis route is the differential solubility and partitioning behavior of the furanose intermediate within a specifically designed solvent system. The intermediate, characterized by the structure in Formula I where R1 can be acetyl, bromo, or nicotinamide groups, possesses amphiphilic properties that are exploited during the extraction. In the first stage, the use of solvents like dichloromethane or ethyl acetate against water ensures that hydrophobic impurities, such as excess tetraacetyl ribose or ethyl nicotinate, are sequestered in the organic layer, while the more polar, partially deprotected sugar intermediate remains in the water.  The second stage is the mechanistic masterpiece: the introduction of the phosphorylation auxiliary agent. Agents like trimethyl phosphate or triethyl phosphate have a high affinity for the specific hydroxyl configurations of the furanose ring. When mixed with the aqueous phase, these phosphate esters act as a 'magnetic' phase for the target molecule, pulling it out of the water and leaving behind inorganic salts and highly polar degradation products. This pre-concentration not only purifies the substrate but also pre-solvates it in a medium that is chemically inert yet perfectly suited for the subsequent nucleophilic attack by phosphorus oxychloride.
The second stage is the mechanistic masterpiece: the introduction of the phosphorylation auxiliary agent. Agents like trimethyl phosphate or triethyl phosphate have a high affinity for the specific hydroxyl configurations of the furanose ring. When mixed with the aqueous phase, these phosphate esters act as a 'magnetic' phase for the target molecule, pulling it out of the water and leaving behind inorganic salts and highly polar degradation products. This pre-concentration not only purifies the substrate but also pre-solvates it in a medium that is chemically inert yet perfectly suited for the subsequent nucleophilic attack by phosphorus oxychloride.
Furthermore, the control of impurities is rigorously maintained during the phosphorylation step itself. By ensuring the substrate is free from ammonia and methanol before the addition of POCl3, the process eliminates the formation of phosphoramidates or methyl phosphates, which are notoriously difficult to separate from the final nucleotide. The reaction is conducted at cryogenic temperatures, typically between -15°C and -10°C. This thermal control is vital for kinetic selectivity; it suppresses the hydrolysis of the acid chloride and prevents the degradation of the sensitive glycosidic bond. The result is a reaction profile that favors the formation of the desired 5'-monophosphate with minimal formation of cyclic phosphates or pyrophosphates. This mechanistic precision translates directly into a cleaner crude product, reducing the burden on the final resin elution step and ensuring that the 'commercial scale-up of complex pharmaceutical intermediates' is not hampered by purification bottlenecks.
How to Synthesize Beta-Nicotinamide Mononucleotide Efficiently
The implementation of this synthesis route requires precise adherence to the extraction ratios and temperature profiles outlined in the patent to replicate the high yields observed in the laboratory. The process begins with the preparation of the raw furanose substrate, typically derived from the condensation of nicotinic acid esters and tetraacetyl ribose, followed by deacetylation. Once this crude intermediate is obtained, the critical purification sequence described above must be executed. Operators must ensure that the mass ratio of the water phase to the intermediate is maintained between 8:1 and 15:1 to ensure complete dissolution and effective partitioning. Similarly, the volume of the phosphorylation auxiliary agent must be optimized to maximize extraction efficiency without diluting the reaction mixture excessively for the subsequent step. The detailed standardized synthesis steps, including specific mixing times and separation protocols, are provided in the guide below to assist technical teams in replicating this high-efficiency pathway.
- Perform a biphasic extraction of the raw furanose substrate intermediate using a water phase and an oil phase (e.g., dichloromethane) to isolate the target compound in the aqueous layer.
- Extract the aqueous phase containing the intermediate using a phosphorylation auxiliary agent (such as triethyl phosphate) to transfer the compound into the organic phosphate phase, effectively purifying it.
- Conduct the phosphorylation reaction by adding phosphorus oxychloride to the purified phosphate phase at low temperatures (-15°C to -10°C) to generate the final beta-nicotinamide mononucleotide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic advantages beyond mere technical elegance. The primary value driver is the significant improvement in process yield. By elevating the total yield from the baseline of roughly 61% to over 73%, the process drastically reduces the consumption of raw materials per kilogram of final product. This efficiency gain directly correlates to a reduction in the cost of goods sold (COGS), making the final NMN product more competitive in the volatile nutraceutical and pharmaceutical markets. Furthermore, the ability to recycle solvents such as dichloromethane and the phosphorylation auxiliary agents contributes to a leaner manufacturing footprint. The elimination of complex protection and deprotection sequences, which are common in alternative chemical routes, simplifies the bill of materials and reduces the dependency on exotic reagents that might face supply chain disruptions.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the purification workflow. Traditional methods often require multiple chromatographic columns or crystallization steps to remove the complex mixture of byproducts generated during uncontrolled phosphorylation. By pre-purifying the intermediate, the downstream processing is streamlined to a simple resin elution. This reduction in unit operations lowers energy consumption, reduces labor hours, and minimizes the loss of product during handling. Additionally, the high atom economy achieved by avoiding unnecessary functional group protection means that less waste is generated, lowering the costs associated with waste disposal and environmental compliance.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route enhances supply chain stability by relying on commodity chemicals rather than specialized biological enzymes or scarce catalysts. The raw materials, such as tetraacetyl ribose and ethyl nicotinate, are widely available from established chemical suppliers, reducing the risk of raw material shortages. Moreover, the process tolerance demonstrated in the examples—where variations in solvent choice (dichloromethane, ethyl acetate, toluene) still yield high-quality results—provides flexibility in sourcing. This adaptability ensures that production schedules remain uninterrupted even if specific solvent grades face temporary market constraints, securing a consistent flow of high-purity intermediates.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is designed for industrial viability. The use of biphasic extractions is a unit operation that scales linearly from laboratory glassware to large-scale industrial reactors without the engineering challenges associated with enzymatic bioreactors. The ability to recover and reuse the phosphorylation auxiliary agent and organic solvents aligns with green chemistry principles, reducing the facility's environmental footprint. This compliance with stringent environmental standards facilitates faster regulatory approvals and reduces the risk of production halts due to environmental audits, ensuring long-term business continuity for partners investing in this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NMN synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for R&D and quality assurance teams evaluating the process feasibility.
Q: How does the new purification method improve NMN yield compared to conventional chemical synthesis?
A: The patented method introduces a critical purification step for the furanose substrate intermediate prior to phosphorylation. By utilizing a specific extraction sequence involving water, oil, and a phosphorylation auxiliary agent, the process removes impurities like unreacted raw materials and ammonia. This pre-purification prevents competitive side reactions during the phosphorylation step, raising the total yield from approximately 61% in conventional methods to over 73%.
Q: What are the specific reaction conditions required for the phosphorylation step in this process?
A: The phosphorylation reaction requires strict temperature control to ensure selectivity and prevent degradation. The process dictates reacting the purified intermediate with phosphorus oxychloride at temperatures between -20°C and -5°C, with an optimal range of -15°C to -10°C. The mass ratio of phosphorus oxychloride to the intermediate is maintained between 1:1 and 2:1, typically around 1.3:1, to ensure complete conversion while minimizing byproduct formation.
Q: Why is the use of a phosphorylation auxiliary agent significant in this synthesis route?
A: The phosphorylation auxiliary agent, such as trimethyl or triethyl phosphate, serves a dual purpose. Firstly, it acts as a highly selective solvent during the extraction phase, pulling the polar furanose intermediate out of the aqueous layer while leaving other impurities behind. Secondly, since it is compatible with the subsequent phosphorylation reagents, it simplifies the workflow by reducing the need for solvent swapping, thereby enhancing atom economy and facilitating solvent recycling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nicotinamide Mononucleotide Supplier
As the global market for NAD+ precursors continues to expand, the ability to produce beta-nicotinamide mononucleotide with high purity and consistent quality is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN111253448B to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial material supply and full-scale commercial manufacturing. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of NMN meets the highest international standards for pharmaceutical and nutraceutical applications.
We invite potential partners to engage with us to explore how this optimized synthesis route can benefit your specific product pipeline. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the technical feasibility of this route for your own facilities, our technical procurement team is ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
