Scalable One-Pot Synthesis of Mopiravir Intermediates for Global API Manufacturing
The global demand for effective antiviral therapeutics has intensified the search for robust, scalable synthetic routes for key nucleoside analogs. Patent CN115785181A introduces a transformative one-pot methodology for the preparation of the critical Mopiravir intermediate, ((3AR,4R,6R,6AR)-6-(4-(hydroxylamine)-2-oxazimidine-1(2H)-methyl)-2,2-dimethyltetrahydrofuran[3,4-D][1,3]dioxol-4-yl)methyl isobutyrate. This compound serves as a pivotal precursor in the manufacturing of Mopiravir, an oral antiviral agent demonstrating superior efficacy against SARS-CoV-2 compared to existing treatments. The disclosed technology addresses longstanding bottlenecks in nucleoside chemistry by eliminating cumbersome isolation steps and hazardous reagents, offering a pathway to high-purity materials suitable for stringent pharmaceutical applications. 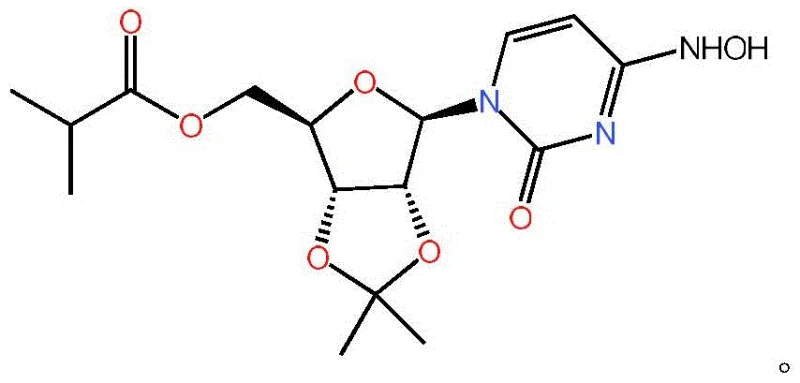
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing this specific nucleoside intermediate have been plagued by significant operational inefficiencies and safety concerns that hinder large-scale adoption. For instance, literature routes often rely on expensive organic bases like DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene) which drastically inflate raw material costs without guaranteeing high crude purity. Furthermore, traditional processes frequently necessitate column chromatography for purification, a technique that is notoriously difficult to scale and generates substantial volumes of hazardous solvent waste. Other patented approaches utilize phosphorus oxychloride, creating severe environmental disposal challenges due to acidic wastewater, and require multiple solvent swaps involving acetonitrile, ethyl acetate, and ether, complicating solvent recovery and increasing the carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the one-pot strategy detailed in CN115785181A streamlines the synthesis into a cohesive, continuous operation that bypasses the isolation of the intermediate acylated species. By utilizing cost-effective triethylamine as the base and employing water-immiscible solvents such as dichloromethane or n-hexane, the process facilitates a unique "reaction-distillation" sequence. This allows the organic solvent to be removed in situ while the reaction temperature is ramped up for the oximation step, effectively switching the reaction medium from organic to aqueous without manual intervention. This innovation not only simplifies the post-reaction workup by removing the need for extraction and chromatography but also significantly enhances the safety profile by stabilizing the hydroxylamine sulfate reagent in an aqueous environment prior to heating. 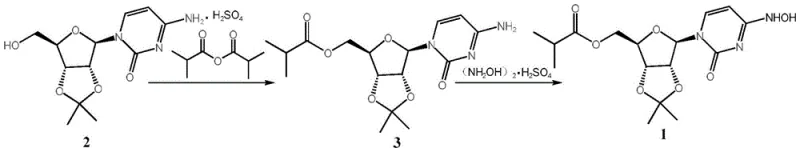
Mechanistic Insights into One-Pot Acylation and Oximation
The core of this technological advancement lies in the precise orchestration of acylation followed immediately by nucleophilic substitution in a changing solvent environment. Initially, the protected cytidine derivative (Compound 2) undergoes acylation with isobutyric anhydride catalyzed by DMAP and triethylamine at mild temperatures ranging from 10°C to 50°C. This step is critical for installing the isobutyrate protecting group which modulates the lipophilicity and metabolic stability of the final drug. Unlike conventional methods that isolate this acylated intermediate, the novel process proceeds directly by introducing water, hydroxylamine sulfate, and sodium acetate into the same vessel. The subsequent heating to 20-90°C serves a dual purpose: it drives the distillation of the low-boiling organic solvent and simultaneously provides the thermal energy required for the conversion of the cytosine base to the N4-hydroxycytosine moiety (Compound 1).
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the exposure of sensitive intermediates to harsh conditions. The use of sodium acetate acts as a buffer, maintaining a pH environment that favors the formation of the desired hydroxylamine product while suppressing side reactions that typically lead to degradation or polymerization. Furthermore, the direct crystallization from the aqueous phase upon cooling leverages the differential solubility of the product versus inorganic salts, effectively purifying the compound without the need for organic recrystallization solvents. This results in a final intermediate with liquid phase purity exceeding 99.1%, providing a robust foundation for the subsequent deprotection step to Mopiravir, which achieves purity levels greater than 99.9%.
How to Synthesize Mopiravir Intermediate Efficiently
The implementation of this one-pot synthesis requires careful attention to solvent selection and temperature profiling to maximize yield and safety. The process begins with the suspension of the starting material in a water-immiscible solvent, followed by the controlled addition of reagents to manage exotherms. The critical transition point occurs when water is added to the reaction mixture containing the acylated intermediate; this creates a biphasic system that enables the safe removal of the organic layer via distillation while retaining the reactants in the aqueous phase for the final transformation. Detailed standardized operating procedures regarding reagent ratios, specifically the mass ratio of hydroxylamine sulfate to the starting material, are essential for reproducibility.
- Acylation Step: React Compound 2 with isobutyric anhydride using DMAP and triethylamine in an organic solvent (e.g., DCM or n-hexane) at 10-50°C to form Compound 3 in situ.
- Oximation and Crystallization: Add water, hydroxylamine sulfate, and sodium acetate directly to the reaction mixture. Heat to 20-90°C to distill off the organic solvent and complete the reaction, then cool to crystallize Compound 1.
- Isolation: Centrifuge the cooled mixture, wash the wet cake with deionized water, and dry to obtain the high-purity Mopiravir intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this one-pot methodology represents a significant opportunity to optimize the cost structure and reliability of the antiviral supply chain. By eliminating the requirement for column chromatography and reducing the number of solvent exchanges, the process inherently lowers the consumption of high-purity organic solvents and silica gel, which are major cost drivers in fine chemical manufacturing. The ability to recover and reuse solvents like dichloromethane or n-hexane directly from the distillation step further compounds these savings, creating a closed-loop system that minimizes waste disposal fees and raw material procurement needs. This efficiency translates directly into a more competitive pricing model for the final API intermediate without compromising on quality standards.
- Cost Reduction in Manufacturing: The replacement of expensive bases like DBU with commodity chemicals such as triethylamine significantly reduces the bill of materials. Additionally, the elimination of multi-step workups, including extractions and chromatographic purifications, reduces labor hours and equipment occupancy time. The process design inherently minimizes the generation of mother liquor and waste streams, leading to substantial cost savings in waste treatment and environmental compliance, thereby enhancing the overall economic viability of large-scale production.
- Enhanced Supply Chain Reliability: The simplified operational workflow reduces the number of potential failure points in the manufacturing process, ensuring higher batch success rates and consistent delivery schedules. The use of stable, commercially available reagents like isobutyric anhydride and hydroxylamine sulfate mitigates the risk of supply disruptions associated with specialized or hazardous catalysts. Furthermore, the robustness of the crystallization step ensures that the physical properties of the intermediate, such as particle size and flowability, are consistent, facilitating smoother downstream processing and packaging operations.
- Scalability and Environmental Compliance: The one-pot nature of the reaction is ideally suited for scale-up in standard stainless steel reactors, avoiding the need for specialized equipment required for handling unstable intermediates or high-vacuum distillations. The reduction in solvent variety simplifies the solvent recovery infrastructure, making it easier to meet stringent environmental regulations regarding volatile organic compound (VOC) emissions. By designing a process that generates less hazardous waste and utilizes safer reaction conditions, manufacturers can achieve better sustainability metrics and reduce their regulatory burden.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing control strategies. The answers provided are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for industrial application.
Q: How does this one-pot method improve safety compared to traditional routes?
A: Traditional methods often use thermally unstable hydroxylamine sulfate in organic solvents like acetonitrile. This patent utilizes a biphasic system where water is added early, stabilizing the hydroxylamine salt and allowing safe distillation of the organic solvent before the high-temperature oximation step.
Q: What are the purity specifications achievable with this process?
A: The process consistently yields the intermediate (Compound 1) with liquid phase purity exceeding 99.1%. Furthermore, the subsequent conversion to Mopiravir achieves purity levels greater than 99.9% with single impurities below 0.1%.
Q: Can the solvents used in this process be recycled?
A: Yes, a key advantage is the use of water-immiscible solvents like dichloromethane or n-hexane. These can be distilled off during the reaction, washed, dried, and directly reused in subsequent batches, significantly reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mopiravir Intermediate Supplier
As the pharmaceutical industry continues to evolve, the need for partners who can navigate complex synthetic challenges while maintaining rigorous quality standards has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced process chemistry expertise to deliver high-value intermediates like the Mopiravir precursor described in CN115785181A. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global clinical and commercial programs. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for antiviral drug substance manufacturing.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of adopting this one-pot technology for your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and reliability in your antiviral drug development pipeline.
