Optimizing Skeletal Muscle Relaxant Intermediates: A Technical Breakthrough in 5 Alpha-Androst-2-Ene-17 Ketone Production
The pharmaceutical industry's demand for high-purity skeletal muscle relaxant intermediates continues to drive innovation in synthetic organic chemistry, particularly for steroidal compounds used in anesthesia and critical care. Patent CN113637043A introduces a significant advancement in the preparation of 5 alpha-androst-2-ene-17 ketone, a pivotal building block for neuromuscular blocking agents such as rocuronium bromide and vecuronium bromide. This novel methodology addresses long-standing challenges regarding isomeric impurity control and process scalability that have plagued previous synthetic routes. By shifting from traditional tosylation to a specialized sulfonate ester strategy, the technology offers a robust pathway to minimize the formation of the difficult-to-separate 5 alpha-androst-3-ene-17-one isomer. 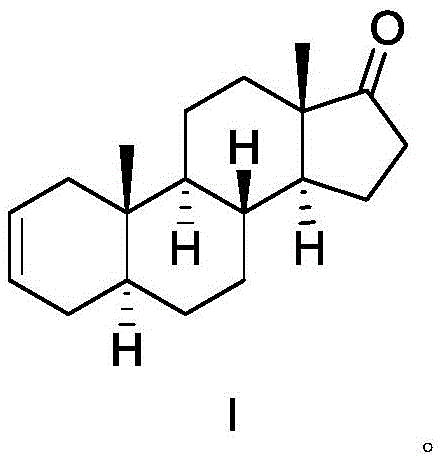 . For R&D directors and procurement specialists, understanding this structural nuance is vital, as the position of the double bond directly dictates the biological activity and downstream synthesis efficiency of the final drug substance. This report analyzes the technical merits and commercial implications of adopting this refined synthesis for reliable pharmaceutical intermediate supplier partnerships.
. For R&D directors and procurement specialists, understanding this structural nuance is vital, as the position of the double bond directly dictates the biological activity and downstream synthesis efficiency of the final drug substance. This report analyzes the technical merits and commercial implications of adopting this refined synthesis for reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5 alpha-androst-2-ene-17 ketone has relied on methods that compromise either yield or purity, creating significant bottlenecks for commercial scale-up of complex pharmaceutical intermediates. One established route involves the reaction of androsterone under severe conditions with boric acid and acetic anhydride at elevated temperatures, which often results in low yields and a complex mixture of byproducts that are difficult to purify. Another common approach utilizes epiandrosterone reacted with p-toluenesulfonyl chloride to form a tosylate, followed by high-temperature elimination; however, literature indicates this method generates approximately 20 percent of the unwanted 5 alpha-androst-3-ene-17-one isomer. This specific isomer possesses similar physical properties to the target molecule, making separation via standard crystallization nearly impossible and often necessitating expensive and time-consuming column chromatography. Furthermore, alternative methods involving zinc powder and trimethylchlorosilane in tetrahydrofuran suffer from incomplete reactions and require extensive purification, rendering them unsuitable for cost reduction in pharmaceutical intermediate manufacturing on an industrial scale.
The Novel Approach
The patented methodology overcomes these deficiencies by introducing p-dodecylbenzene sulfonyl chloride as the sulfonating agent, coupled with a specific elimination protocol using substituted pyridines. This strategic modification in the leaving group structure fundamentally alters the reaction kinetics and thermodynamics during the elimination phase. Instead of the smaller tosylate group, the bulky dodecylbenzene sulfonyl moiety imposes steric constraints that favor the formation of the desired delta-2 double bond over the thermodynamic delta-3 isomer. Experimental data from the patent demonstrates that this approach can achieve yields exceeding 90 percent while drastically reducing the isomeric impurity to levels around 7 percent or lower, depending on the specific base used. This improvement eliminates the need for chromatographic purification, allowing for a streamlined workflow that relies on crystallization and filtration. For supply chain heads, this transition represents a move towards reducing lead time for high-purity pharmaceutical intermediates by simplifying the downstream processing units and minimizing solvent consumption associated with column chromatography.
Mechanistic Insights into Sulfonate Ester Elimination
The core of this technological breakthrough lies in the mechanistic details of the elimination reaction, specifically how the choice of sulfonate ester influences the regioselectivity of the double bond formation. In the initial step, epiandrosterone reacts with p-dodecylbenzene sulfonyl chloride in the presence of catalysts like DMAP and DCC to form a stable sulfonate ester intermediate. This intermediate is then subjected to thermal elimination in solvents such as 2-methylpyridine or 2,6-dimethylpyridine at temperatures between 110°C and 130°C. The bulky nature of the dodecylbenzene sulfonate group likely hinders the abstraction of protons at the C-4 position, which would lead to the undesired delta-3 isomer, thereby kinetically favoring the abstraction at the C-1 position to form the delta-2 olefin. This level of control is critical for R&D directors focusing on purity and impurity profiles, as it ensures that the crude product already meets stringent specifications before final recrystallization. The use of hindered pyridine bases further supports this selectivity by acting as non-nucleophilic bases that facilitate elimination without participating in side reactions.
Impurity control is further enhanced by the crystallization behavior of the intermediate and final products, which is optimized by the specific solvent systems described in the patent. The process utilizes organic solvents like dichloromethane or toluene for the sulfonation step, followed by an aqueous workup and recrystallization from ethanol for the final product. This solvent system is designed to maximize the solubility difference between the target 5 alpha-androst-2-ene-17 ketone and potential byproducts, including unreacted starting materials and isomeric impurities. By avoiding the use of transition metal catalysts or exotic reagents that might leave behind toxic residues, the process also aligns with strict environmental compliance and safety standards required for API intermediate production. The ability to control the crystal lattice formation through controlled cooling rates and solvent composition ensures that the final solid form has consistent physical properties, which is essential for downstream processing in the formulation of skeletal muscle relaxant drugs.
How to Synthesize 5 Alpha-Androst-2-Ene-17 Ketone Efficiently
Implementing this synthesis route requires precise control over reaction parameters to replicate the high yields and purity reported in the patent documentation. The process begins with the activation of epiandrosterone, followed by a carefully monitored elimination step that dictates the final isomeric ratio. Operators must maintain strict temperature controls during the addition of reagents and the subsequent heating phases to prevent thermal degradation or the formation of side products. The following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility and safety in a GMP environment. Detailed standardized synthesis steps are provided in the section below for technical review.
- React epiandrosterone with p-dodecylbenzene sulfonyl chloride using DMAP and DCC catalysts in dichloromethane or toluene to form the sulfonate ester intermediate.
- Perform thermal elimination of the sulfonate ester in 2-methylpyridine or 2,6-dimethylpyridine at 110-130°C to generate the delta-2 double bond.
- Purify the crude product via aqueous acid workup and recrystallization in ethanol to achieve high purity without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial cost savings and supply chain reliability improvements for manufacturers of neuromuscular blocking agents. The primary economic driver is the elimination of column chromatography, which is notoriously expensive due to high solvent usage, silica gel costs, and low throughput. By replacing this batch-intensive process with continuous or semi-continuous crystallization, manufacturers can significantly increase production capacity without proportional increases in capital expenditure or operating costs. This efficiency translates directly into cost reduction in pharmaceutical intermediate manufacturing, allowing suppliers to offer more competitive pricing while maintaining healthy margins. Additionally, the reduced impurity profile minimizes the risk of batch rejection during quality control testing, thereby enhancing overall yield and resource utilization.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps leads to a drastic simplification of the production workflow, reducing both material and labor costs associated with downstream processing. Without the need for large volumes of elution solvents and stationary phases, the environmental footprint of the manufacturing process is also significantly lowered, aligning with green chemistry initiatives. This streamlined approach allows for faster batch turnover times, enabling facilities to respond more agilely to market demand fluctuations. Consequently, the overall cost of goods sold (COGS) for the intermediate is optimized, providing a strategic advantage in price-sensitive global markets.
- Enhanced Supply Chain Reliability: The reagents used in this process, such as p-dodecylbenzene sulfonyl chloride and substituted pyridines, are commercially available and stable, reducing the risk of raw material shortages. Unlike methods relying on specialized catalysts or hard-to-source precursors, this route utilizes commodity chemicals that can be sourced from multiple vendors, ensuring supply continuity. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. For procurement managers, this reliability is crucial for maintaining consistent inventory levels and meeting delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like stirring, heating, and filtration that are easily transferable from pilot plant to full commercial production. The avoidance of heavy metal catalysts and the reduction in hazardous solvent waste simplify waste treatment protocols and regulatory compliance. This makes the technology attractive for manufacturing in regions with strict environmental regulations, expanding the geographical options for production sites. The ability to produce high-purity material consistently at scale ensures that the supply chain can support the growing global demand for skeletal muscle relaxants without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this key steroid intermediate. These answers are derived from the specific technical disclosures and beneficial effects outlined in the patent literature, providing clarity for stakeholders evaluating this technology. Understanding these details is essential for making informed decisions about sourcing and process integration.
Q: How does this method improve impurity profiles compared to traditional tosylation?
A: By utilizing p-dodecylbenzene sulfonyl chloride instead of p-toluenesulfonyl chloride, the steric bulk of the leaving group alters the elimination transition state, significantly suppressing the formation of the unwanted 5 alpha-androst-3-ene-17-one isomer.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process replaces labor-intensive column chromatography with standard crystallization and filtration steps, making it highly scalable and cost-effective for commercial production volumes.
Q: What are the key quality control parameters for this intermediate?
A: Critical QC metrics include the ratio of the delta-2 olefin versus the delta-3 isomer, overall chemical purity via HPLC, and residual solvent levels, all of which are tightly controlled in this optimized route.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5 Alpha-Androst-2-Ene-17 Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5 alpha-androst-2-ene-17 ketone meets the highest industry standards. We are committed to leveraging advanced synthetic technologies, such as the optimized sulfonate elimination route, to deliver superior value to our partners.
We invite you to collaborate with us to explore how this innovative synthesis can enhance your supply chain and reduce your overall production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing goals, ensuring a seamless transition from development to commercial supply.
