Advanced Palladium-Catalyzed Ortho-Deuteration Technology for High-Purity Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Ortho-Deuteration Technology for High-Purity Pharmaceutical Intermediates
The landscape of modern drug discovery is increasingly shifting towards deuterated pharmaceuticals, driven by the need for improved metabolic stability and optimized pharmacokinetic profiles. In this context, the recent disclosure in patent CN113651663A represents a significant technological breakthrough for the synthesis of deuterated aromatic carbonyl compounds. This patent details a novel, one-pot preparation method that utilizes a sophisticated palladium catalytic system to achieve highly selective ortho-deuteration of aromatic aldehydes and ketones. By leveraging a unique combination of palladium catalysts, amino acid ligands, and specific auxiliary agents, this technology overcomes the historical limitations of multi-step syntheses and expensive transition metal catalysts. For R&D directors and procurement specialists alike, this innovation offers a pathway to accessing high-purity deuterated building blocks with enhanced economic efficiency and supply chain reliability.
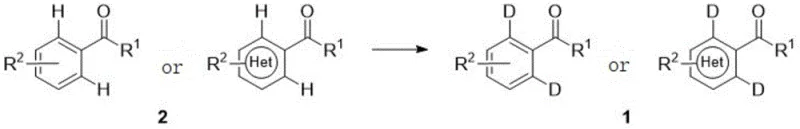
The strategic value of this patent lies in its ability to transform simple aromatic carbonyl derivatives into valuable deuterated intermediates in a single operational step. Traditionally, introducing deuterium atoms at specific positions on an aromatic ring required complex, multi-step sequences starting from deuterated benzene precursors, often resulting in low overall yields and significant waste generation. The method described in CN113651663A streamlines this process by employing a direct hydrogen-deuterium exchange mechanism. This not only reduces the number of unit operations but also minimizes the environmental footprint, aligning perfectly with the green chemistry principles that modern chemical manufacturers strive to uphold. The versatility of the substrate scope, covering various substituted phenyl, naphthyl, and heterocyclic groups, further underscores its potential as a platform technology for diverse medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the preparation of deuterated aromatic carbonyl derivatives was fraught with significant technical and economic challenges. Conventional approaches often relied on classical organic reactions starting from deuterium-substituted benzene, necessitating lengthy multi-step synthetic routes that were both time-consuming and costly. Furthermore, while transition metal-catalyzed hydrogen-deuterium exchange using ruthenium, rhodium, or iridium complexes offered a more direct route, these methods frequently demanded harsh reaction conditions, such as extremely high temperatures or the use of deuterium gas under pressure. The reliance on scarce and expensive noble metals like iridium and rhodium also posed a substantial burden on production costs, making the final deuterated intermediates prohibitively expensive for large-scale applications. Additionally, achieving high regioselectivity, particularly for the ortho-position, remained a persistent challenge, often leading to mixtures of isotopologues that required difficult and yield-loss-inducing separation processes.
The Novel Approach
The methodology presented in CN113651663A fundamentally disrupts these established paradigms by introducing a highly efficient palladium-catalyzed system. This novel approach utilizes a readily available palladium source, such as palladium acetate or palladium chloride, in conjunction with a specifically designed amino acid ligand. The reaction proceeds under an inert atmosphere in a mixture of organic alcohol and strong acid, using heavy water or deuterated acids as the deuterium source. This system enables the direct activation of the ortho-C-H bond adjacent to the carbonyl group, facilitating a clean and selective exchange with deuterium. The process is characterized by its operational simplicity, as it is a one-pot reaction that does not require intermediate solvent swaps or complex workup procedures. The result is a robust, scalable, and cost-effective method that delivers high deuteration degrees and excellent yields across a broad range of substrates, effectively solving the pain points of cost, complexity, and selectivity associated with previous technologies.
Mechanistic Insights into Pd-Catalyzed Ortho-C-H Activation
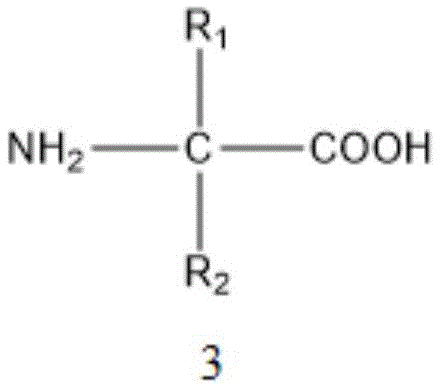
The core of this technological advancement lies in the intricate interplay between the palladium catalyst and the amino acid ligand, which together orchestrate the selective activation of the aromatic C-H bond. The proposed mechanism involves the coordination of the carbonyl oxygen to the palladium center, directing the metal to the ortho-position through a chelation-assisted process. The amino acid ligand, depicted in the patent as Formula 3, plays a critical role in modulating the electronic and steric environment of the palladium center, thereby enhancing its reactivity and selectivity. This ligand assistance is crucial for overcoming the high energy barrier associated with breaking the strong carbon-hydrogen bond on the aromatic ring. The presence of the inorganic oxidant, such as silver trifluoroacetate, facilitates the regeneration of the active palladium species, ensuring the catalytic cycle continues efficiently. Furthermore, the specific solvent system comprising hexafluoroisopropanol (HFIP) and trifluoroacetic acid (TFA) creates a highly polar and acidic environment that stabilizes the cationic palladium intermediates and promotes the protonolysis step necessary for the incorporation of the deuterium atom.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over non-directed deuteration methods. The chelation effect ensures that deuteration occurs predominantly at the ortho-position relative to the carbonyl group, minimizing the formation of meta- or para-deuterated byproducts. This high regioselectivity is paramount for pharmaceutical applications where the position of the deuterium atom can significantly influence the metabolic fate of the drug molecule. The use of mild acidic conditions also helps to suppress side reactions such as polymerization or decomposition of sensitive functional groups that might be present on the aromatic ring. Consequently, the crude reaction mixture is cleaner, simplifying the downstream purification process and leading to higher overall recovery of the desired high-purity product. This level of control over the reaction outcome is a testament to the sophistication of the catalytic system design and provides R&D teams with a reliable tool for synthesizing complex deuterated standards and intermediates.
How to Synthesize Deuterated Aromatic Carbonyl Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol typically involves charging a reaction vessel with the aromatic carbonyl substrate, the palladium catalyst, the amino acid ligand, and the inorganic oxidant under a nitrogen or argon atmosphere. Subsequently, the deuterium source, organic alcohol solvent, and organic strong acid are added, and the mixture is heated to temperatures ranging between 120°C and 150°C. The reaction progress is monitored until completion, after which a straightforward aqueous workup involving dilute acid and organic extraction is performed. The detailed standardized synthesis steps, including specific molar ratios and purification techniques, are outlined in the guide below to ensure reproducibility and optimal results for your specific substrate.
- Combine aromatic carbonyl substrate, palladium catalyst (e.g., Pd(OAc)2), amino acid ligand, and inorganic oxidant in a reaction vessel under inert atmosphere.
- Add the deuterium source (e.g., D2O), organic alcohol solvent (e.g., HFIP), and organic strong acid, then heat the mixture to 120-150°C.
- Upon completion, perform aqueous workup with dilute acid and organic extraction, followed by column chromatography to isolate the pure ortho-deuterated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed deuteration technology translates into tangible strategic benefits that extend beyond mere technical performance. The shift from expensive and scarce noble metals like iridium and rhodium to more abundant and affordable palladium significantly alters the cost structure of deuterated intermediate production. Moreover, the simplification of the process into a one-pot operation reduces labor costs, energy consumption, and waste disposal fees, contributing to a leaner and more sustainable manufacturing model. The robustness of the reaction conditions ensures consistent quality and yield, mitigating the risks of batch failures that can disrupt supply chains. By securing a reliable deuterated aromatic carbonyl supplier who utilizes this advanced technology, pharmaceutical companies can safeguard their development timelines and reduce the overall cost of goods sold for their deuterated drug candidates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as iridium and rhodium, replacing them with cost-effective palladium systems, leads to substantial raw material savings. Additionally, the one-pot nature of the reaction removes the need for intermediate isolation and solvent exchange steps, drastically reducing processing time and utility consumption. This streamlined workflow allows for significant cost reduction in pharmaceutical intermediates manufacturing, making deuterated compounds more accessible for early-stage research and late-stage clinical supply.
- Enhanced Supply Chain Reliability: The reagents required for this process, including palladium salts, amino acids, and heavy water, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The high operational stability of the catalytic system means that production schedules are less likely to be impacted by technical difficulties or sensitivity to minor variations in reaction parameters. This reliability is crucial for reducing lead time for high-purity deuterated aromatic carbonyls, ensuring that critical materials are available when needed for tight drug development timelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple setup and lack of hazardous reagents like deuterium gas under high pressure. The use of heavy water as the primary deuterium source is environmentally benign compared to other deuterating agents, aligning with strict environmental regulations. The ability to scale up complex deuterated aromatics from gram to kilogram quantities without compromising yield or selectivity supports the commercial scale-up of complex pharmaceutical intermediates, facilitating a smooth transition from clinical trials to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuteration technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of this route for your specific project requirements and for engaging in informed discussions with your chemical partners.
Q: What are the advantages of this palladium-catalyzed method over traditional ruthenium or rhodium systems?
A: Unlike traditional methods requiring expensive Ru, Rh, or Ir catalysts and harsh conditions, this patent utilizes a more cost-effective palladium system with amino acid ligands. It operates under milder acidic conditions with high regioselectivity for ortho-position deuteration, significantly simplifying the purification process and reducing raw material costs.
Q: What is the typical deuteration degree achievable with this protocol?
A: According to the experimental data in CN113651663A, the method achieves high deuteration degrees, often exceeding 90% for many substrates (e.g., 91-97% for various benzaldehyde derivatives), ensuring high isotopic purity required for metabolic stability studies in drug development.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the process is designed as a one-pot reaction without the need for intermediate solvent exchanges. The use of commercially available reagents like heavy water and standard palladium salts, combined with robust reaction conditions (120-150°C), makes it highly suitable for scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Aromatic Carbonyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality deuterated intermediates play in the advancement of next-generation therapeutics. Our team of expert chemists has extensively evaluated the technology disclosed in CN113651663A and possesses the capability to implement this palladium-catalyzed protocol with precision and efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise deuteration levels and isotopic distribution, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your upcoming projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this efficient route can optimize your budget without compromising quality. We encourage you to reach out today to request specific COA data for our deuterated catalog or to discuss route feasibility assessments for your custom synthesis needs, ensuring a seamless and successful supply chain partnership.
