Revolutionizing p-Nitrobenzoic Acid Production via Safe Biomimetic Catalytic Oxidation
Revolutionizing p-Nitrobenzoic Acid Production via Safe Biomimetic Catalytic Oxidation
The chemical industry is constantly seeking methods that balance high efficiency with stringent safety and environmental standards. Patent CN100395227C introduces a groundbreaking approach for the preparation of p-nitrobenzoic acid, a critical intermediate in the synthesis of pharmaceuticals, dyes, and agrochemicals. This technology leverages biomimetic catalytic oxygen oxidation of p-nitrotoluene, replacing hazardous traditional solvents with a benign ethanol-water system. By utilizing advanced transition metal catalysts such as metal phthalocyanines and metalloporphyrins, this method achieves exceptional yields under mild conditions. For global procurement teams and R&D directors, this represents a significant shift towards greener, safer, and more cost-effective manufacturing protocols that do not compromise on output quality or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-nitrobenzoic acid via catalytic oxygen oxidation has relied heavily on anhydrous methanol as the primary solvent medium. While effective in dissolving reactants, anhydrous methanol presents severe safety liabilities, particularly when used in conjunction with pure oxygen under pressure. The narrow explosion limits and low flash point of methanol create a high-risk environment for industrial-scale operations, necessitating expensive explosion-proof infrastructure and rigorous safety monitoring. Furthermore, the toxicity of methanol poses long-term health risks to laboratory and plant personnel, requiring extensive personal protective equipment and ventilation systems. These factors collectively drive up operational costs and complicate regulatory compliance, making the conventional methanol-based route increasingly unattractive for modern sustainable manufacturing initiatives.
The Novel Approach
The innovative methodology described in the patent data fundamentally alters the solvent landscape by introducing an ethanol-water mixture as the reaction medium. This substitution effectively mitigates the explosion hazards inherent to anhydrous methanol, as ethanol possesses a significantly higher flash point and narrower explosion limits, especially when diluted with water. Beyond safety, this solvent system demonstrates superior performance, with experimental data indicating that product yields in ethanol-water solutions consistently exceed those obtained in anhydrous methanol. The ability to conduct radical-type oxidation reactions efficiently in an aqueous environment challenges previous chemical assumptions and opens new avenues for process optimization. This approach not only enhances operator safety but also simplifies waste treatment and solvent recovery processes, aligning perfectly with the goals of a reliable p-nitrobenzoic acid supplier aiming for sustainable growth.
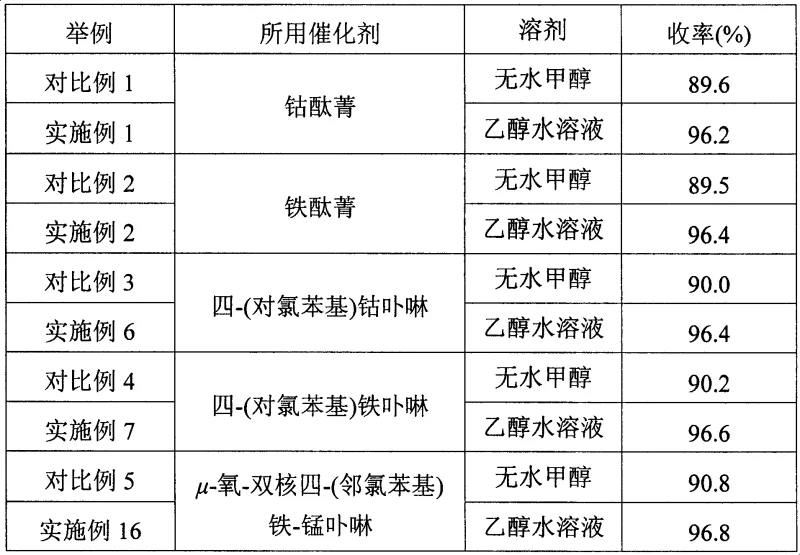
Mechanistic Insights into Biomimetic Catalytic Oxygen Oxidation
The core of this technological advancement lies in the utilization of biomimetic catalysts that mimic the activity of cytochrome P450 enzymes found in biological systems. These catalysts, specifically metal phthalocyanines and metalloporphyrins, facilitate the activation of molecular oxygen to generate reactive oxygen species capable of oxidizing the methyl group of p-nitrotoluene to a carboxyl group. The transition metal centers, which can include Iron, Cobalt, Manganese, Copper, or Zinc, play a pivotal role in electron transfer processes. The coordination environment around the metal atom, influenced by the macrocyclic ligand structure, dictates the selectivity and turnover frequency of the reaction. Understanding these mechanistic details is crucial for R&D teams looking to optimize reaction parameters such as temperature, pressure, and catalyst loading to maximize throughput while minimizing impurity formation.
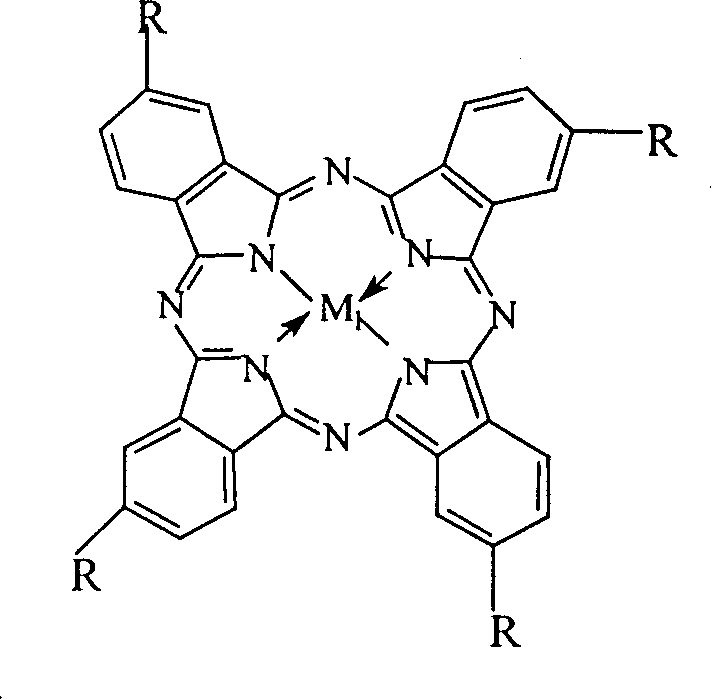
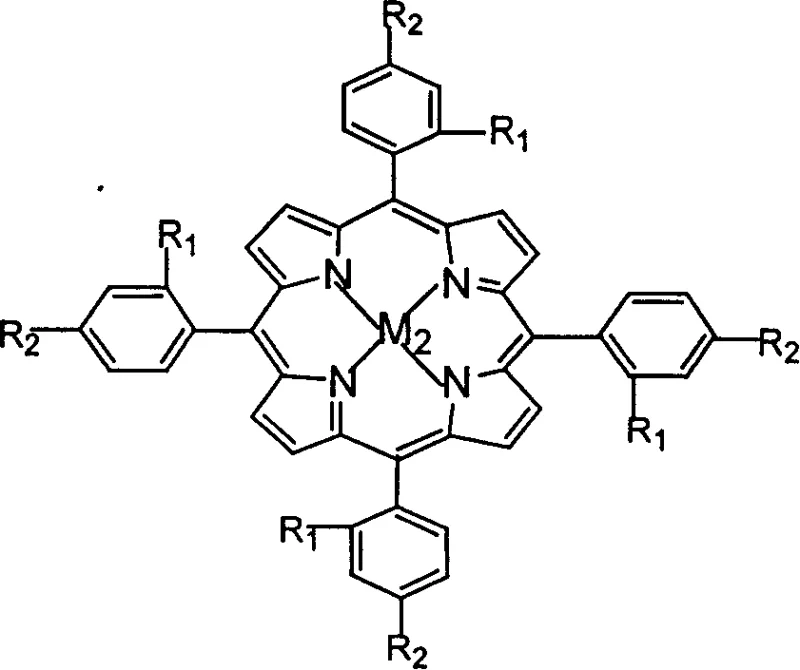
Furthermore, the specific structural variations of the catalysts, such as the presence of electron-withdrawing or electron-donating substituents on the macrocycle, can fine-tune the electronic properties of the metal center. For instance, the use of μ-oxygen binuclear metalloporphyrins offers a unique cooperative effect where two metal centers work in tandem to activate oxygen, potentially lowering the activation energy required for the oxidation step. This sophisticated catalytic design ensures that the reaction proceeds smoothly even in the presence of water, which is traditionally known to quench free radical reactions. By controlling the impurity profile through precise catalyst selection, manufacturers can achieve high-purity p-nitrobenzoic acid suitable for sensitive downstream applications in the pharmaceutical and electronic materials sectors without the need for extensive purification steps.
How to Synthesize p-Nitrobenzoic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing p-nitrobenzoic acid with high consistency and yield. The process involves charging a high-pressure reactor with p-nitrotoluene, a selected biomimetic catalyst, and a strong base dissolved in an ethanol-water mixture. The reaction is then initiated by introducing oxygen gas at controlled pressures ranging from 0.8 to 3.0 MPa while maintaining temperatures between 35°C and 55°C. This mild condition window is critical for preventing thermal degradation of the product and ensuring the stability of the catalyst. Detailed standard operating procedures regarding catalyst preparation, solvent ratios, and work-up techniques are essential for replicating these results on a commercial scale. The following guide summarizes the critical steps for implementation.
- Prepare the reaction mixture by combining p-nitrotoluene, a biomimetic catalyst (such as cobalt phthalocyanine or iron porphyrin), and a strong base in an ethanol-water solvent.
- Pressurize the reactor with oxygen gas to between 0.8 and 3.0 MPa and maintain the temperature between 35°C and 55°C for 2 to 14 hours.
- Filter the reaction mixture to remove the catalyst, neutralize with acid, and isolate the high-purity p-nitrobenzoic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ethanol-water based biomimetic oxidation process offers tangible strategic benefits beyond mere technical feasibility. The elimination of hazardous solvents like anhydrous methanol directly translates to reduced insurance premiums, lower safety compliance costs, and decreased downtime associated with safety audits. Additionally, the higher yields reported with this method mean that less raw material is wasted per unit of product, effectively lowering the cost of goods sold. The simplicity of the work-up procedure, which involves filtration and neutralization, reduces the complexity of downstream processing equipment requirements. These factors combine to create a more resilient and cost-efficient supply chain capable of meeting fluctuating market demands without compromising on delivery schedules or product quality standards.
- Cost Reduction in Manufacturing: The shift to ethanol-water solvents eliminates the need for specialized explosion-proof equipment required for anhydrous methanol, leading to substantial capital expenditure savings. Furthermore, ethanol is generally more affordable and easier to source globally than high-purity anhydrous methanol, stabilizing raw material costs. The higher reaction yields observed in this system mean that less starting material is consumed to produce the same amount of final product, directly improving the overall material efficiency and reducing waste disposal costs associated with unreacted feedstock and by-products.
- Enhanced Supply Chain Reliability: By utilizing a solvent system that is less hazardous and easier to transport, logistics become simpler and less prone to regulatory delays. Ethanol and water are ubiquitous commodities with stable supply chains, reducing the risk of shortages that can occur with specialized solvents. The robustness of the catalyst system, which tolerates aqueous conditions well, ensures consistent batch-to-batch performance, allowing production planners to forecast output with greater accuracy and reliability, thereby securing the continuity of supply for downstream customers.
- Scalability and Environmental Compliance: The process operates at relatively low temperatures and moderate pressures, making it highly scalable from pilot plants to multi-ton commercial reactors without significant engineering hurdles. The reduced toxicity of the solvent system simplifies wastewater treatment and vent gas scrubbing requirements, ensuring easier compliance with increasingly stringent environmental regulations. This green chemistry approach enhances the corporate sustainability profile, appealing to end-users who prioritize environmentally responsible sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomimetic catalytic oxidation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. They are designed to clarify the operational advantages and safety improvements offered by this novel method compared to traditional synthesis routes. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing lines or sourcing strategies.
Q: Why is ethanol-water solvent preferred over anhydrous methanol for this oxidation?
A: Ethanol-water mixtures eliminate the explosion risks associated with anhydrous methanol under pure oxygen conditions and significantly reduce toxicity hazards for operators while maintaining or improving reaction yields.
Q: What types of catalysts are effective for this biomimetic oxidation process?
A: The process utilizes transition metal complexes including metal phthalocyanines, mononuclear metalloporphyrins, and μ-oxygen binuclear metalloporphyrins containing metals like Iron, Cobalt, Manganese, or Copper.
Q: How does this method impact the purity of the final p-nitrobenzoic acid?
A: By avoiding harsh acidic conditions and utilizing selective biomimetic catalysts, the method achieves high conversion rates with minimal by-product formation, resulting in superior product purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Nitrobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced manufacturing technologies to meet the evolving needs of the global pharmaceutical and fine chemical industries. Our expertise extends to scaling diverse synthetic pathways, including complex biomimetic catalytic oxidations, from initial development stages of 100 kgs to full commercial production capacities of 100 MT annually. We are committed to maintaining stringent purity specifications through our rigorous QC labs, ensuring that every batch of p-nitrobenzoic acid meets the highest international standards. Our team of experienced chemists and engineers is dedicated to optimizing these green processes to deliver cost-effective and sustainable solutions for our partners.
We invite you to collaborate with us to explore how this innovative oxidation technology can enhance your supply chain efficiency and product quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable p-nitrobenzoic acid supplier dedicated to driving value through scientific excellence and operational integrity.
