Breakthrough Catalyst-Free Oxidation Technology for Commercial Scale-Up of Complex Nitro Compounds
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical building blocks. A significant technological advancement in this domain is documented in patent CN108238948B, which details a novel method for preparing alpha-o-nitrobenzene ethanol (α-o-nitrophenylethanol) through the direct oxidation of o-nitroethylbenzene. This innovation represents a paradigm shift from traditional reduction-based syntheses or complex catalytic oxidations to a streamlined, catalyst-free aerobic oxidation process. By leveraging molecular oxygen as the sole oxidant and sodium hydroxide as a promoter, this technology addresses long-standing challenges regarding heavy metal contamination, catalyst recovery, and environmental impact. For R&D directors and procurement strategists, understanding the nuances of this patent is crucial, as it offers a viable route to high-purity alpha-o-nitrobenzene ethanol that aligns perfectly with modern green chemistry principles and supply chain resilience goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-o-nitrophenylethanol has been plagued by significant inefficiencies and environmental burdens. The predominant industrial route involves the reduction of acetophenone derivatives, a method that often necessitates expensive starting materials and generates substantial chemical waste. Alternatively, direct oxidation methods reported in prior art, such as those utilizing silica-supported Cobalt(II) or biomimetic metalloporphyrin catalysts, have failed to gain widespread industrial traction due to severe limitations. For instance, earlier catalytic systems required harsh acidic solvents like acetic acid, leading to equipment corrosion and increased maintenance costs. Furthermore, these metal-catalyzed processes typically resulted in complex product mixtures containing unwanted ketones and carboxylic acids, with selectivity for the target alcohol often dropping below 50%. The necessity of synthesizing and purifying complex catalysts like metalloporphyrins adds a layer of operational complexity and cost that renders these methods economically unviable for large-scale cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN108238948B introduces a remarkably simple yet powerful catalytic-free system. By utilizing o-nitroethylbenzene as the substrate and molecular oxygen under basic conditions, the process achieves exceptional conversion rates and selectivity without the need for any transition metal catalysts. The reaction operates efficiently in alcohol solvents, particularly aqueous ethanol, which are non-toxic, inexpensive, and easily recoverable. This elimination of catalyst not only removes the risk of heavy metal residues in the final API intermediate but also drastically simplifies the workup procedure. The ability to achieve yields as high as 88% with conversion rates reaching 98% under moderate temperatures (25-65°C) demonstrates the robustness of this chemistry. This approach effectively bypasses the purification nightmares associated with catalyst removal, positioning it as a superior choice for commercial scale-up of complex nitro compounds.
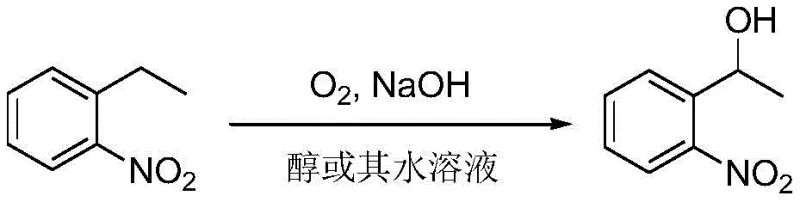
Mechanistic Insights into Base-Promoted Aerobic Oxidation
The core of this technological breakthrough lies in the unique interaction between the benzylic hydrogen of o-nitroethylbenzene and the activated oxygen species generated in the alkaline alcoholic medium. Although the process is termed "catalyst-free," the presence of sodium hydroxide plays a critical role in initiating the radical chain reaction. The base likely facilitates the formation of a benzylic radical or anion intermediate, which then reacts rapidly with dissolved molecular oxygen. This mechanism avoids the high energy barriers typically associated with uncatalyzed auto-oxidation, allowing the reaction to proceed smoothly at temperatures as low as 65°C. The absence of metal centers means there are no coordination complexes to stabilize unwanted side reactions, thereby inherently favoring the formation of the alcohol over the corresponding ketone or acid. This intrinsic selectivity is a major advantage for R&D teams focused on impurity profiling, as it minimizes the formation of hard-to-remove byproducts that often plague metal-catalyzed oxidations.
Furthermore, the choice of solvent system profoundly influences the reaction kinetics and product distribution. The patent data indicates that while pure methanol or ethanol can be used, an 80% (V/V) aqueous ethanol solution provides an optimal balance of solubility and reactivity. Water acts not just as a diluent but potentially participates in stabilizing transition states or facilitating proton transfer steps essential for the regeneration of the active species. This solvent flexibility allows manufacturers to tune the process for maximum efficiency. For example, adjusting the neutralization pH post-reaction to between 6 and 7 ensures the stability of the product during isolation. Such precise control over reaction parameters underscores the maturity of this method, making it a reliable option for reducing lead time for high-purity intermediates by minimizing the need for extensive chromatographic purification steps.
How to Synthesize Alpha-O-Nitrophenylethanol Efficiently
Implementing this synthesis route requires careful attention to pressure and temperature controls to maximize the benefits of the aerobic oxidation. The standard protocol involves charging the reactor with o-nitroethylbenzene and sodium hydroxide, followed by the addition of the alcoholic solvent. The system must be thoroughly purged with oxygen to ensure an inert atmosphere free of nitrogen before pressurization. Maintaining the oxygen pressure between 1.0 and 1.8 MPa is critical for driving the reaction to completion within a reasonable timeframe, typically around 24 hours. Detailed standardized synthetic steps see the guide below.
- Charge o-nitroethylbenzene and sodium hydroxide into a high-pressure autoclave with 80% aqueous ethanol solvent.
- Purge the system with oxygen three times, then pressurize to 1.8 MPa and heat to 65°C for 24 hours.
- Neutralize the reaction mixture to pH 6-7, remove solvent under reduced pressure, and purify via chromatography to achieve up to 88% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this catalyst-free technology are profound. The most immediate benefit is the drastic reduction in raw material costs associated with catalyst procurement. Traditional methods often rely on precious metals or complex organic ligands that are subject to volatile market pricing and supply constraints. By eliminating the catalyst entirely, this process removes a significant variable from the cost structure. Additionally, the use of ethanol and water as solvents replaces hazardous and expensive organic solvents like acetic acid or chlorinated hydrocarbons. This switch not only lowers the direct cost of goods sold but also reduces the expenses related to solvent recovery and hazardous waste disposal, contributing to substantial overall cost savings without compromising on quality or throughput.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for expensive metal scavengers and rigorous purification steps required to meet strict residual metal limits in pharmaceutical products. This simplification of the downstream processing workflow translates directly into lower operational expenditures. Furthermore, the high atom economy of using oxygen as the oxidant means that the only byproduct is water, minimizing waste treatment costs. The ability to recycle the aqueous ethanol solvent further enhances the economic viability, creating a closed-loop system that maximizes resource utilization and minimizes environmental fees.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like o-nitroethylbenzene, sodium hydroxide, and ethanol ensures a stable and resilient supply chain. Unlike specialized catalysts that may have single-source suppliers or long lead times, these raw materials are globally available in bulk quantities. This availability mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions—operating at moderate temperatures and pressures—means that the process can be easily transferred between different manufacturing sites or CDMO partners without requiring specialized high-tech equipment, ensuring consistent supply continuity for global clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in high-pressure autoclaves which are standard equipment in fine chemical plants. The use of a closed system for oxygen oxidation addresses safety concerns regarding flammability, allowing for safe operation at larger scales. From an environmental perspective, the absence of heavy metals and the use of green solvents align perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturers and enhances the sustainability profile of the final product, a key selling point for eco-conscious pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this catalyst-free method compare to traditional metalloporphyrin catalysis?
A: Unlike traditional methods requiring complex metalloporphyrin catalysts which suffer from low synthesis yields and difficult separation, this novel approach eliminates the catalyst entirely. This avoids heavy metal contamination issues and significantly simplifies the downstream purification process, resulting in higher selectivity (up to 88% yield) compared to the mixtures of alcohols, ketones, and acids produced by older methods.
Q: What are the safety implications of using pure oxygen in this oxidation process?
A: While handling pure oxygen requires caution, the patent specifies conducting the reaction in a closed autoclave system. This closed environment effectively mitigates the potential explosion hazards associated with organic solvent and oxygen mixtures found in open systems. Furthermore, operating at moderate temperatures (25-65°C) enhances process safety compared to high-temperature solvent-free methods that require heating up to 160°C.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly amenable to scale-up. It utilizes inexpensive and readily available raw materials like o-nitroethylbenzene and ethanol. The absence of expensive catalysts and the use of a recyclable aqueous ethanol solvent system drastically reduce production costs and environmental waste, making it an ideal candidate for reliable pharma intermediate supplier operations aiming for green manufacturing standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-O-Nitrobenzene Ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free oxidation technology described in CN108238948B for the production of high-value nitro intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into robust commercial reality. Our facilities are equipped with advanced high-pressure reactors capable of handling aerobic oxidations safely and efficiently. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall production costs for alpha-o-nitrobenzene ethanol and related intermediates.
