Scalable Synthesis of (13 cis, 15 trans)-Octadecadienal for Commercial Pheromone Production
The agricultural sector faces persistent challenges from defoliating pests such as the Clostera anastomosis, commonly known as the poplar moth, which causes significant economic losses and ecological damage across vast regions. To combat this without the environmental drawbacks of broad-spectrum chemical pesticides, the industry is increasingly turning to species-specific insect sex pheromones. Patent CN111943821B discloses a robust and highly efficient synthesis method for (13 cis, 15 trans)-octadecadienal, the active pheromone component for this pest. This technical breakthrough addresses critical bottlenecks in prior art by utilizing inexpensive starting materials like 12-bromo-1-dodecanol and acetylene gas, streamlining the reaction pathway into five distinct unit operations. For R&D directors and procurement specialists, this patent represents a pivotal shift towards cost-effective and scalable manufacturing of high-purity agrochemical intermediates, ensuring a stable supply chain for green pest control solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex dienal pheromones has been plagued by convoluted reaction sequences that drive up production costs and limit scalability. Previous methods, such as those referenced in background patent CN201910688701.1, often suffer from low production efficiency and complicated operational procedures that are ill-suited for industrial magnification. These conventional routes frequently rely on expensive reagents or require harsh reaction conditions that compromise safety and increase waste generation. Furthermore, the cumulative yield loss across multiple steps in traditional syntheses results in a final product that is economically unviable for large-area field applications, where tons of pheromone dispensers are required. The inability to consistently achieve high stereochemical purity in older methods also leads to reduced biological activity in the field, necessitating higher application rates and further inflating costs.
The Novel Approach
The methodology outlined in CN111943821B introduces a streamlined five-step strategy that fundamentally重构 s the production logic for (13 cis, 15 trans)-octadecadienal. By initiating the synthesis with the nucleophilic substitution of 12-bromo-1-dodecanol with acetylene, the process establishes the carbon backbone efficiently using commodity chemicals. The subsequent addition reaction with n-butyraldehyde and controlled dehydration allows for precise construction of the conjugated diene system with the requisite trans-geometry. This novel approach eliminates the need for exotic catalysts or cryogenic conditions that are difficult to maintain on a kiloton scale. The integration of a selective hydrogenation step using P-2 nickel catalyst ensures the correct cis-configuration is installed late in the sequence, minimizing isomerization risks. Overall, this route offers a direct path to high-purity products with significantly simplified post-reaction processing.
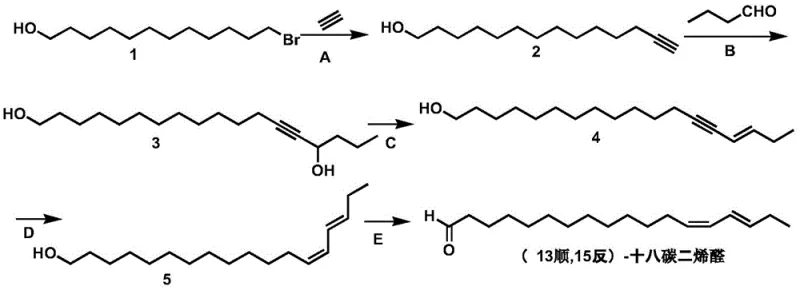
Mechanistic Insights into the Five-Step Synthetic Route
The core of this synthesis lies in the precise control of carbon-carbon bond formation and stereochemistry. Step A involves the generation of an acetylide salt using strong bases like potassium hydroxide or sodium amide in solvents such as methyl tert-butyl ether (MTBE) or THF. This acetylide attacks the primary bromide of 12-bromo-1-dodecanol, extending the chain to form 13-tetradecyn-1-ol with yields reaching approximately 90 percent. In Step B, the terminal alkyne is deprotonated using n-butyllithium at low temperatures ranging from minus 30 to 0 degrees Celsius, followed by nucleophilic attack on n-butyraldehyde to create the secondary alcohol intermediate. Step C is critical for geometry control; acid-catalyzed dehydration using trifluoroacetic acid or p-toluenesulfonic acid at 30 to 45 degrees Celsius selectively eliminates water to form the trans-alkene, yielding octadec-(15-trans)-13-yn-1-ol with high fidelity.
The final stages focus on functional group manipulation while preserving the delicate double bond geometry. Step D utilizes a P-2 type nickel catalyst or Lindlar catalyst for the semi-hydrogenation of the internal alkyne. Conducted at mild temperatures between 0 and 25 degrees Celsius, this step selectively reduces the triple bond to a cis-double bond without affecting the existing trans-alkene or the terminal hydroxyl group, achieving conversion rates near 99 percent. The final Step E employs mild oxidants such as Dess-Martin periodinane or manganese dioxide to convert the primary alcohol to the target aldehyde. This oxidation is performed at controlled temperatures to prevent over-oxidation to the carboxylic acid or isomerization of the sensitive conjugated diene system, ensuring the final product meets the stringent purity specifications required for effective pheromone activity.
How to Synthesize (13 cis, 15 trans)-Octadecadienal Efficiently
Implementing this synthesis requires strict adherence to the temperature profiles and stoichiometric ratios defined in the patent to maximize yield and purity. The process begins with the preparation of the acetylene salt, followed by sequential chain elongation and functional group transformations. Operators must ensure rigorous exclusion of moisture during the organometallic steps and precise temperature control during the dehydration and hydrogenation phases to maintain stereochemical integrity. While the general workflow is robust, specific parameters regarding solvent choice and catalyst loading are critical for reproducibility. For detailed standardized operating procedures and specific reaction conditions tailored to your facility, please refer to the technical guide below.
- Perform nucleophilic substitution of 12-bromo-1-dodecanol with acetylene salt to form 13-tetradecyn-1-ol.
- Conduct alkynylation with n-butyraldehyde followed by acid-catalyzed dehydration to establish the trans-alkene geometry.
- Execute selective cis-hydrogenation using P-2 nickel catalyst and final oxidation to yield the target aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible strategic benefits beyond mere technical feasibility. The reliance on bulk commodity chemicals like acetylene gas and long-chain bromo-alcohols significantly decouples production costs from the volatility of specialized fine chemical markets. By simplifying the reaction sequence and utilizing standard industrial solvents such as dichloromethane and MTBE, the process reduces the complexity of waste treatment and solvent recovery systems. This streamlined approach translates directly into a more resilient supply chain capable of responding rapidly to seasonal demand spikes for pest control agents without the lead times associated with complex multi-step custom syntheses.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of high-yielding reactions drastically lower the cost of goods sold. By avoiding low-yield steps that typically plague pheromone synthesis, the overall material throughput is maximized, reducing the amount of raw material required per kilogram of final product. Furthermore, the ability to perform reactions at near-ambient temperatures in several steps reduces energy consumption for heating and cooling, contributing to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: The starting materials identified in this patent are widely available from global chemical suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility supplies or environmental conditions, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining the continuous availability of pheromone dispensers needed for area-wide pest management programs.
- Scalability and Environmental Compliance: The process is explicitly designed for mass production, with workup procedures involving standard extraction and distillation techniques that scale linearly from pilot to plant scale. The reduction in reaction steps inherently minimizes the total volume of chemical waste generated, simplifying compliance with increasingly stringent environmental regulations. The high purity of the intermediate and final products also reduces the burden on downstream formulation teams, facilitating faster time-to-market for end-user products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (13 cis, 15 trans)-octadecadienal. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: What are the key advantages of this synthesis method over prior art?
A: This method utilizes cheap and readily available raw materials like acetylene gas and 12-bromo-1-dodecanol, avoiding complex multi-step sequences found in previous patents. It offers simplified operation conditions, higher overall yields, and is specifically optimized for mass production scalability.
Q: How is stereochemical control achieved in the hydrogenation step?
A: The process employs a P-2 type nickel catalyst or Lindlar catalyst under controlled temperatures (0 to 25°C). This ensures selective cis-hydrogenation of the triple bond while preserving the existing trans-double bond geometry formed during the dehydration step.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method is suitable for volume production. The reaction conditions are mild, solvents are standard industrial grades (e.g., MTBE, DCM), and the purification steps involve standard workups like washing and column chromatography which can be adapted for plant-scale operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (13 cis, 15 trans)-Octadecadienal Supplier
The synthesis of (13 cis, 15 trans)-octadecadienal represents a sophisticated intersection of organic chemistry and agricultural necessity, demanding a partner with deep technical expertise. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of pheromone intermediate delivers the biological efficacy required for effective pest control.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your trusted partner in sustainable agrochemical manufacturing.
