Advanced Industrial Synthesis of Melengestrol Acetate for Global Veterinary Markets
Advanced Industrial Synthesis of Melengestrol Acetate for Global Veterinary Markets
The global demand for high-purity veterinary pharmaceutical intermediates requires robust, scalable, and economically viable synthetic pathways. Patent CN102134266B introduces a transformative preparation method for Melengestrol Acetate, a potent progestogen widely used in livestock weight gain and estrus synchronization. This technical insight report analyzes the proprietary 6-step synthesis route which utilizes 3β-hydroxy-16β-methyl-16α,17α-epoxy pregna-5-ene-20-one as a readily available initiator. By shifting away from the arduous 16-step sequences derived from Diosgenin, this methodology offers a streamlined approach that addresses critical pain points in steroid manufacturing, including yield optimization, operational safety, and supply chain continuity. For R&D directors and procurement strategists, understanding the mechanistic advantages of this route is essential for securing a reliable melengestrol acetate supplier capable of meeting stringent regulatory and volume requirements.
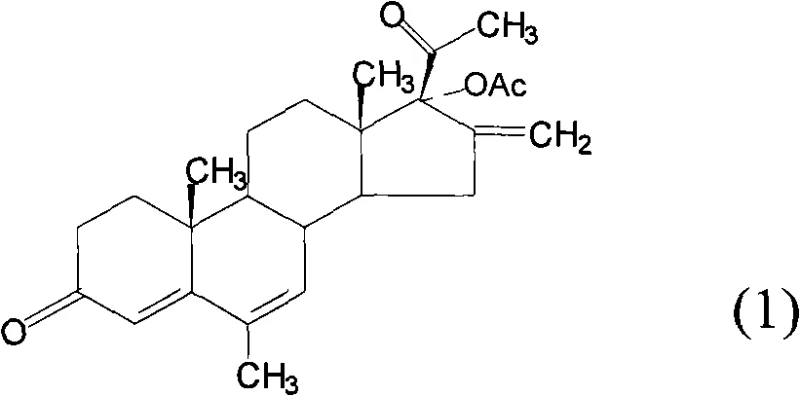
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Melengestrol Acetate has been plagued by inefficient synthetic architectures that hinder cost reduction in veterinary drug manufacturing. Early methodologies, such as those disclosed in GB886619 and US3359287, relied on initiators like 6,16-dimethyl-16-dehydroprogesterone acetate, which itself required a complex 10-step synthesis from Diosgenin. Consequently, the total process spanned approximately 16 steps, leading to substantial cumulative yield losses and reliance on scarce raw materials. Furthermore, alternative routes utilizing 4-AD (Androstadienedione) as a starting point, while shorter, introduced severe operational hazards including the use of toxic reagents like red precipitate and dimethyl oxalate, alongside harsh cryogenic conditions that complicated reactor design and increased energy consumption.
The Novel Approach
In stark contrast, the methodology defined in CN102134266B leverages a commercially accessible epoxy-pregnane starter to achieve the target molecule in merely six high-yielding transformations. This route eliminates the need for hazardous sealed-tube reactions previously required for D-ring construction, replacing them with standard reflux operations in open vessels. The strategic selection of reaction conditions allows for simplified post-processing, such as direct crystallization and filtration, rather than complex chromatographic separations. By integrating the ring-opening of the 16,17-epoxy group with simultaneous esterification, the process achieves significant step economy. This architectural efficiency not only boosts the overall recovery rate to approximately 18.47% compared to the 13.09% of older methods but also drastically simplifies the commercial scale-up of complex steroid intermediates.
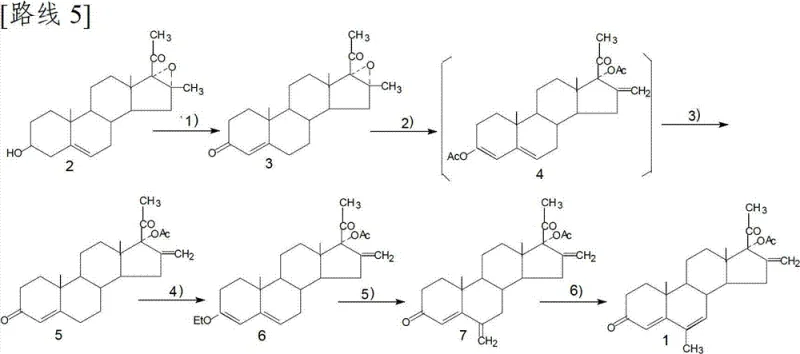
Mechanistic Insights into Steroid Functionalization and Translocation
The core of this synthetic strategy lies in the precise manipulation of the steroid nucleus through Oppenauer oxidation and selective catalytic translocation. The initial conversion of the 3β-hydroxyl group to a 3-ketone is achieved using aluminum isopropoxide in toluene, a classic yet highly effective hydride transfer mechanism that ensures the formation of the conjugated 4-ene-3-one system essential for biological activity. Subsequent steps involve the acid-catalyzed opening of the 16α,17α-epoxide ring, which proceeds with high regioselectivity to install the critical 17α-acetoxy group while generating the 16-methylene exocyclic double bond. This tandem transformation is pivotal, as it constructs the bioactive D-ring configuration in a single operational unit, minimizing the exposure of sensitive intermediates to degradative conditions.
Furthermore, the final translocation of the 6-methylene group to the 6-methyl position represents a masterclass in impurity control. Traditional hydrogenation methods often suffer from over-reduction, saturating the desirable Δ4 or Δ6 double bonds. This patent specifies the use of a palladium/lime carbonate (Pd/CaCO3) catalyst system, which exhibits superior chemoselectivity. This specific catalyst formulation suppresses the reduction of the conjugated diene system while facilitating the migration of the exocyclic methylene. For quality assurance teams, this mechanistic nuance is vital, as it inherently limits the formation of saturated by-products, thereby reducing the load on final recrystallization steps and ensuring the delivery of high-purity API intermediates that meet rigorous pharmacopeial standards.
How to Synthesize Melengestrol Acetate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process begins with the oxidation of the starting material in anhydrous toluene, requiring careful moisture control to maintain catalyst activity. Following the formation of the tri-enone intermediate, the subsequent acetylation and hydrolysis steps utilize common industrial solvents like acetic acid and ethanol, facilitating easy solvent recovery and recycling. The Mannich reaction and elimination sequence are conducted at moderate temperatures, avoiding thermal degradation.
- Perform Oppenauer oxidation on the 3-hydroxyl group of the starting epoxy-pregnane using aluminum isopropoxide and cyclohexanone to form the 3-keto-4-ene intermediate.
- Execute ring-opening of the 16,17-epoxy group followed by simultaneous esterification of the 3-carbonyl enol using acetic anhydride and p-toluenesulfonic acid.
- Hydrolyze the 3-acetate group under acidic conditions to restore the 3-ketone functionality while retaining the 17α-acetoxy-16-methylene structure.
- Conduct etherification of the 3-carbonyl enol using triethyl orthoformate to protect the A-ring prior to methylene introduction.
- Perform a Mannich reaction followed by elimination to introduce the 6-methylene group, creating the 6,16-dimethylene intermediate.
- Finalize the synthesis via translocation of the 6-methylene group to the 6-methyl position using a palladium/lime carbonate catalyst system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the supply chain; by utilizing a starting material that is commercially available from multiple vendors, manufacturers can mitigate the risks associated with single-source bottlenecks common in Diosgenin-derived pathways. The reduction in step count from sixteen to six directly correlates to a significant reduction in manufacturing costs, as it lowers the consumption of solvents, reagents, and labor hours per kilogram of output. Additionally, the elimination of toxic reagents like dimethyl oxalate reduces the environmental compliance burden and waste disposal costs, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The streamlined six-step process inherently lowers the cost of goods sold (COGS) by minimizing unit operations. Each skipped step represents savings in reactor time, utility consumption, and quality control testing. Moreover, the use of robust catalysts like Pd/CaCO3, which can be recovered and reused, further optimizes the economic profile of the synthesis. The avoidance of specialized high-pressure equipment for sealed-tube reactions also reduces capital expenditure requirements for production facilities, making the technology accessible for rapid deployment.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-step initiators derived from natural products like Diosgenin often leads to supply volatility due to agricultural fluctuations. By shifting to a semi-synthetic route starting from stable, commercially sourced epoxy-pregnanes, the supply chain becomes more resilient and predictable. The robustness of the reaction conditions, which tolerate standard industrial-grade solvents and do not require extreme cryogenic cooling, ensures consistent batch-to-batch production schedules, effectively reducing lead time for high-purity veterinary hormone intermediates.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as reflux, filtration, and recrystallization. The absence of hazardous sealed-tube steps removes a major barrier to scaling from kilograms to metric tons. Furthermore, the improved selectivity of the catalytic steps results in fewer by-products, simplifying wastewater treatment and solid waste management. This environmental efficiency not only lowers operational costs but also ensures compliance with increasingly strict global environmental regulations regarding pharmaceutical manufacturing emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental embodiments provided in the patent documentation, offering clarity on yield expectations, safety protocols, and purity profiles. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: How does this new synthesis route improve upon traditional Diosgenin-based methods?
A: Traditional routes starting from Diosgenin often require up to 16 steps with difficult-to-source initiators like 6,16-dimethyl-16-dehydroprogesterone acetate. This novel patent CN102134266B reduces the sequence to just 6 steps from a commercially available epoxy-pregnane starter, significantly shortening lead times and reducing cumulative yield losses associated with long synthetic chains.
Q: What are the safety advantages regarding reaction conditions compared to previous art?
A: Previous methods, such as those described in EP104054, often necessitated hazardous sealed-tube reactions at high temperatures, posing significant risks for industrial scale-up. The disclosed method operates in open vessels under reflux conditions with standard solvents like toluene and ethanol, eliminating the need for high-pressure equipment and simplifying post-processing operations.
Q: How is product purity maintained during the critical 6-methylene translocation step?
A: The process utilizes a specific palladium/lime carbonate (Pd/CaCO3) catalyst system rather than standard Pd/C. This selective catalyst minimizes the unwanted reduction of the Δ6 double bond, a common side reaction in prior art, thereby ensuring higher chemical purity and reducing the burden on downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Melengestrol Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of veterinary pharmaceuticals depends on a partnership grounded in technical excellence and supply reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering Melengestrol Acetate with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for animal health applications. Our infrastructure is designed to support the complex chemistry of steroid synthesis, providing a secure and compliant source for your critical raw materials.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to serve as your long-term strategic partner in the global veterinary market.
