Advanced Synthesis of 3-Hydroxymethyltetrahydrofuran via Gamma-Butyrolactone for Commercial Scale-Up
Advanced Synthesis of 3-Hydroxymethyltetrahydrofuran via Gamma-Butyrolactone for Commercial Scale-Up
The chemical landscape for producing key agrochemical intermediates is constantly evolving, driven by the need for safer, more cost-effective, and environmentally sustainable processes. Patent CN102276559A introduces a groundbreaking methodology for the synthesis of 3-hydroxymethyltetrahydrofuran, a critical building block for third-generation neonicotinoid insecticides like dinotefuran. This innovative approach shifts the paradigm from traditional, hazardous multi-step sequences to a streamlined protocol centered on gamma-butyrolactone. By leveraging advanced Lewis acid catalysis and optimized reduction strategies, this technology addresses long-standing inefficiencies in yield and safety. For global supply chains, this represents a significant opportunity to secure a reliable agrochemical intermediate supplier capable of delivering high-purity materials with reduced environmental impact. The following analysis dissects the technical merits and commercial implications of this proprietary synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxymethyltetrahydrofuran has relied on cumbersome pathways that pose significant operational and economic challenges. As illustrated in the prior art referenced in the patent, such as WO2005065689, the conventional route typically initiates with diethyl malonate and ethyl chloroacetate. This initial condensation step often suffers from mediocre yields, reported around 64%, which immediately impacts the overall process efficiency. Furthermore, the subsequent reduction of the resulting 2-ethoxyacyl-diethyl succinate requires a massive excess of reducing agents, specifically sodium borohydride, in a ratio as high as 1:6. This stoichiometric imbalance not only drives up raw material costs but also generates substantial quantities of hydrogen gas, creating severe safety hazards in a manufacturing environment. Additionally, the final cyclization step often necessitates harsh acidic dehydration conditions that produce complex impurity profiles, frequently requiring resource-intensive column chromatography for purification.
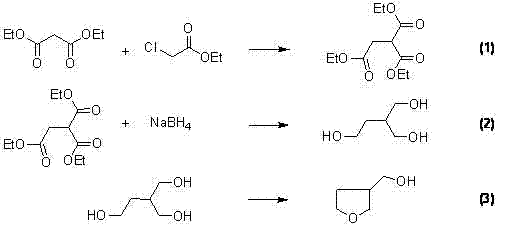
The Novel Approach
In stark contrast, the novel methodology disclosed in CN102276559A utilizes gamma-butyrolactone as the primary starting material, offering a fundamentally more efficient architectural framework for the molecule. This new strategy bypasses the hazardous hydrogen evolution associated with the malonate route by employing a direct reduction mechanism or a controlled ring-opening followed by recyclization. The process begins with the alpha-substitution of gamma-butyrolactone using strong bases and carbonyl-containing compounds, achieving yields exceeding 90% in optimized examples. This high-efficiency entry point sets the stage for a robust downstream process. By eliminating the need for excessive reducing agents and avoiding the dangerous gas evolution of the legacy method, this approach drastically simplifies the operational workflow. The result is a synthesis pathway that is not only chemically superior but also inherently safer and more economically viable for large-scale production facilities.
Mechanistic Insights into Lewis Acid-Catalyzed Reduction
The core innovation of this technology lies in the sophisticated application of Lewis acid catalysis during the reduction phase. In the direct reduction pathway, the alpha-substituted gamma-butyrolactone is treated with a metal borohydride, such as sodium borohydride, in the presence of a catalytic amount of Boron Trifluoride (BF3) or Aluminum Chloride (AlCl3). The Lewis acid activates the carbonyl group of the lactone, facilitating a highly selective hydride attack that preserves the cyclic structure while reducing the ester functionality to a hydroxymethyl group. This reaction proceeds under mild thermal conditions, typically ranging from -5°C to 50°C, which minimizes side reactions and thermal degradation. The mechanistic precision allows for a molar ratio of substrate to reducing agent close to 1:1.5, a dramatic improvement over the 1:6 ratio required in conventional methods. This efficiency translates directly into reduced waste generation and lower consumption of expensive reagents.
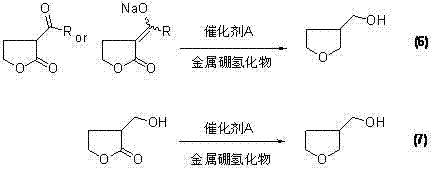
Furthermore, the process demonstrates remarkable versatility through an alternative ring-opening pathway. In this variation, the alpha-substituted intermediate undergoes reductive ring-opening to form 2-hydroxymethyl-1,4-butanediol, utilizing metal catalysts like Zinc Chloride or Titanium Tetrachloride. This linear diol is subsequently cyclized under acidic conditions to reform the tetrahydrofuran ring. This dual-pathway capability provides manufacturers with flexibility to optimize for specific equipment constraints or raw material availability. Crucially, the impurity profile in both pathways is significantly cleaner than that of the diethyl malonate route. The avoidance of harsh dehydration conditions in the direct route, or the use of optimized polyphosphoric acid in the cyclization route, ensures that the final product requires only standard fractional distillation for purification, eliminating the need for costly and slow column chromatography steps.
How to Synthesize 3-Hydroxymethyltetrahydrofuran Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the novel catalytic system. The process generally involves two main stages: the initial alpha-functionalization of the lactone ring and the subsequent reductive transformation. Operators must maintain strict temperature controls during the addition of strong bases to prevent polymerization or decomposition of the sensitive intermediates. The choice of solvent, typically toluene or tetrahydrofuran, plays a critical role in solubilizing the intermediates and managing the exotherm during the reduction phase. Detailed standard operating procedures regarding the order of addition for the Lewis acid and borohydride are essential to ensure safety and reproducibility. For a comprehensive guide on the specific experimental conditions and workup procedures, please refer to the standardized synthesis steps outlined below.
- Perform alpha-substitution of gamma-butyrolactone using a strong base and an ester compound or formaldehyde to generate alpha-substituted gamma-butyrolactone.
- Conduct a direct reduction of the alpha-substituted intermediate using a metal borohydride and a Lewis acid catalyst like BF3 or AlCl3.
- Neutralize the reaction mixture with acid, filter, remove solvents, and purify via fractional distillation to obtain high-purity 3-hydroxymethyltetrahydrofuran.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this gamma-butyrolactone-based synthesis offers profound advantages in cost structure and supply reliability. The shift away from diethyl malonate to gamma-butyrolactone leverages a commodity chemical that is widely available and significantly less expensive on a global scale. This raw material substitution alone drives a substantial reduction in the Bill of Materials (BOM) cost. Moreover, the drastic decrease in the consumption of sodium borohydride—from a six-fold excess to near-stoichiometric amounts—further compounds these savings. For procurement managers, this means a more stable cost base that is less susceptible to volatility in the pricing of specialized reagents. The simplified purification process also reduces the demand for silica gel and organic solvents used in chromatography, contributing to lower operational expenditures and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The elimination of hazardous hydrogen gas evolution removes the need for specialized venting and explosion-proof equipment, leading to significant capital expenditure savings. Additionally, the high yield of the initial substitution step ensures that raw material utilization is maximized, minimizing waste disposal costs. The overall process efficiency allows for cost reduction in agrochemical intermediates manufacturing by streamlining the number of unit operations required. By reducing the reliance on expensive purification techniques like column chromatography, the production cost per kilogram is drastically lowered, enhancing the margin potential for downstream insecticide producers.
- Enhanced Supply Chain Reliability: Utilizing gamma-butyrolactone, a bulk chemical with a robust global supply chain, mitigates the risk of raw material shortages that can plague specialty ester supplies. The simplified process flow, characterized by fewer reaction steps and easier workup procedures, enhances the throughput capacity of existing manufacturing assets. This increased efficiency translates to shorter lead times and the ability to respond more rapidly to fluctuations in market demand. For supply chain heads, this reliability is crucial for maintaining continuous production of finished insecticide formulations without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex agrochemical intermediates, with reaction conditions that are easily manageable in large reactors. The reduction in wastewater generation and the avoidance of toxic byproducts align with increasingly stringent environmental regulations. This green chemistry profile facilitates easier permitting and reduces the liability associated with hazardous waste management. The ability to scale from pilot batches to multi-ton production without significant process re-engineering ensures a smooth transition from R&D to full commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their specific production needs. The answers highlight the practical benefits regarding yield optimization, safety protocols, and purification strategies that distinguish this method from legacy technologies.
Q: What are the primary advantages of the gamma-butyrolactone route over the diethyl malonate method?
A: The gamma-butyrolactone route significantly reduces raw material costs and eliminates the dangerous evolution of large volumes of hydrogen gas associated with the traditional diethyl malonate reduction, while also shortening the synthetic sequence.
Q: What catalysts are utilized in the novel reduction step?
A: The process employs Lewis acid catalysts such as Boron Trifluoride (BF3) or Aluminum Chloride (AlCl3) in conjunction with alkali metal borohydrides to achieve high selectivity and yield.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method is designed for industrial production, featuring simple operation, readily available raw materials, and reduced wastewater generation compared to conventional chromatographic purification methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxymethyltetrahydrofuran Supplier
The technological advancements detailed in patent CN102276559A underscore the immense potential for optimizing the production of high-purity 3-hydroxymethyltetrahydrofuran. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory innovations into robust commercial realities. As a premier CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest industry standards for agrochemical applications.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your dinotefuran supply chain. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you secure not just a supplier, but a strategic ally dedicated to driving efficiency and innovation in your agrochemical manufacturing operations.
